Abstract
Ferroptosis is an iron-dependent form of regulated cell death, driven by the extensive peroxidation of cellular membrane phospholipids, particularly those enriched with oxidation-sensitive polyunsaturated fatty acids. Given its role in diverse pathologies, ferroptosis inhibition represents a compelling therapeutic target. Among the strategies being explored, modulating cellular membrane lipid composition through exogenous supplementation with less oxidizable fatty acids, such as monounsaturated fatty acids, has gained significant attention. Nevertheless, the influence of endogenous regulators on membrane lipid dynamics and ferroptosis susceptibility is not yet fully elucidated and represents a fertile frontier for discovery. While the gut microbiota is well established as a systemic regulator of host physiology, its potential role in modulating membrane lipid composition and ferroptosis susceptibility remains largely unexplored. This Perspective opens by examining the study by Zhang et al., which suggests that bacterial extracellular vesicles (BEVs) from the gut commensal Lactobacillus amylovorus deliver oleic acid to the mammary gland. This mechanism suppresses ferroptosis and helps sustain lactation in mice under oxidative stress. The work provides a proof-of-concept for BEVs as endogenous lipid delivery vectors that may modulate ferroptosis susceptibility across different organs. Building on these findings, this Perspective critically evaluates the conceptual advance represented by Zhang et al. and integrates it with the broader literature and future scientific opportunities. Specifically, the Perspective dissects the mechanistic underpinnings of this pathway within the context of extracellular vesicle biology and inter-organ lipid trafficking. It also maps the unresolved questions poised to shape the future of the field and examines the key translational hurdles that must be overcome to harness BEV-mediated lipid delivery for therapeutic benefit.
Graphical Abstract
Keywords
1. Introduction
Ferroptosis is a regulated form of cell death driven by iron-dependent lipid peroxidation[1]. Over the past decade, its causal role in diverse pathological conditions, from kidney injury to neurodegeneration, has become firmly established, positioning its inhibition as a compelling therapeutic target[2,3]. A central strategy to suppress ferroptosis involves modulating the lipid composition of cell membranes. Phospholipids containing polyunsaturated fatty acids (PUFAs) are primary peroxidation substrates and potent drivers of ferroptotic death[4]. Conversely, exogenous monounsaturated fatty acids (MUFAs), such as oleic acid, exert a protective effect by incorporating into membranes. This was first established by studies in cultured cells, showing that exogenous MUFAs suppress ferroptosis by displacing oxidizable PUFAs to reduce lipid peroxidation[5]. Later research corroborated this mechanism in vivo, confirming that exogenous MUFAs can also inhibit ferroptosis in living animals[6,7]. While this principle is well-documented for dietary or administered lipids, the endogenous systems that dynamically regulate membrane lipid unsaturation and ferroptosis susceptibility remain an active and not-yet-fully-understood area of investigation.
This gap in knowledge intersects with a broader paradigm in host physiology: the systemic influence of the gut microbiota. Beyond producing soluble metabolites, gut bacteria communicate with distant organs via bacterial extracellular vesicles (BEVs)[8]. Within the broader framework of human health, BEVs have emerged as key mediators of gut homeostasis, facilitating non-contact communication between the microbiota and host cells. Enclosed by a lipid bilayer, these nanoparticles serve as vehicles for the delivery of diverse bioactive molecules (including proteins, nucleic acids, low-molecular-weight metabolites, and lipids) across the intestinal barrier. This process, referred to as BEV-mediated transport, enables the translocation of microbial cargo to host tissues, thereby modulating both local intestinal function and systemic physiological processes[9].
A recent work by Zhang et al.[10] suggests that BEVs from Lactobacillus amylovorus deliver oleic acid to mammary epithelial cells, inhibiting ferroptosis and sustaining proper milk production and lactation in mice. This study points to a mechanistic explanation for the gut-mammary axis and, more broadly, offers a proof-of-concept for BEVs as endogenous, targeted lipid carriers capable of altering cell fate by modulating ferroptosis susceptibility.
This Perspective critically evaluates this conceptual advance, arguing that the study by Zhang et al.[10] represents a significant emerging paradigm, although its findings must be contextualized within the broader landscape of inter-organ lipid trafficking and extracellular vesicle biology.
2. BEVs in the Landscape of Inter-Organ Lipid Trafficking
To appreciate the conceptual advance offered by Zhang et al.[10], it is essential to position their findings within the established framework of inter-organ lipid transport. Mammalian systems have evolved sophisticated mechanisms for lipid distribution, primarily through lipoprotein particles such as low-density lipoproteins (LDL) and high-density lipoproteins (HDL), which transport cholesterol, triglycerides, and phospholipids between tissues via the circulation[11]. These lipoproteins are recognized by specific receptors (e.g., LDL receptor, scavenger receptor class B type I) that mediate cellular uptake and lipid unloading. More recently, mammalian extracellular vesicles (EVs), including exosomes and microvesicles derived from host cells, have been recognized as additional lipid carriers capable of transferring not only proteins and nucleic acids but also bioactive lipids between cells[12]. Complementing this paradigm, BEVs derived from the gut microbiota have emerged as important mediators of host physiology, capable of delivering complex lipid cargos that influence metabolism and immune function[13]. However, the quantitative contribution of both host-derived and bacterial EVs to the delivery of specific lipid species, as well as their resulting impact on host physiology and systemic lipid homeostasis, is only beginning to be appreciated as a biologically relevant axis of inter-organ communication.
A recent study[14] showed that BEVs interact with host cells through membrane fusion, endocytosis, or receptor recognition, processes that enable them to dynamically influence host physiology. For Gram-positive bacteria, membrane fusion is a key mechanism for delivering cargo molecules, including lipids. BEVs preferentially fuse with liquid-ordered regions of host cell membranes, altering membrane fluidity, dipole potential, and compressibility. Fusion efficiency is modulated by lipid composition (e.g., phosphatidylcholine and C16 acyl chains promote fusion, while cholesterol inhibits it), with implications for bacterial manipulation of host cellular machinery[14]. These mechanistic insights suggest that bacteria may actively regulate host physiology by modulating membrane biophysical properties through BEV fusion and lipid delivery. Despite this progress in understanding BEV-host interactions, no study had yet established a physiological role for BEV-delivered lipids in regulating specific cell fate decisions, such as ferroptosis.
What distinguishes the work by Zhang et al.[10] is the provision of experimental support for the concept that BEV-mediated lipid delivery serves a physiological, organ-protective function. The authors show that BEVs from a commensal bacterium (Limosilactobacillus amylovorus) are enriched in oleic acid, and that this MUFA constitutes the primary bioactive cargo. By directly treating mice under oxidative stress with L. amylovorus-derived BEVs, the authors observed that these vesicles trafficked to the mammary gland and markedly improved mammary function, as evidenced by reduced ferroptosis markers (e.g., malondialdehyde [MDA], a lipid peroxidation end-product), preserved mitochondrial integrity, and enhanced lactation outcomes. These findings significantly extend existing knowledge by showing that lipids carried by BEVs can reach a distal organ (the mammary gland) and exert a protective effect by suppressing ferroptosis, likely through the modulation of target membrane lipid composition. Nevertheless, the novelty of this paradigm should be appropriately qualified: the core principle that exogenous MUFAs protect against ferroptosis by displacing membrane PUFAs was already established[5,15]; the conceptual advance lies in identifying an endogenous, microbiota-derived delivery system that exploits this principle for inter-organ communication. Furthermore, while the proposed displacement of membrane PUFAs by BEV-derived oleic acid remains hypothetical (a point discussed later in this Perspective), it represents a potentially pivotal mechanism that warrants further investigation.
3. Microbial Regulation of Mammary Gland Ferroptosis and Lactation: From Sows to Mice
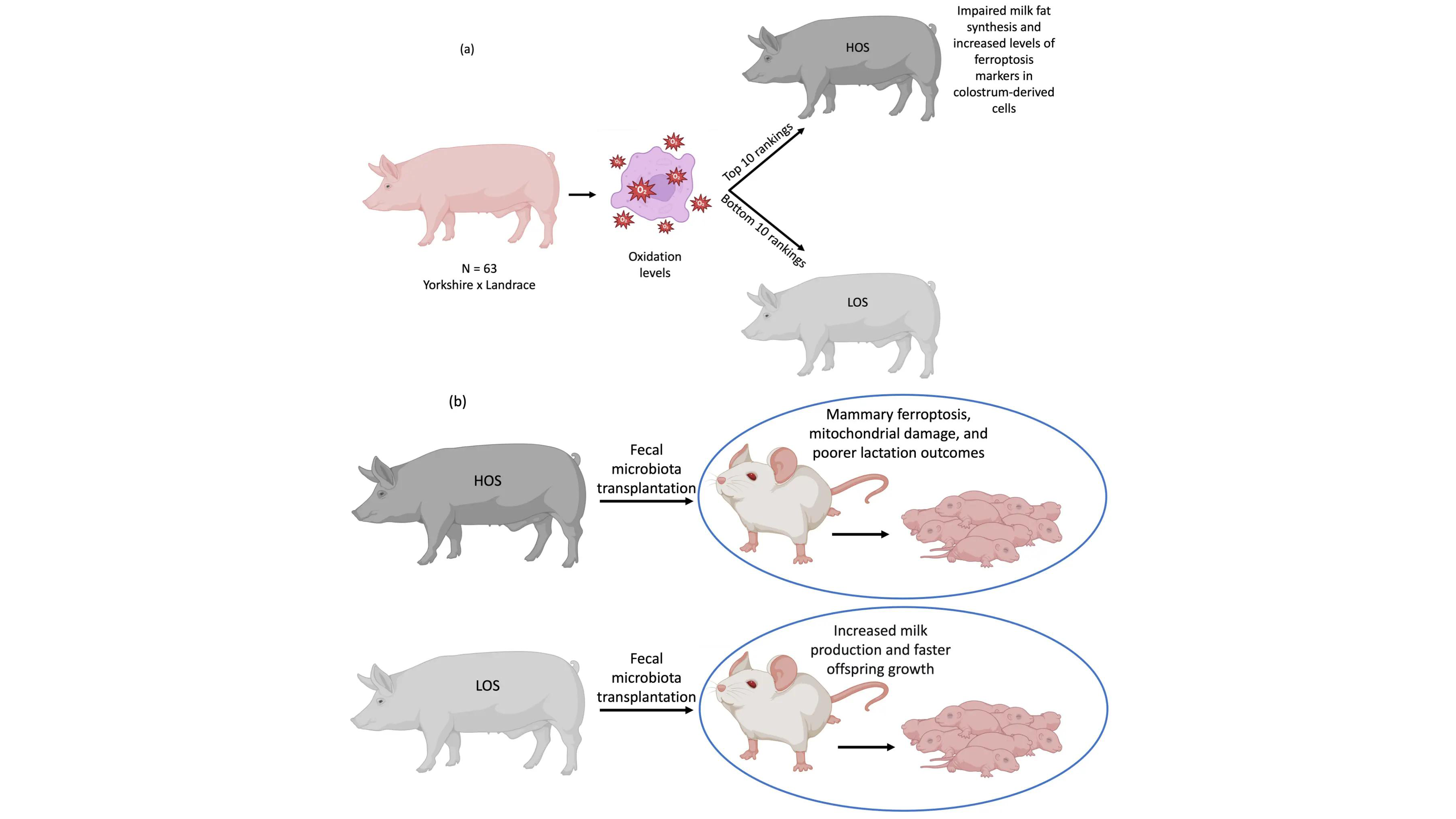
Zhang et al.[10] adopted a translational approach, initiating their investigation in sows (a physiologically relevant large-animal model for human mammary development and lactation) before moving to mechanistic studies in mice and cell culture. By categorizing sows based on oxidative stress levels, designating them as high oxidative stress (HOS) and low oxidative stress (LOS) groups (Figure 1a), they identified that the HOS group presented impaired milk fat synthesis and elevated ferroptosis markers in colostrum-derived cells, including increased PTGS2 (prostaglandin-endoperoxide synthase 2, a transcriptional marker of ferroptosis) and MDA, and decreased levels of the anti-ferroptosis proteins glutathione peroxidase 4 and ferritin heavy chain. These results suggested a foundational phenotype linking systemic oxidative stress to mammary dysfunction through ferroptosis.
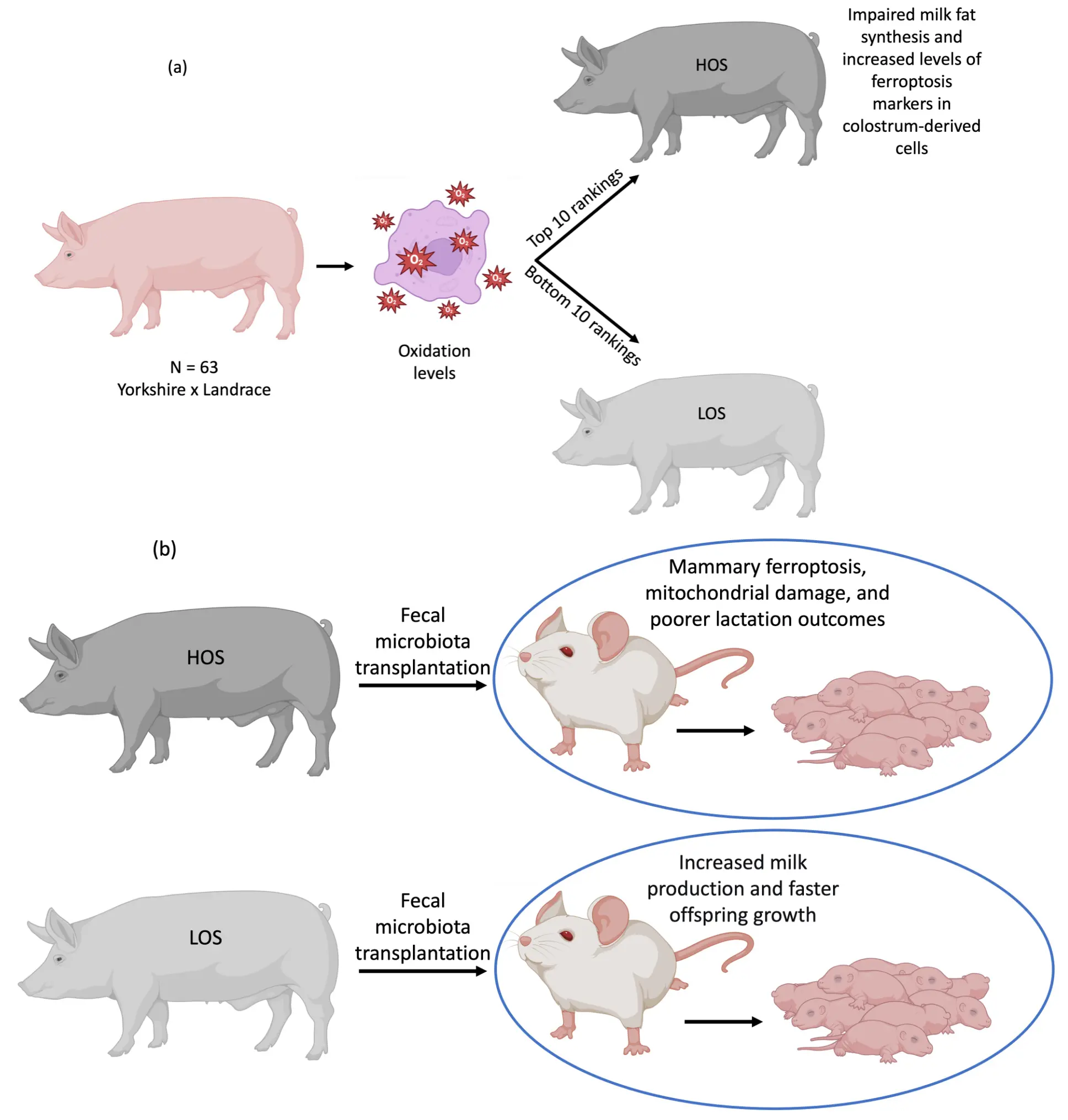
Figure 1. (a) Basis for experimental group formation in sows. Sixty-three post-farrowing sows were assessed for systemic oxidative stress using the OSI. From this assessment, the ten animals displaying the highest OSI values formed the HOS group, while the ten with the lowest OSI comprised the LOS group. Compared to the LOS group, HOS sows showed impaired milk fat synthesis and elevated ferroptosis markers in colostrum-derived cells; (b) FMT in mice. Donor fecal microbiota collected from HOS and LOS sows was transferred to recipient mice. Dams receiving HOS-derived FMT exhibited increased mammary ferroptosis markers and poorer lactation performance. Their pups showed significantly reduced body weight and size compared to pups from dams receiving LOS-derived FMT. Created with BioRender. OSI: oxidative stress index; HOS: high oxidative stress; LOS: low oxidative stress; FMT: fecal microbiota transplantation.
Fecal microbiota transplantation (Figure 1b) from HOS and LOS sows to recipient mice successfully recapitulated the phenotype: mice receiving HOS microbiota displayed heightened mammary ferroptosis, mitochondrial damage, and poorer lactation outcomes (as evidenced by reduced pup weight gain), whereas LOS microbiota conferred protection. This cross-species validation is methodologically significant, as it demonstrates that the microbiota from sows with different oxidative stress phenotypes is sufficient to transfer the mammary ferroptosis phenotype to mice, suggesting a causal role for gut microbes in regulating this process.
Through systematic 16S rRNA sequencing and species-level screening, Lactobacillus amylovorus was identified as a keystone species enriched in the protective LOS microbiota. To assess functional relevance, the authors established an in vitro oxidative stress model in HC11 mammary epithelial cells using hydrogen peroxide (H2O2). Screening cell-free supernatants from five Lactobacillus species enriched in the LOS group (L. amylovorus, L. johnsonii, L. reuteri, L. plantarum, and L. salivarius) revealed that L. amylovorus supernatant exhibited the strongest antioxidant and anti-ferroptotic activity, significantly reducing free radicals and lipid peroxides.
Crucially, BEVs derived from L. amylovorus were identified as the primary functional vectors. Purified BEVs, when administered to mice under oxidative stress, localized to the mammary gland (as shown by fluorescent labeling and imaging) and markedly attenuated ferroptosis, preserved mitochondrial integrity, and enhanced milk fat synthesis and secretion. To establish causality, the authors inhibited bacterial vesicle biogenesis using GW4869, a compound originally characterized as an inhibitor of neutral sphingomyelinase that has been widely used to suppress exosome biogenesis in mammalian cells[16]. However, its efficacy and specificity in inhibiting BEV biogenesis in Gram-positive bacteria warrant careful consideration. While GW4869 treatment reduced BEV production by L. amylovorus and abolished the protective effects, the potential for off-target effects on bacterial physiology or other secretion pathways cannot be excluded. Complementary approaches that specifically disrupt BEV biogenesis would strengthen causal inference in future studies. Nevertheless, within the context of the current study, the combination of BEV purification, characterization, and inhibition experiments provides convergent evidence supporting BEVs as essential mediators of the observed inter-organ protection.
4. Lipid-Centric Mechanism: Oleic Acid as the Primary Bioactive Cargo
A pivotal mechanistic insight from Zhang et al.[10] is the identification of lipids, specifically MUFAs, as the bioactive cargo within L. amylovorus BEVs. To determine which biomolecular component drives the protective effects, the authors exposed BEVs to enzymatic digestion with Proteinase K, DNase I, or RNase A prior to evaluating their capacity to suppress ferroptosis in HC11 cells under oxidative challenge. Degradation of nucleic acids or proteins did not diminish the protective capacity of the BEVs, shifting the focus to their lipid components. When BEV molecules were separated into hydrophilic and lipophilic components, only the lipophilic fraction retained the capacity to protect HC11 cells from ferroptosis, pointing to lipids as the primary functional mediators. Lipidomic analysis showed that MUFAs constituted nearly half of the BEV fatty acid profile, with oleic acid as the predominant species.
While these experiments point to lipids as the primary functional moiety, it is important to acknowledge certain caveats. Certain proteins or small metabolites may resist enzymatic degradation. Moreover, such molecules could co-partition into the lipophilic fraction during separation, potentially contributing to the observed protective effects even after protease, nuclease, or lipophilic extraction steps. Therefore, while the lipid-centric model is the most parsimonious explanation, synergistic or contributory roles of other components cannot be entirely ruled out.
Zhang et al.[10] demonstrated that exogenous supplementation of oleic acid in mammary epithelial cells recapitulated the BEV phenotype: it reduced lipid peroxidation and reactive oxygen species and enhanced cellular resilience to ferroptosis without altering ferroptosis-related gene expression. These findings align with established evidence that oleic acid protects against ferroptosis by directly altering membrane lipid composition, specifically by displacing oxidation-prone PUFAs from cell membranes, thereby increasing ferroptosis resistance[5,7,15], rather than by inducing antioxidant or anti-ferroptotic gene programs.
The hypothesis that BEV-delivered oleic acid incorporates into membrane phospholipids and displaces oxidation-prone PUFAs is supported by prior literature on exogenous MUFA supplementation. Magtanong and colleagues demonstrated in cultured cells that oleic acid supplementation increases MUFA-containing phospholipids at the expense of PUFA-containing species, reducing the availability of peroxidation substrates without altering the expression of ferroptosis machinery[5]. However, it is important to explicitly distinguish between demonstrated mechanisms and speculative extensions in the context of BEV-delivered lipids. Zhang et al.[10] directly demonstrated that: (i) L. amylovorus BEVs are enriched in oleic acid, (ii) oleic acid supplementation in vitro recapitulates BEV-mediated ferroptosis protection, and (iii) BEV-delivered lipids reach the mammary gland. The specific mechanism of membrane PUFA displacement by BEV-delivered oleic acid, while a logical and well-supported hypothesis, remains to be directly demonstrated through lipidomic analysis of membrane phospholipids in BEV-treated mammary epithelial cells in vivo.
Integrating these findings into the broader ferroptosis framework reveals multiple points of potential interaction. Membrane phospholipid remodeling is a dynamic process regulated by enzymes that determine fatty acyl composition. Acyl-CoA synthetase long-chain family member 4 (ACSL4) activates PUFAs for incorporation into membrane phospholipids and is a critical pro-ferroptotic factor[17]. Lysophosphatidylcholine acyltransferase 3 (LPCAT3) incorporates activated PUFAs into phospholipids, further promoting ferroptosis susceptibility[18]. Conversely, stearoyl-CoA desaturase 1 (SCD1) generates MUFAs from saturated fatty acids and has been implicated in ferroptosis resistance[19]. Whether BEV-delivered oleic acid influences the activity or substrate preference of these enzymes, or whether it simply competes with PUFAs for incorporation into the phospholipid pool through mass action, remains to be determined. Future studies should examine whether BEV treatment alters ACSL4-dependent PUFA activation, LPCAT3-mediated phospholipid remodeling, or SCD1 expression and/or activity in target tissues.
An important unresolved question is the precise molecular form in which oleic acid is delivered. It is unknown whether BEVs carry free fatty acids, esterified within complex bacterial phospholipids, or as part of other lipid species. The metabolic fate of these lipids upon uptake, whether they are directly incorporated into host membranes or first processed by host enzymes, will determine how they interface with the ferroptosis machinery and represents a key area for future investigation.
5. Evidence Map for BEV-Mediated Inter-Organ Communication and Ferroptosis Inhibition
To provide a structured assessment of the current evidence strength for each step in the proposed BEV-mediated inter-organ communication pathway, an evidence map is presented in Table 1. This framework distinguishes between steps that are established (directly demonstrated by Zhang et al. with rigorous methodology), supported (indirect evidence from Zhang et al. or consistent with prior studies), and hypothetical (proposed model lacking direct evidence). This grading is essential for identifying priorities for future investigation.
| Pathway Step | Evidence Grade | Supporting Observations | Knowledge Gaps |
| Biogenesis of L. amylovorus BEVs | Supported | BEVs isolated and characterized from bacterial cultures; GW4869 inhibition reduces BEV production and abrogates protection | GW4869 specificity for bacterial vesicle biogenesis requires validation |
| Cargo Packaging (Oleic Acid Enrichment) | Established | Lipidomic analysis demonstrates MUFA enrichment (48% of total fatty acids) with oleic acid as predominant species | Mechanisms governing selective lipid packaging into BEVs unknown |
| Systemic Transit | Supported | Fluorescently labeled BEVs detected in circulation after oral administration | Quantitative pharmacokinetics (half-life, clearance, stability) undefined; BEV integrity during transit not directly assessed |
| Organ Targeting (Mammary Gland) | Established | Fluorescent BEV signal detected in mammary tissue after oral administration | Targeting mechanisms (passive vs. receptor-mediated) unknown; specificity for mammary versus other organs not systematically examined |
| Cellular Uptake | Supported | BEV-associated signal localized to mammary epithelial cells | Uptake mechanism (endocytosis, fusion, receptor-mediated) undefined |
| Lipid Delivery and Membrane Remodeling | Hypothetical | Oleic acid supplementation recapitulates protection (although only in vitro experiments with cultured cells were performed); prior literature establishes MUFA-mediated PUFA displacement | Direct evidence for BEV-delivered oleic acid incorporation into membrane phospholipids lacking; lipidomic analysis of BEV-treated cell membranes needed |
| Ferroptosis Inhibition | Established | BEV treatment reduces lipid peroxidation, preserves mitochondrial integrity, and improves functional outcomes (lactation) | Molecular mechanism linking lipid delivery to ferroptosis suppression (PUFA displacement vs. other effects) not definitively established |
BEV: bacterial extracellular vesicle; MUFA: monounsaturated fatty acid; PUFA: polyunsaturated fatty acid.
6. Beyond the Gut-Mammary Axis: Expanding the BEV-Ferroptosis Paradigm
The study by Zhang et al.[10] raises the possibility that BEV-mediated lipid delivery may represent a general mechanism for inter-organ regulation of ferroptosis susceptibility. If gut-derived BEVs can transport bioactive lipids to the mammary gland, it is plausible that similar pathways operate for other ferroptosis-sensitive tissues.
Emerging evidence, though preliminary, supports this broader perspective. Several studies have reported that BEVs can reach multiple organs[20,21]. The lipid composition of BEVs varies substantially across bacterial species and growth conditions[22], suggesting that different microbial communities may produce vesicles with distinct fatty acid profiles and, consequently, varying capacities to modulate host ferroptosis susceptibility. This raises the possibility that the gut microbiota collectively generates a diverse repertoire of lipid-rich vesicles that differentially influence ferroptosis sensitivity across organs, depending on BEV tropism and organ-specific lipid metabolism.
However, a “dark side” analysis is equally important. If BEVs can deliver protective MUFAs, they might also deliver pro-ferroptotic lipids under certain conditions. Bacterial species that produce BEVs enriched in PUFAs could potentially promote ferroptosis in host tissues. Moreover, systemic delivery of MUFAs via BEVs could have context-dependent deleterious effects. While MUFA-induced ferroptosis resistance is beneficial in conditions of oxidative stress (e.g., ischemia-reperfusion injury, neurodegenerative diseases), it could theoretically promote tumorigenesis or cancer progression by suppressing ferroptosis-mediated tumor suppression[23]. Many cancer cells upregulate MUFA synthesis via SCD1 to evade ferroptosis[24], and exogenous MUFA delivery might inadvertently support this adaptive mechanism. Whether sustained or excessive BEV-mediated MUFA delivery could influence cancer risk, metabolic reprogramming, or other ferroptosis-related pathologies remains entirely unknown and represents an important consideration for therapeutic development.
7. Methodological Considerations and Study Limitations
Some methodological considerations and knowledge gaps in the study by Zhang et al.[10] warrant discussion:
First, the efficacy and specificity of GW4869 for inhibiting bacterial EV biogenesis require careful consideration. GW4869 is a well-characterized inhibitor of neutral sphingomyelinase 2 (nSMase2), an enzyme involved in ceramide biosynthesis and exosome biogenesis in mammalian cells[16]. Its use in bacterial systems is less well validated. While Zhang et al.[10] demonstrate that GW4869 reduces BEV production by L. amylovorus and abrogates protective effects, the molecular target in Gram-positive bacteria remains unclear; there is no direct evidence confirming that Lactobacillus species possess canonical nSMase2 homologs. Thus, as already discussed, potential off-target effects on bacterial growth, membrane integrity, or other secretion pathways cannot be excluded. Complementary approaches that specifically disrupt BEV biogenesis would strengthen causal inference in future studies.
Second, while the authors demonstrate that BEVs reach the mammary gland, the mechanisms governing organotropism remain undefined. BEV targeting could reflect passive accumulation based on vascular permeability (the mammary gland undergoes substantial remodeling and increased vascularization during lactation), active receptor-mediated uptake (mammary epithelial cells may express receptors recognizing BEV surface components), or simply the high blood flow to lactating tissue. Despite the preliminary evidence indicating that BEVs can reach multiple organs[20,21], systematic biodistribution studies examining BEV accumulation in multiple organs under various physiological conditions are needed.
Third, the quantitative contribution of BEV-delivered oleic acid to mammary gland lipid pools remains uncharacterized. While fluorescent labeling demonstrates BEV presence, it does not quantify lipid transfer. Stable isotope-labeled oleic acid incorporated into BEVs could definitively track its incorporation into mammary membrane phospholipids and distinguish BEV-derived from endogenous or dietary lipid sources.
Fourth, the translational extrapolation from sow and mouse lactation models to human disease contexts requires careful qualification. Lactation involves unique physiological adaptations, including profound metabolic changes, increased mammary blood flow, and specialized lipid secretion mechanisms. These events may not generalize to other organs or pathological conditions. While the gut-mammary axis is biologically interesting, its relevance to, for example, neurodegenerative diseases or acute kidney injury remains speculative until direct evidence for BEV-mediated ferroptosis protection in those contexts emerges.
8. Conclusions and Future Perspectives
The study by Zhang et al.[10] expands our understanding of gut-organ communication by unveiling a vesicle-mediated lipid delivery route that regulates ferroptosis and tissue function. It provides a proof-of-concept that commensal bacteria can influence host lipid metabolism and cellular stress responses through BEVs, highlighting a conceptual advance for microbiota-host interaction. By framing their findings within the broader context of inter-organ lipid trafficking, we can appreciate both the conceptual advance and the substantial knowledge gaps that remain.
Several priority questions should guide future research:
(i) What are the molecular mechanisms governing lipid packaging into BEVs? Elucidating the mechanisms behind selective oleic acid packaging into L. amylovorus BEVs may provide clues about how bacterial vesicles acquire their lipid signatures.
(ii) What determines BEV organotropism? Identifying receptors or targeting signals that direct BEVs to specific tissues would be transformative for developing organ-selective lipid delivery systems.
(iii) Is BEV-mediated ferroptosis modulation a general phenomenon? Systematic examination of BEVs from diverse bacterial species for their capacity to modulate ferroptosis in multiple organs (kidney, liver, brain, heart) is needed.
(iv) What are the context-dependent consequences of BEV-mediated MUFA delivery? Studies examining both protective effects (injury models) and potential deleterious effects (cancer models, metabolic disease) are essential.
(v) Can BEVs be engineered for therapeutic lipid delivery? Proof-of-concept studies using BEVs loaded with specific lipid species to modulate ferroptosis in disease models would establish translational potential.
8.1 Translational challenges and opportunities
The therapeutic potential of BEV-mediated lipid delivery, as exemplified by the protective role of L. amylovorus-derived vesicles loaded with MUFAs in a murine model of mammary gland homeostasis[10], is considerable. This concept opens a new paradigm for modulating host physiology, specifically the susceptibility of cells to ferroptosis, via microbiota-derived vectors. However, while these initial findings in preclinical models are promising, their relevance to human disease remains to be established. Translating this concept to human applications presents substantial obstacles that require critical analysis. The current lack of fundamental evidence underscores the need for a major future research effort to advance the field and enable such a translation. This evidence includes: (i) a definitive demonstration of a similar gut-mammary axis in humans, (ii) the identification of human commensal bacteria capable of producing analogous protective BEVs, and (iii) robust validation that the proposed lipid-based protective mechanism operates effectively within the complex and dynamic milieu of human tissues and pathologies. Addressing these fundamental gaps is essential to guide the rigorous investigation of the following translational challenges:
(i) Cargo Heterogeneity and Safety. BEVs derived from commensal bacteria like L. amylovorus carry a complex mixture of lipids, proteins, and nucleic acids beyond the putative therapeutic cargo. Consequently, the safety profile of chronic or long-term BEV administration, encompassing potential immunogenicity, toxicity, and off-target effects, remains critically undefined and represents a key knowledge gap requiring scientific investigation.
(ii) Immunogenicity and Toxicity. As bacterial products, BEVs are inherently immunogenic. Although Lactobacillus species are generally regarded as safe, their vesicles may still elicit innate immune responses, particularly upon repeated systemic administration. Potential consequences include Toll-like receptor activation and induction of pro-inflammatory cytokines, and the formation of anti-vector antibodies[25], all of which could compromise therapeutic efficacy or lead to adverse effects. Accordingly, a comprehensive assessment of BEV immunogenicity and toxicity represents a critical priority prior to clinical translation.
(iii) Biodistribution and Targeting Control. The organotropism of L. amylovorus BEVs for mammary tissue[10] may not generalize to other therapeutic targets. Uncontrolled biodistribution could lead to off-target effects, including unintended ferroptosis modulation in healthy tissues. Engineering BEVs for targeted delivery (through surface display of targeting ligands, receptor-specific modifications, or hybrid vesicle technologies) represents a major technical challenge requiring substantial investment.
(iv) Dosing and Route of Administration. The optimal route (oral, systemic, local), dosing regimen, and formulation for BEV-based therapies are unknown. Oral administration offers convenience but faces gastrointestinal degradation and variable absorption. Systemic administration provides more controlled delivery but raises immunogenicity and safety concerns.
Despite these considerable challenges (ranging from the fundamental lack of human data to unresolved complexities regarding safety and optimal administration routes), the conceptual advance provided by Zhang et al.[10] opens exciting new directions. If BEVs can be engineered to deliver specific lipid cargos to defined organs, they could enable precision modulation of ferroptosis susceptibility in ways not possible with systemic lipid supplementation. The coming years will likely see intensive efforts to: (i) elucidate the basic biology of BEV-mediated lipid trafficking; (ii) map the landscape of BEV lipid composition across the human microbiota; and (iii) develop the tools and platforms needed to translate these insights into therapies. The journey from a mechanistic discovery in a lactation model to human therapeutics is long and uncertain, but the paradigm of microbiota-derived vesicles as inter-organ lipid carriers represents a fundamentally new way of thinking about host-microbiota interactions and their impact on cell fate decisions.
Acknowledgements
DeepSeek AI assistant was used solely for language polishing. After using this tool, the author reviewed and edited the content as needed and take full responsibility for the final content.
Authors contribution
The author contributed solely to the article.
Conflicts of interest
The author declares no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by a grant from Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina (Process FAPESC/TO/2024TR002498; Process FAPESC/TO/2023TR000318) and National Council for Scientific and Technological Development (CNPq-Brazil; grant 303121/2022-0).
Copyright
© The Author(s) 2026.
References
-
1. Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: An iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060-1072.[DOI]
-
2. Stockwell BR. Ferroptosis turns 10: Emerging mechanisms, physiological functions, and therapeutic applications. Cell. 2022;185(14):2401-2421.[DOI]
-
3. Berndt C, Alborzinia H, Amen VS, Ayton S, Barayeu U, Bartelt A, et al. Ferroptosis in health and disease. Redox Biol. 2024;75:103211.[DOI]
-
6. Perez MA, Clostio AJ, Houston IR, Ruiz J, Magtanong L, Dixon SJ, et al. Ether lipid deficiency disrupts lipid homeostasis leading to ferroptosis sensitivity. PLoS Genet. 2022;18(9):e1010436.[DOI]
-
7. Mann J, Reznik E, Santer M, Fongheiser MA, Smith N, Hirschhorn T, et al. Ferroptosis inhibition by oleic acid mitigates iron-overload-induced injury. Cell Chem Biol. 2024;31(2):249-264.e7.[DOI]
-
8. Kang SG, Choi YY, Mo SJ, Kim TH, Ha JH, Hong DK, et al. Effect of gut microbiome-derived metabolites and extracellular vesicles on hepatocyte functions in a gut-liver axis chip. Nano Convergence. 2023;10(1):5.[DOI]
-
9. Wang Y, Luo X, Xiang X, Hao C, Ma D. Roles of bacterial extracellular vesicles in systemic diseases. Front Microbiol. 2023;14:1258860.[DOI]
-
11. Feingold KR. Lipid and lipoprotein metabolism. Endocrinol Metab Clin N Am. 2022;51(3):437-458.[DOI]
-
13. Huang J, Yu Y, Feng Z, Yin Y, Liu Y, Liu X, et al. Cross - Kingdom dialogue of microbial messengers: Multi - target regulatory mechanisms and therapeutic strategies of gut microbiota - derived extracellular vesicles in metabolic diseases. Int J Nanomed. 2025;20:12573-12591.[DOI]
-
17. Doll S, Freitas FP, Shah R, Aldrovandi M, da Silva MC, Ingold I, et al. FSP1 is a glutathione-independent ferroptosis suppressor. Nature. 2019;575:693-698.[DOI]
-
20. Choi Y, Kwon Y, Kim DK, Jeon J, Jang SC, Wang T, et al. Gut microbe-derived extracellular vesicles induce insulin resistance, thereby impairing glucose metabolism in skeletal muscle. Sci Rep. 2015;5:15878.[DOI]
-
21. Chen X, Li Q, Zhang W, Xu Y, Nie X, Wang X, et al. Akkermansia muciniphila-derived extracellular vesicles alleviate colitis-related cognitive impairment via tryptophan metabolic reprogramming of the gut‒brain axis. Gut Microbes. 2026;18:2611546.[DOI]
-
22. Liu Y, Defourny KAY, Smid EJ, Abee T. Gram-positive bacterial extracellular vesicles and their impact on health and disease. Front Microbiol. 2018;9:1502.[DOI]
-
24. Yi J, Zhu J, Wu J, Thompson CB, Jiang X. Oncogenic activation of PI3K-AKT-mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis. Proc Natl Acad Sci U S A. 2020;117(49):31189-31197.[DOI]
-
25. Effah CY, Ding X, Drokow EK, Li X, Tong R, Sun T. Bacteria-derived extracellular vesicles: Endogenous roles, therapeutic potentials and their biomimetics for the treatment and prevention of sepsis. Front Immunol. 2024;15:1296061.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite