-
Chiral Chemistry (CC, Online ISSN 3106-8405) is an open-access quarterly journal dedicated to publication and dissemination of high-impact, original, and leading research in the field of chiral chemistry and technologies. The journal provides a platform for pioneering theoretical, experimental, and applied studies, promoting fundamental understanding, advancements, and applied innovations across a broad range of scientific and industrial applications. more >
Articles
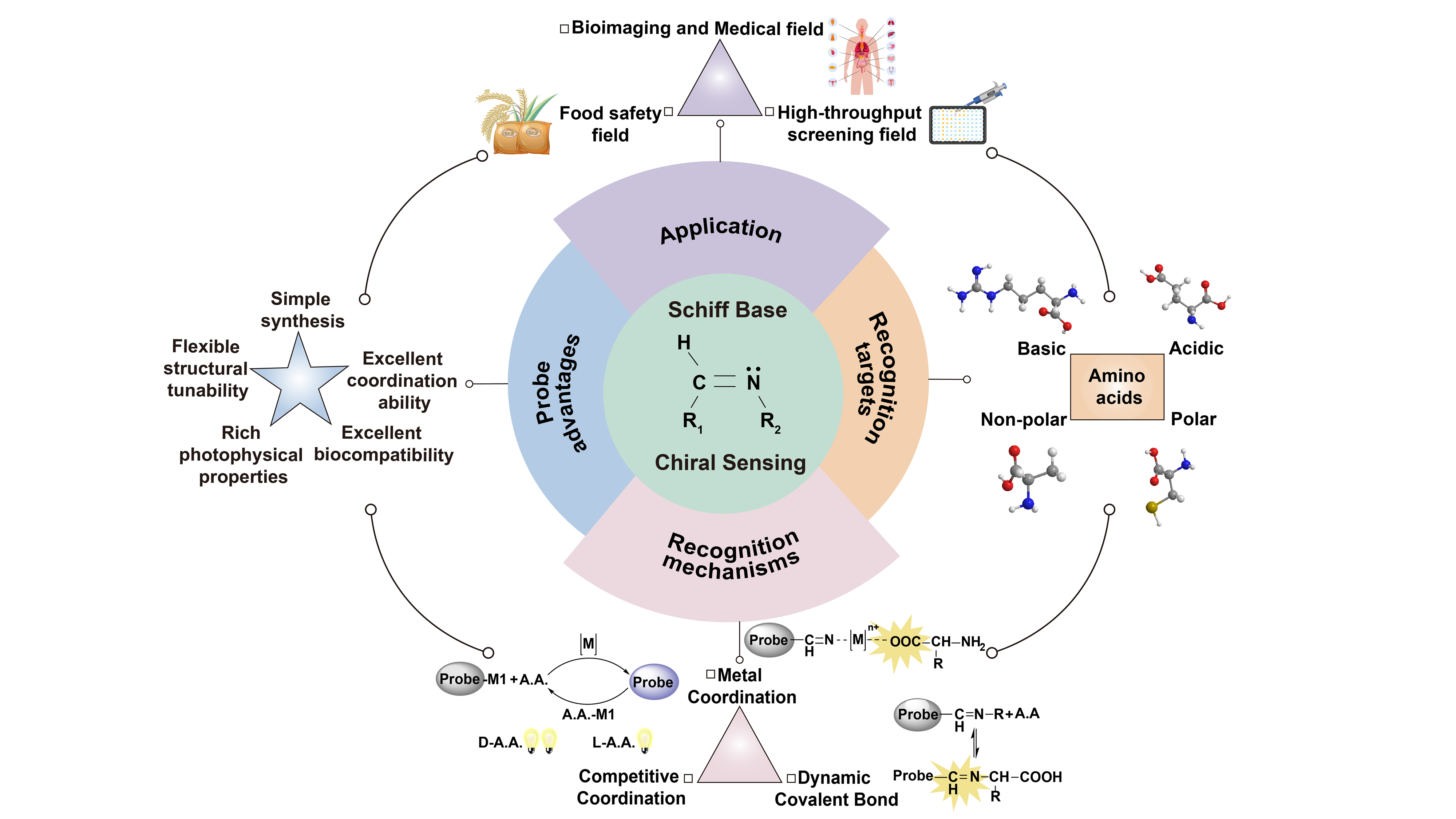
Schiff base fluorescent probes for enantioselective recognition of amino acids
-
Amino acids are typical naturally occurring chiral compounds, whose enantiomeric pairs show distinct biological functions and markedly different application prospects. The precise chiral recognition of amino acid enantiomers is of great significance ...
MoreAmino acids are typical naturally occurring chiral compounds, whose enantiomeric pairs show distinct biological functions and markedly different application prospects. The precise chiral recognition of amino acid enantiomers is of great significance in diverse fields including life sciences, medical diagnosis, and pharmaceutical development. Over the past decade, chiral fluorescent probes toward amino acids have gained considerable attention for enantiomer recognition. Among these probes, Schiff base chiral fluorescent probes have emerged as a powerful class of sensors for achieving high enantioselective recognition of amino acids, owing to their advantages of facile synthesis, tunable structure, facile functionalization, and excellent photophysical and coordination properties. This review systematically summarizes the research progress of such probes in the enantiomer recognition of different types of amino acids, including acidic, basic, non-polar, and polar amino acids. It focuses on discussing probe design strategies, enantioselective recognition, interaction mechanisms, and application developments. In addition, this paper also highlights and outlines the major challenges existing in this field and the difficult issues that need to be addressed in the future.
Less -
Jinyu Wei, ... Shuangxi Gu
-
DOI: https://doi.org/10.70401/cc.2026.0027 - May 15, 2026
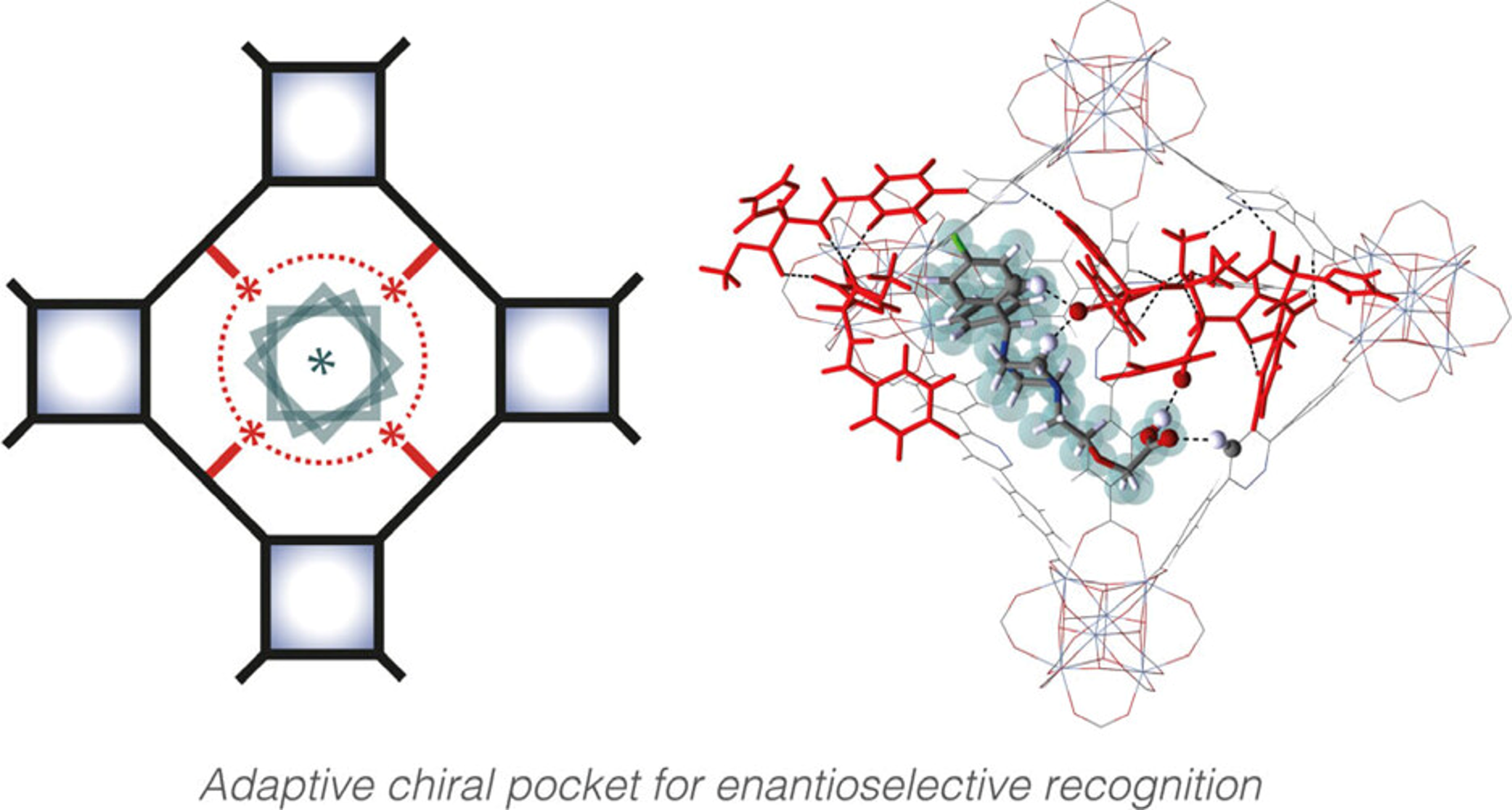
Click reactions in a robust metal-organic framework enable adaptive chiral recognition
-
Wenqiang Zhang, Wei Gong
-
DOI: https://doi.org/10.70401/cc.2026.0028 - May 15, 2026
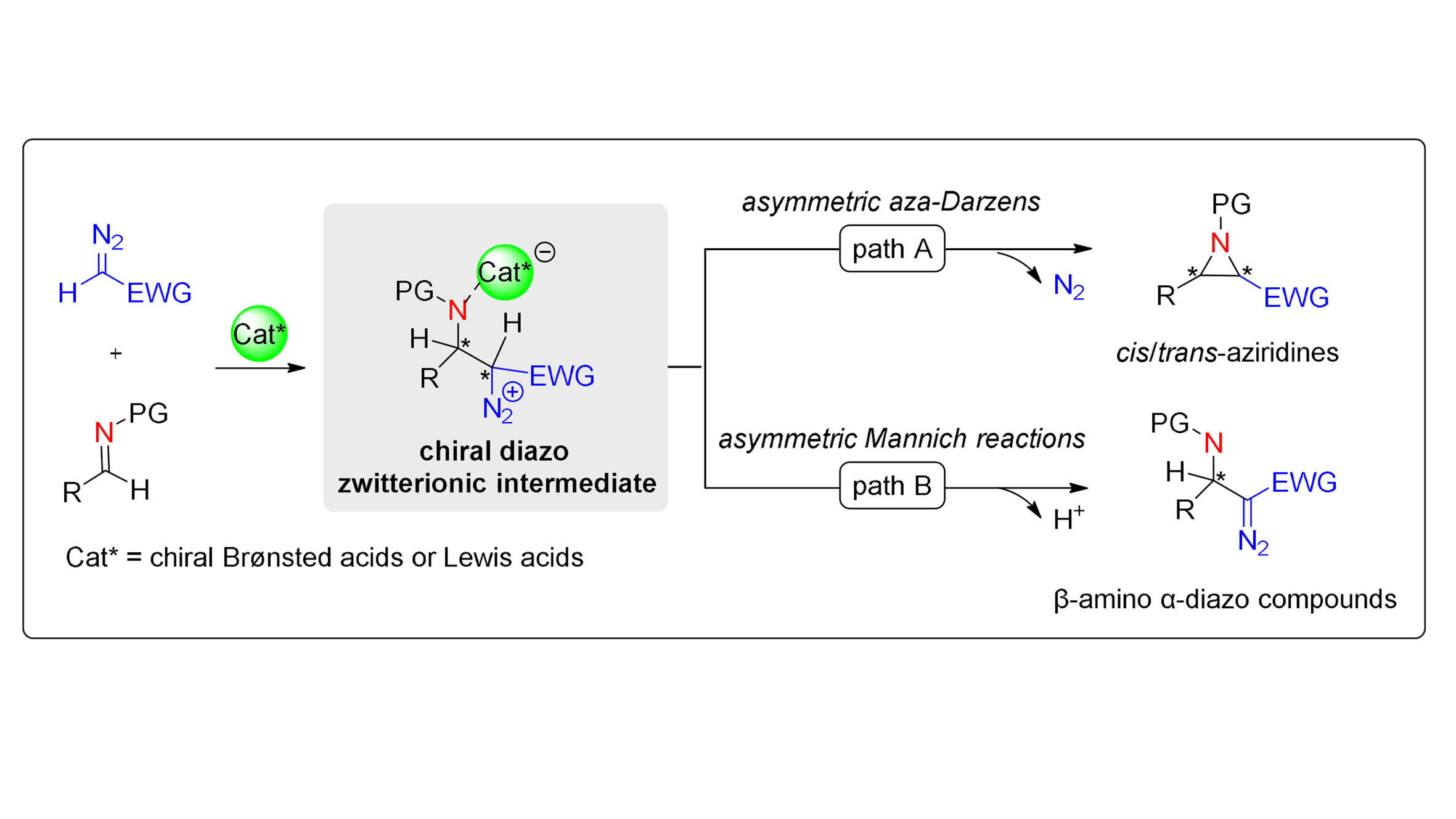
Transition-metal-free chiral Brønsted/Lewis acid-catalyzed asymmetric diazo nucleophile-imine reactions
-
Over the past few decades, transition-metal-free chiral Brønsted acid or Lewis
Moreacid-catalyzed asymmetric reactions between diazo nucleophiles and imines have emerged as a pivotal strategy for constructing chiral ...Over the past few decades, transition-metal-free chiral Brønsted acid or Lewis
Lessacid-catalyzed asymmetric reactions between diazo nucleophiles and imines have emerged as a pivotal strategy for constructing chiral aziridines or β-amino α-diazo derivatives, achieving remarkable synthetic progress. This review summarizes the research advances in asymmetric aza-Darzens reactions and asymmetric Mannich-type reactions of diazo nucleophiles with imines. It encompasses diverse catalytic systems, including chiral boroxinates, chiral dicarboxylic acids, chiral phosphoric acids, chiral N-triflylphosphoramides, and chiral Lewis acids, with an emphasis on the effects of substrate and catalyst on chemoselectivity. Remaining challenges and future perspectives are presented in this review. -
Pei-Run Xie, ... Jun Xuan
-
DOI: https://doi.org/10.70401/cc.2026.0026 - May 12, 2026
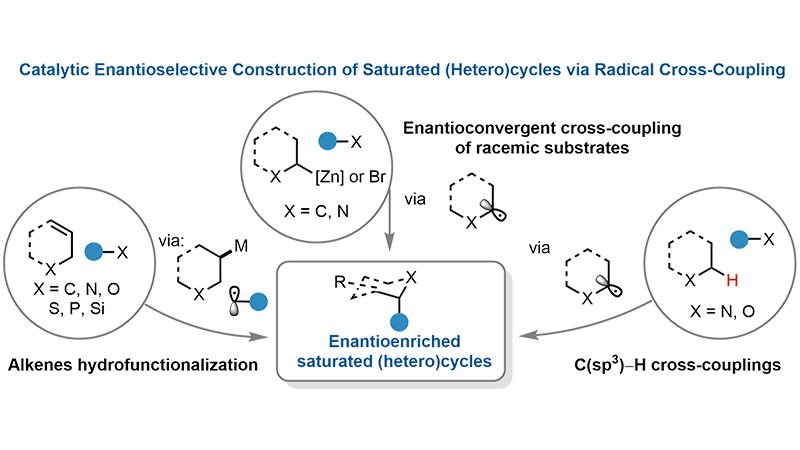
Catalytic enantioselective construction of saturated (hetero)cycles via radical cross-coupling
-
Chiral saturated (hetero)cyclic scaffolds constitute privileged structural motifs in natural products, pharmaceuticals, and bioactive molecules, where their three-dimensional architectures often confer superior physicochemical and pharmacological ...
MoreChiral saturated (hetero)cyclic scaffolds constitute privileged structural motifs in natural products, pharmaceuticals, and bioactive molecules, where their three-dimensional architectures often confer superior physicochemical and pharmacological properties compared to planar aromatic systems. Despite their importance, general and modular strategies for the enantioselective synthesis of these C(sp3)-rich frameworks remain limited. In recent years, enantioselective radical cross-coupling (RCC) has emerged as a transformative strategy that addresses these limitations by enabling the direct coupling of abundant radical precursors, including alkenes, carboxylic acids, alkyl halides, and C(sp3)–H bonds, in a largely functional-group-agnostic manner. By leveraging transition-metal catalysis with chiral ligands, these methods achieve exquisite stereocontrol while providing modular access to structurally diverse saturated (hetero)cycles under mild conditions. This review comprehensively surveys recent advances in the catalytic enantioselective construction of saturated (hetero)cycles via RCC, organized into three major strategic classes: enantioconvergent cross-coupling of racemic substrates, enantioselective hydrofunctionalization of (hetero)cyclic alkenes, and direct enantioselective C(sp3)–H RCC. We critically analyze synthetic achievements, mechanistic insights, and substrate scope encompassing diverse ring sizes (from three-membered to bridged polycyclic systems) and heteroatom incorporation (N, O, S, P, Si, Ge), and discuss persistent challenges including macrocycle synthesis, C(sp3)–C(sp3) bond formation, multicomponent couplings, catalyst diversification beyond nickel and cobalt, expansion of C(sp3)–H functionalization scope, and mechanistic elucidation. This review provides a comprehensive understanding of the current state of the field and highlights promising directions for future development in accessing enantioenriched (hetero)cyclic architectures with broad applications in pharmaceutical discovery, materials science, and chemical biology.
Less -
Xinlong Luo, ... Haohua Huo
-
DOI: https://doi.org/10.70401/cc.2026.0025 - May 07, 2026
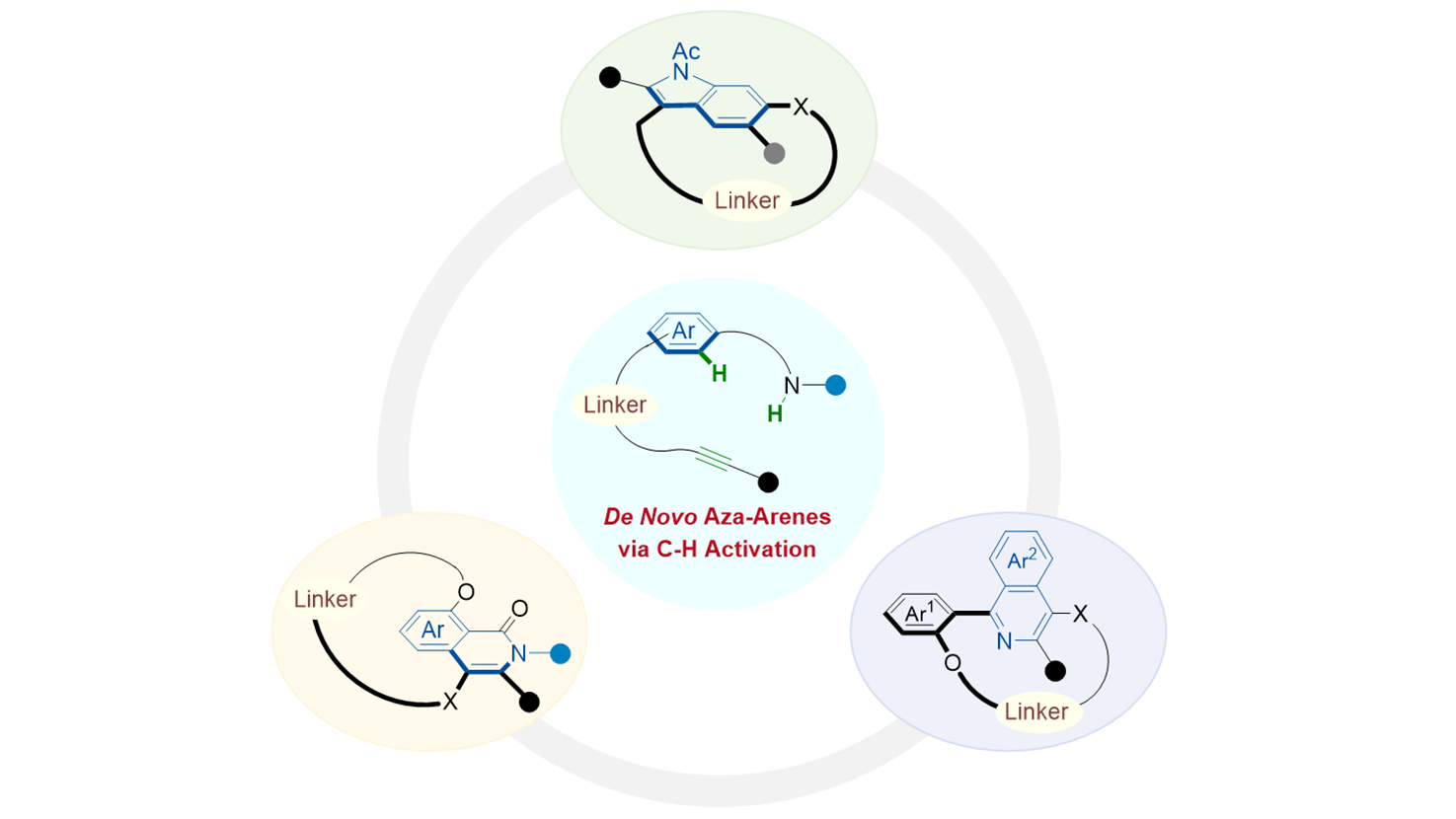
Catalytic enantioselective synthesis of planar-chiral macrocycles by de novo aza-arene construction via C–H activation
-
Zhong-Tian Ye, ... Jin-Sheng Yu
-
DOI: https://doi.org/10.70401/cc.2026.0024 - April 22, 2026
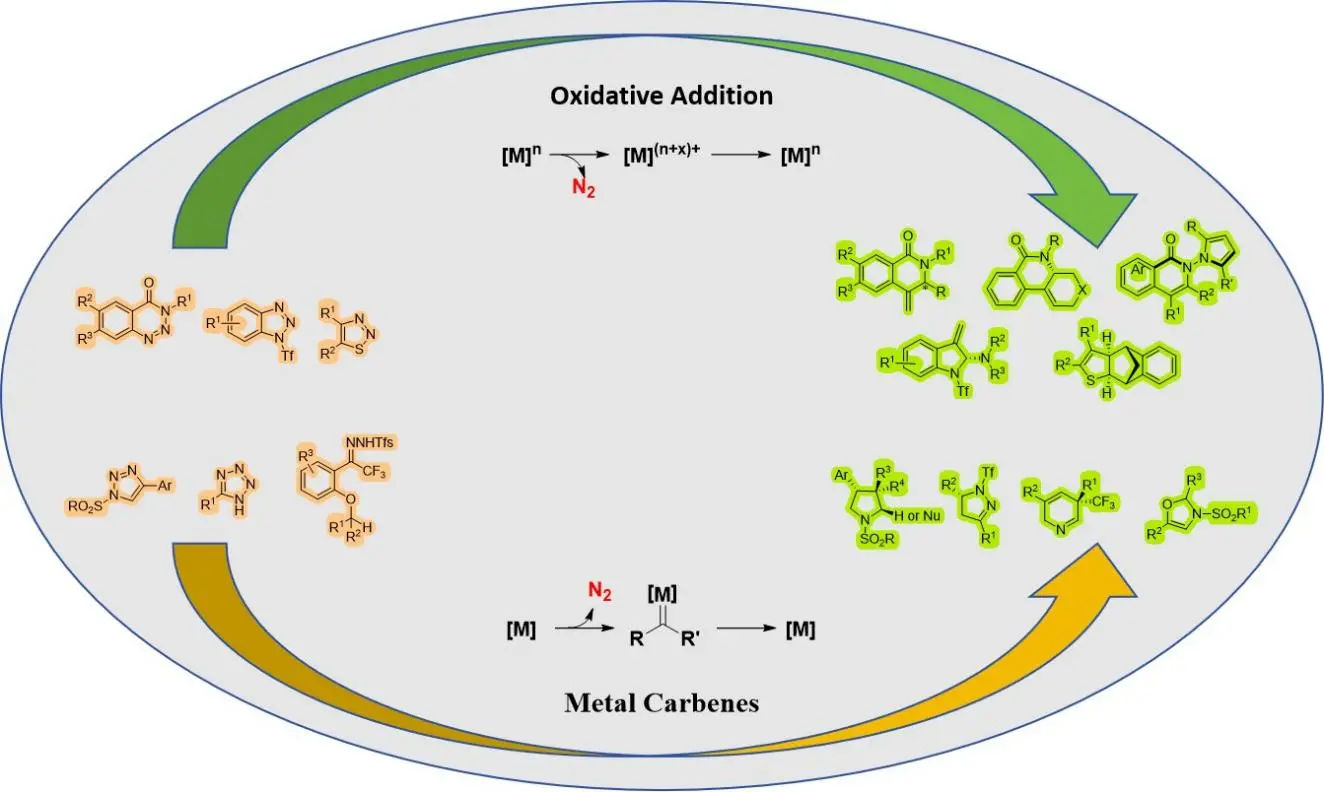
Transition-metal-catalyzed asymmetric denitrogenative transannulation
-
Over the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product ...
MoreOver the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product synthesis. Denitrogenation demonstrates high activity and can engage in cyclization reactions with olefins, alkynes, carbon-heterocycles, aldehydes, and other reagents. This one-step operation enables the rapid construction of chiral heterocycles such as pyrroles and indoles, significantly shortening complex synthetic pathways. Innovations in chiral ligands, optimization of catalytic systems, and detailed studies on mechanisms have significantly enhanced the enantioselectivity and substrate applicability of denitrogenation reactions. This review highlights recent advancements in the synthesis of chiral heterocycles via denitrogenation reactions and systematically examines the reaction characteristics of various metal catalytic systems.
Less -
Wen-Ge Guo, Ren-Rong Liu
-
DOI: https://doi.org/10.70401/cc.2025.0001 - November 06, 2025
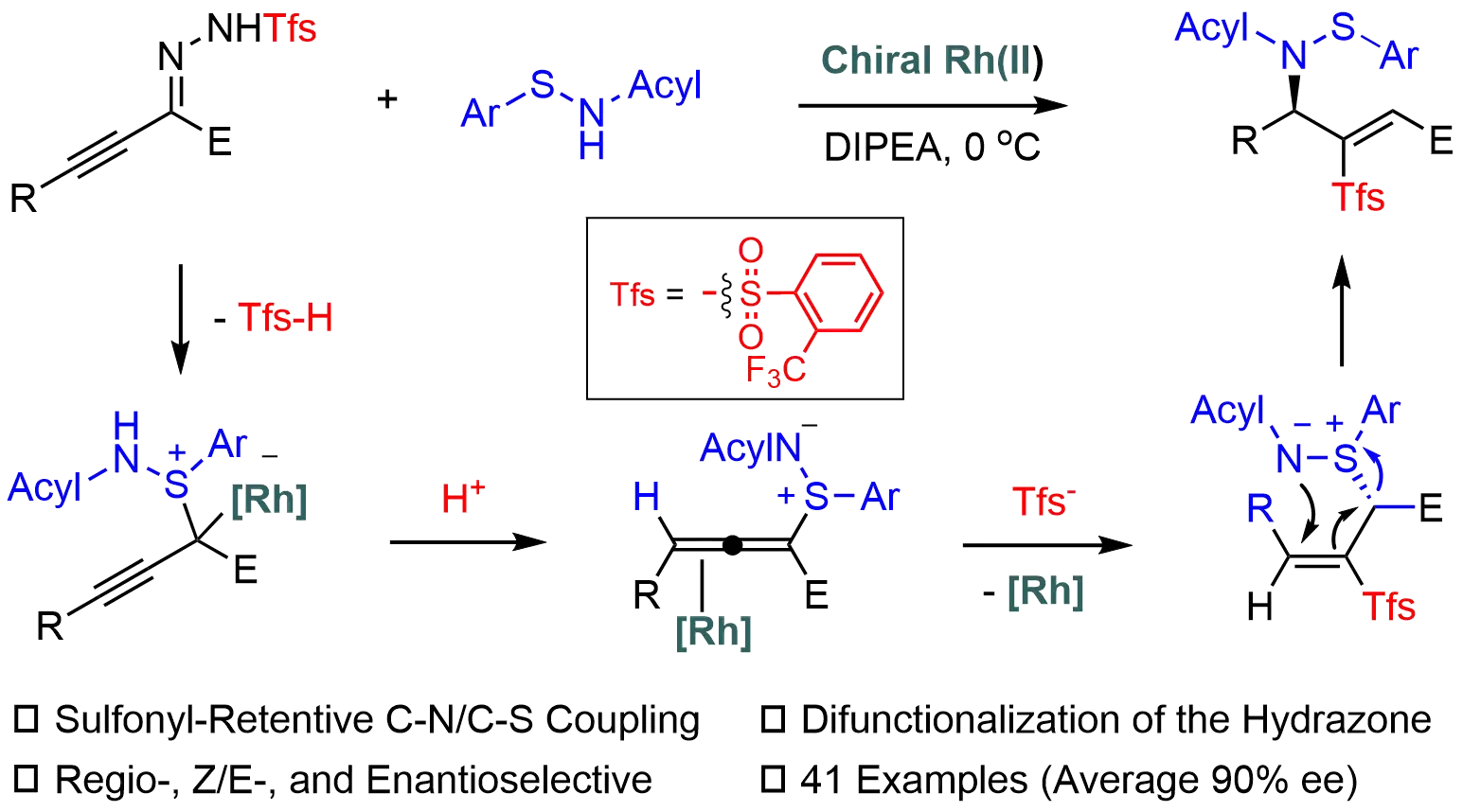
Enantioselective aza-Mislow-Evans rearrangement through S-allenylation of sulfenamides with alkynyl carbenes
-
Sulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated ...
MoreSulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated in Rh-catalyzed asymmetric sulfonyl retentive S,N-difunctionalization of alkynyl N-sulfonylhydrazones with sulfenamides. The sulfonyl group or the corresponding sulfinic acid is retained in a formal migration. This mild and efficient protocol allows for straightforward construction of enantioenriched sulfonyl-based allylic sulfenamides with excellent chemo-, E/Z-, and enantioselectivity, thereby offering a creative strategy for achieving more atom-economic transformations of carbene precursors. Mechanistic studies reveal a three-step process involving S-allenylation, hydrosulfonylation, and an aza-Mislow-Evans rearrangement, with the hydrosulfonylation step governing enantioselectivity.
Less -
Bin Wei, ... Xingwei Li
-
DOI: https://doi.org/10.70401/cc.2025.0004 - December 12, 2025
Advances in catalytic asymmetric hydrogenation of third-row heteroatom-substituted alkenes
-
The asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction ...
MoreThe asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction of the related optically active organosilanes, organosulfides, organophosphine, and organochlorides. These compounds have shown considerable potential for preparing functional molecules and synthesizing natural products. Over the past few decades, considerable research efforts have focused on the design and development of transition-metal catalysts featuring chiral ligands for the asymmetric hydrogenation of such substrates. In parallel, in-depth mechanistic studies have been conducted to elucidate the pathways of these enantioselective hydrogenation reactions, significantly advancing the understanding of their catalytic behavior and stereocontrol. This review focuses on the recent momentum and key advancements in the enantioselective hydrogenation of vinyl silanes, vinyl sulfides, and vinyl chlorides. In addition, given the widespread industrial interest in these compounds, the practical utility of this transformation in the synthesis of chiral silanes, chiral thioethers, chiral alkyl chlorides, as well as related derivatives, is also discussed.
Less -
Jian Zhang, ... Wanbin Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0002 - November 27, 2025
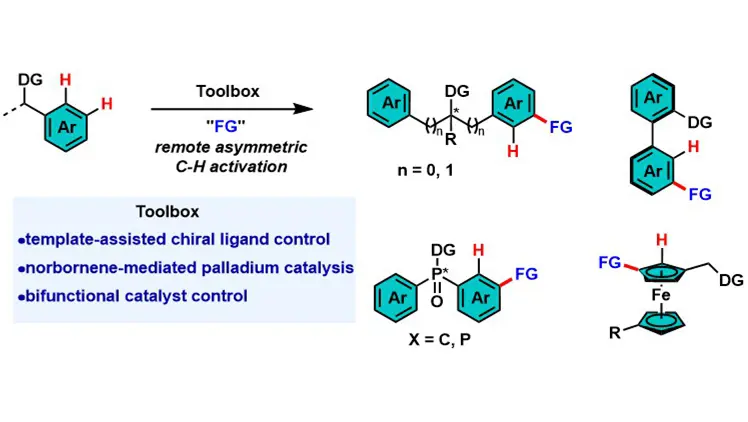
Transition metal-catalyzed remote asymmetric C–H activation of arenes
-
Transition metal-catalyzed asymmetric C–H activation is vital for chiral molecule synthesis but faces challenges in remote C–H functionalization due to traditional metallacycle constraints and difficulties in long-range chiral recognition. This review ...
MoreTransition metal-catalyzed asymmetric C–H activation is vital for chiral molecule synthesis but faces challenges in remote C–H functionalization due to traditional metallacycle constraints and difficulties in long-range chiral recognition. This review summarizes three core strategies to address these issues: template-assisted chiral ligand control, norbornene-mediated palladium catalysis, and bifunctional catalyst control. These strategies achieve high enantioselectivity for diverse chiral architectures. Future directions include expanding to para-C–H bonds of arenes and aliphatic C–H bonds, developing robust chiral mediators/ligands, and applying the methodology to natural products and complex materials.
Less -
Lili Chen, Senmiao Xu
-
DOI: https://doi.org/10.70401/cc.2025.0006 - December 16, 2025
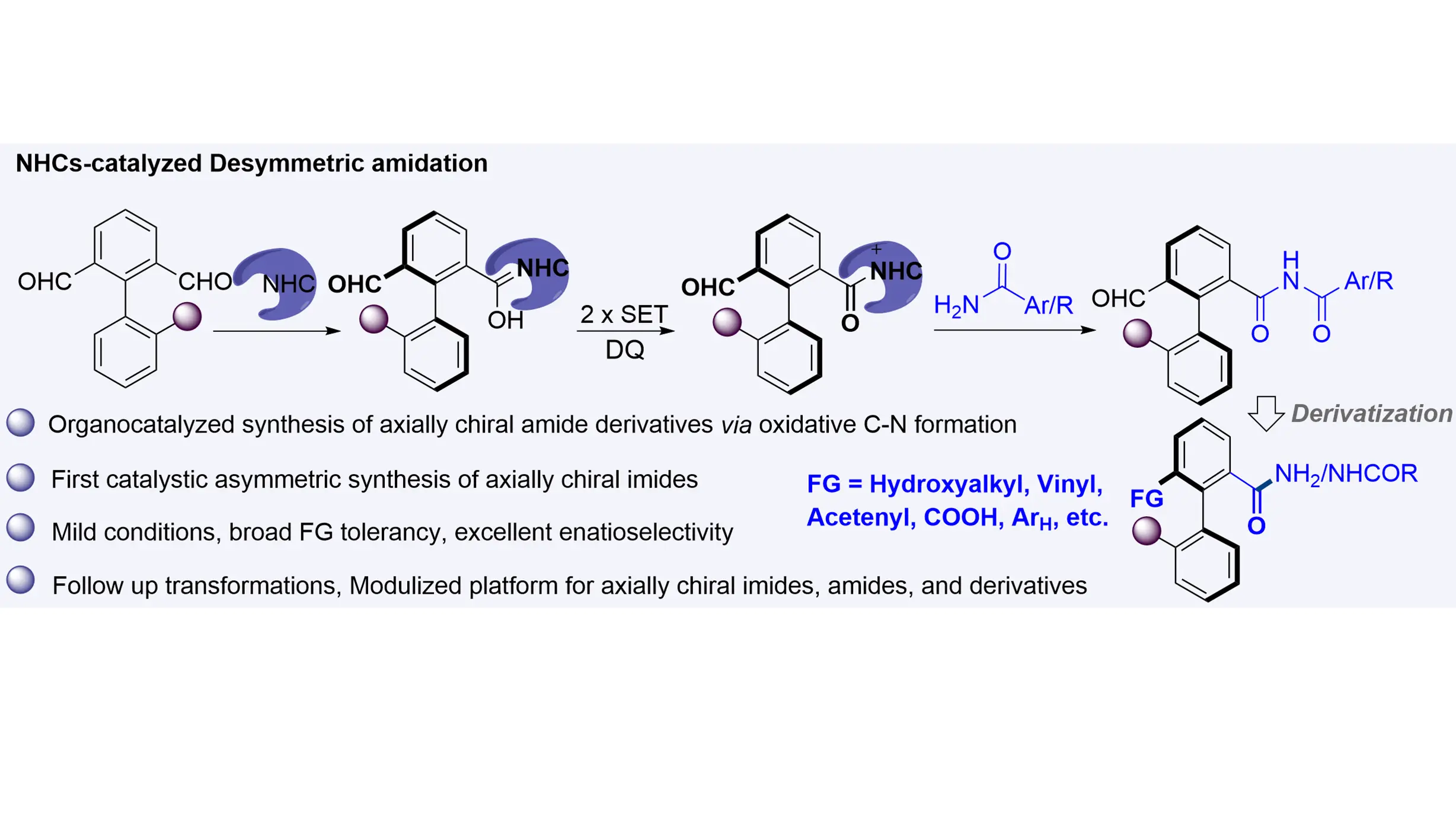
NHCs-catalyzed enantioselective synthesis of biaryl axially chiral imides
-
The synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic ...
MoreThe synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic atropoenantioselective amidation between axially prochiral biaryl dialdehydes and amides that efficiently affords axially chiral imides. This method operates under metal-free and mild conditions, exhibits broad functional group tolerance and substrate scope, and delivers products with excellent enantioselectivities. Furthermore, a wide variety of axially chiral imides, amides, and related derivatives can be accessed through enantio-retentive transformations, offering a versatile and attractive strategy for their synthesis.
Less -
Yingtao Wu, ... Qian Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0005 - December 15, 2025
Advances in catalytic asymmetric hydrogenation of third-row heteroatom-substituted alkenes
-
The asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction ...
MoreThe asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction of the related optically active organosilanes, organosulfides, organophosphine, and organochlorides. These compounds have shown considerable potential for preparing functional molecules and synthesizing natural products. Over the past few decades, considerable research efforts have focused on the design and development of transition-metal catalysts featuring chiral ligands for the asymmetric hydrogenation of such substrates. In parallel, in-depth mechanistic studies have been conducted to elucidate the pathways of these enantioselective hydrogenation reactions, significantly advancing the understanding of their catalytic behavior and stereocontrol. This review focuses on the recent momentum and key advancements in the enantioselective hydrogenation of vinyl silanes, vinyl sulfides, and vinyl chlorides. In addition, given the widespread industrial interest in these compounds, the practical utility of this transformation in the synthesis of chiral silanes, chiral thioethers, chiral alkyl chlorides, as well as related derivatives, is also discussed.
Less -
Jian Zhang, ... Wanbin Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0002 - November 27, 2025
Enantioselective aza-Mislow-Evans rearrangement through S-allenylation of sulfenamides with alkynyl carbenes
-
Sulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated ...
MoreSulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated in Rh-catalyzed asymmetric sulfonyl retentive S,N-difunctionalization of alkynyl N-sulfonylhydrazones with sulfenamides. The sulfonyl group or the corresponding sulfinic acid is retained in a formal migration. This mild and efficient protocol allows for straightforward construction of enantioenriched sulfonyl-based allylic sulfenamides with excellent chemo-, E/Z-, and enantioselectivity, thereby offering a creative strategy for achieving more atom-economic transformations of carbene precursors. Mechanistic studies reveal a three-step process involving S-allenylation, hydrosulfonylation, and an aza-Mislow-Evans rearrangement, with the hydrosulfonylation step governing enantioselectivity.
Less -
Bin Wei, ... Xingwei Li
-
DOI: https://doi.org/10.70401/cc.2025.0004 - December 12, 2025
Transition-metal-catalyzed asymmetric denitrogenative transannulation
-
Over the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product ...
MoreOver the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product synthesis. Denitrogenation demonstrates high activity and can engage in cyclization reactions with olefins, alkynes, carbon-heterocycles, aldehydes, and other reagents. This one-step operation enables the rapid construction of chiral heterocycles such as pyrroles and indoles, significantly shortening complex synthetic pathways. Innovations in chiral ligands, optimization of catalytic systems, and detailed studies on mechanisms have significantly enhanced the enantioselectivity and substrate applicability of denitrogenation reactions. This review highlights recent advancements in the synthesis of chiral heterocycles via denitrogenation reactions and systematically examines the reaction characteristics of various metal catalytic systems.
Less -
Wen-Ge Guo, Ren-Rong Liu
-
DOI: https://doi.org/10.70401/cc.2025.0001 - November 06, 2025
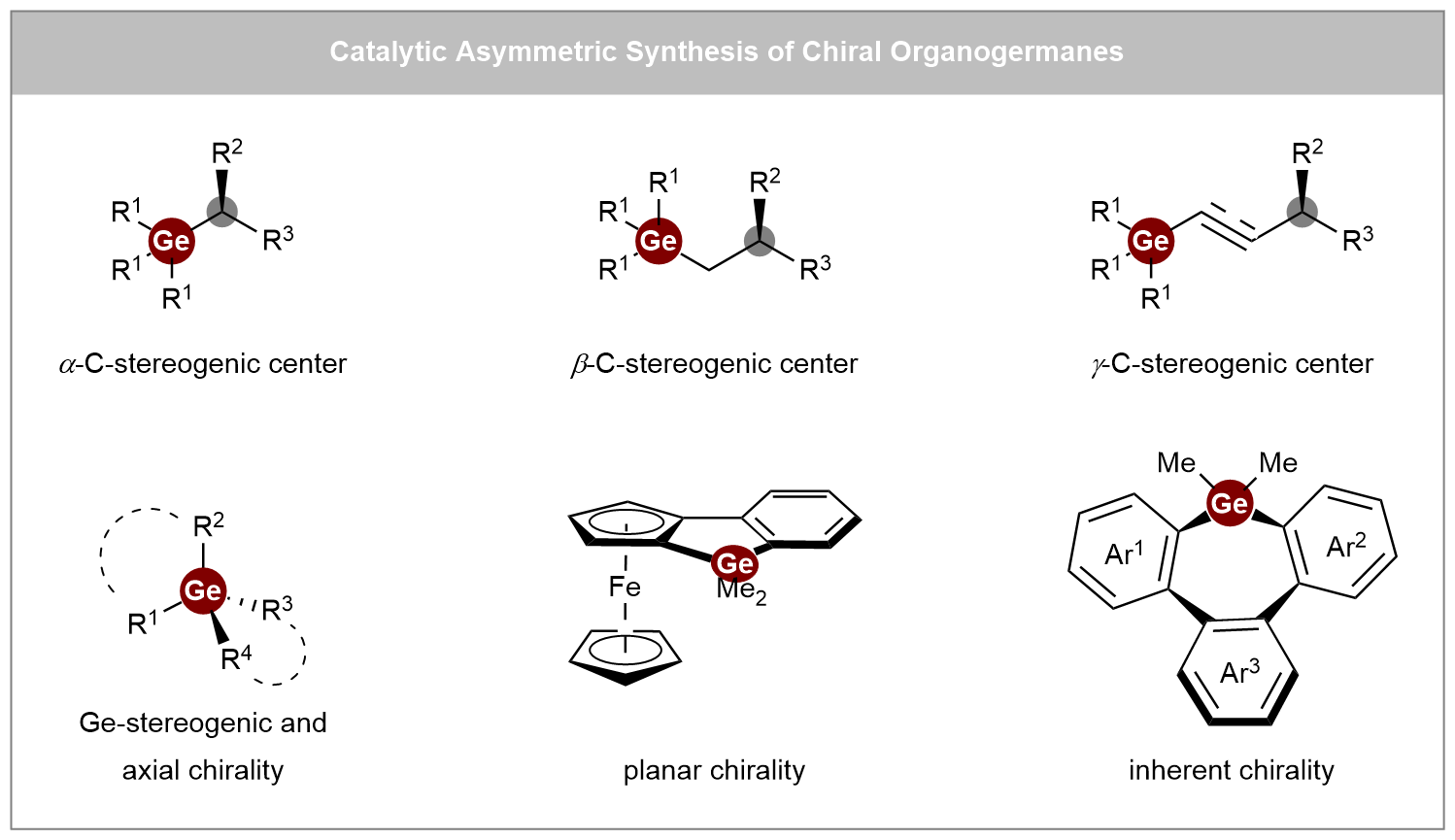
Recent advances in catalytic asymmetric synthesis of chiral organogermanes
-
Chiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent ...
MoreChiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent advances in the catalytic asymmetric synthesis of chiral organogermanes, highlighting a shift from traditional resolution methods toward asymmetric catalytic approaches. The content is organized into three main categories: (i) synthesis of C-stereogenic germanes, (ii) synthesis of Ge-stereogenic germanes, and (iii) synthesis of other chiral germanes, including planar, inherent, and axially chiral types. Key synthetic methodologies are systematically examined, such as enantioselective alkene hydrofunctionalization, carbene insertion, coupling reactions, and [2+2+2] cycloaddition, utilizing a variety of catalytic systems ranging from transition metals (Rh, Cu, Ni, Co) and Lewis acids to engineered metalloenzymes. Particular emphasis is placed on the mechanistic insights and ligand design principles that enable stereochemical control in these transformations. We hope this review will inspire chemists working in related areas and contribute to the future advancement of this field.
Less -
Shao-Wu Liu, ... Chuan He
-
DOI: https://doi.org/10.70401/cc.2026.0013 - February 13, 2026
NHCs-catalyzed enantioselective synthesis of biaryl axially chiral imides
-
The synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic ...
MoreThe synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic atropoenantioselective amidation between axially prochiral biaryl dialdehydes and amides that efficiently affords axially chiral imides. This method operates under metal-free and mild conditions, exhibits broad functional group tolerance and substrate scope, and delivers products with excellent enantioselectivities. Furthermore, a wide variety of axially chiral imides, amides, and related derivatives can be accessed through enantio-retentive transformations, offering a versatile and attractive strategy for their synthesis.
Less -
Yingtao Wu, ... Qian Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0005 - December 15, 2025