Senlin Zhao, Department of Colorectal Surgery, Fudan University Shanghai Cancer Center, Shanghai 200032, China. E-mail: sunshinezsl1989@163.com
Xiaoming Zhang, National Key Laboratory of Immune Response and Immunotherapy, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 200031, China. E-mail: xmzhang@simm.ac.cn
Abstract
Accumulating evidence has indicated that the normal tissue adjacent to tumor (NAT) is distinct from both healthy and tumor tissues. It is suggested that the crosstalk between NAT and tumor tissues helps shape the tumor microenvironment and promotes cancer progression, but the molecular and cellular evidence for this crosstalk is scarce. In this perspective, we propose that NAT tissue constitutes a unique “peritumoral microenvironment (Peri-TME)” mostly conditioned by the tumor. Furthermore, cellular senescence is identified as a key characteristic of Peri-TME, which accelerates tumor growth, as illustrated in our recent studies in colorectal cancer (CRC). Finally, strategies to target the senescent Peri-TME may represent an effective means to disrupt the vicious interaction between Peri-TME and Tumor and enhance therapeutic efficacy.
Keywords
1. Introduction of Peritumoral Microenvironment (Peri-TME)
Paired normal adjacent to tumor (NAT) tissues are often used as controls in cancer studies. However, whether the NAT tissues are truly “normal” remains controversial. A pan-cancer study has shown that the transcriptomic profiles of the NAT tissues are in an intermediate state between healthy and tumor tissues, marked by a unique activation of genes associated with inflammatory responses[1]. Later studies not only confirmed an inflammatory gene signature uniquely existing in the NAT tissues, but also demonstrated its superior value in predicting unfavorable outcomes in lung adenocarcinoma[2] and colorectal cancer[3]. Furthermore, the NAT tissues display other distinct transcriptional features with prognostic significance, including hippo-related genes (HRGs) in hepatocellular carcinoma[4], wound response gene signature in breast cancer[5], Krüppel-like factor 4 (KLF4)[6], vascular endothelial growth factor (VEGF), and epidermal growth factor receptor (EGFR) genes[7] in colorectal cancer.
Several hypotheses have been proposed to implicate the role of the NAT tissues in tumorigenesis and cancer development. The “field cancerization” hypothesis described the NAT tissues as a precancerous state that is histologically normal but genetically changed, primed for early tumor initialization and development[8]. Another “tumor microenvironment” hypothesis advocated that the NAT tissues actively participated in the formation of the tumor microenvironment[5], which was supported at the pan-cancer level[1,9]. Additionally, studies indicated that the transcriptional features of the NAT tissues were mainly shaped by the individual tumor types[10] and the distances to the tumor margin, as evidenced in breast cancer[11] and lung adenocarcinoma[12].
Here, we propose the concept of Peri-TME, a specialized niche located at the critical interface between tumor and normal tissues. Distinct from the field cancerization model, which describes precancerous genetic alterations, and the tumor microenvironment (TME), which characterizes intra-tumoral complexity, the Peri-TME serves as a counterpart to both the TME and the normal tissue microenvironment. It is spatially defined as the transitional interface located between the tumor and truly normal tissue. The Peri-TME enables parallel investigations at cellular, molecular, and tissue levels that are directly comparable to those of the TME. This spatial refinement allows researchers to map dynamic changes along a gradient (tumor - Peri-TME - distant normal tissue) using high-resolution techniques. The Peri-TME framework helps elucidate how tumors initiate, progress, and potentially regress, and provides a new theoretical basis for cancer prevention and treatment. Moreover, because peritumoral cells are not fully transformed, targeting abnormal cells in this region may offer a novel therapeutic strategy and synergize with intratumoral therapies to improve overall efficacy. Thus, the Peri-TME is a complementary niche that enriches our understanding of the tumor-host interface and guides innovative experimental and clinical strategies.
Notably, while our understanding of the Peri-TME is still in its early stages, our recent findings suggest that cellular senescence is a key characteristic of the Peri-TME, which interacts with tumor tissue and exerts a significant impact on tumor progression[13,14]. Furthermore, spatial transcriptomic profiling has revealed that the Peri-TME exhibits distinct cellular composition and signaling dynamics compared to both the tumor and distant normal tissues, highlighting its unique functional role in cancer biology.
2. Senescence and Tumorigenesis
Cellular senescence is a state of stable cell cycle arrest triggered by diverse stress signals, which is closely linked to fundamental biological processes, including tissue repair, regeneration, and vascular remodeling[15]. Key molecular, cellular, and systemic hallmarks have been proposed to underlie the aging process[16]. Moreover, several of the hallmarks of aging are very similar to those observed in cancer, such as genomic instability, epigenetic dysregulation, and chronic inflammation, which might drive both aging and cancer progression[17]. Although induction of senescence can act as a protective response in early-stage tumors, the persistence of senescent cells (SnCs) fuels chronic inflammation through acquiring a senescence-associated secretory phenotype (SASP), which contributes to tumor progression[18,19]. Consequently, the targeted elimination of SnCs or SASP has emerged as a promising strategy to simultaneously mitigate aging pathologies and cancer. While the association between senescence and tumorigenesis has been extensively investigated, the specific roles and underlying mechanisms of senescence within the Peri-TME remain to be explored. We propose the Peri-TME as a distinct entity characterized by a high burden of senescent cells and a pro-inflammatory SASP, which enables spatially resolved analyses, the development of region-specific senolytic interventions, and the exploration of the Peri-TME as an early therapeutic window before irreversible tumor invasion.
3. Senescence-oriented Crosstalk between Peri-TME and Tumor
The Peri-TME is a complex ecosystem composed of a variety of resident and recruited cells, exhibiting spatiotemporal heterogeneity, with its composition and functions evolving continuously as the tumor progresses. Within the Peri-TME niche of colorectal cancer (CRC), the major cell type surrounding the primary tumor is colonic epithelial cells (CECs). Our recent findings have revealed that the peritumoral CECs undergo profound reprogramming into a senescent state during tumorigenesis, thereby contributing to the establishment of a pro-cancer ecosystem[13,14]. Below, we describe two illustrative mechanistic axes from our CRC studies that serve as proof-of-principle examples of senescence-driven intercellular crosstalk between CRC cells and the Peri-TME (Figure 1).
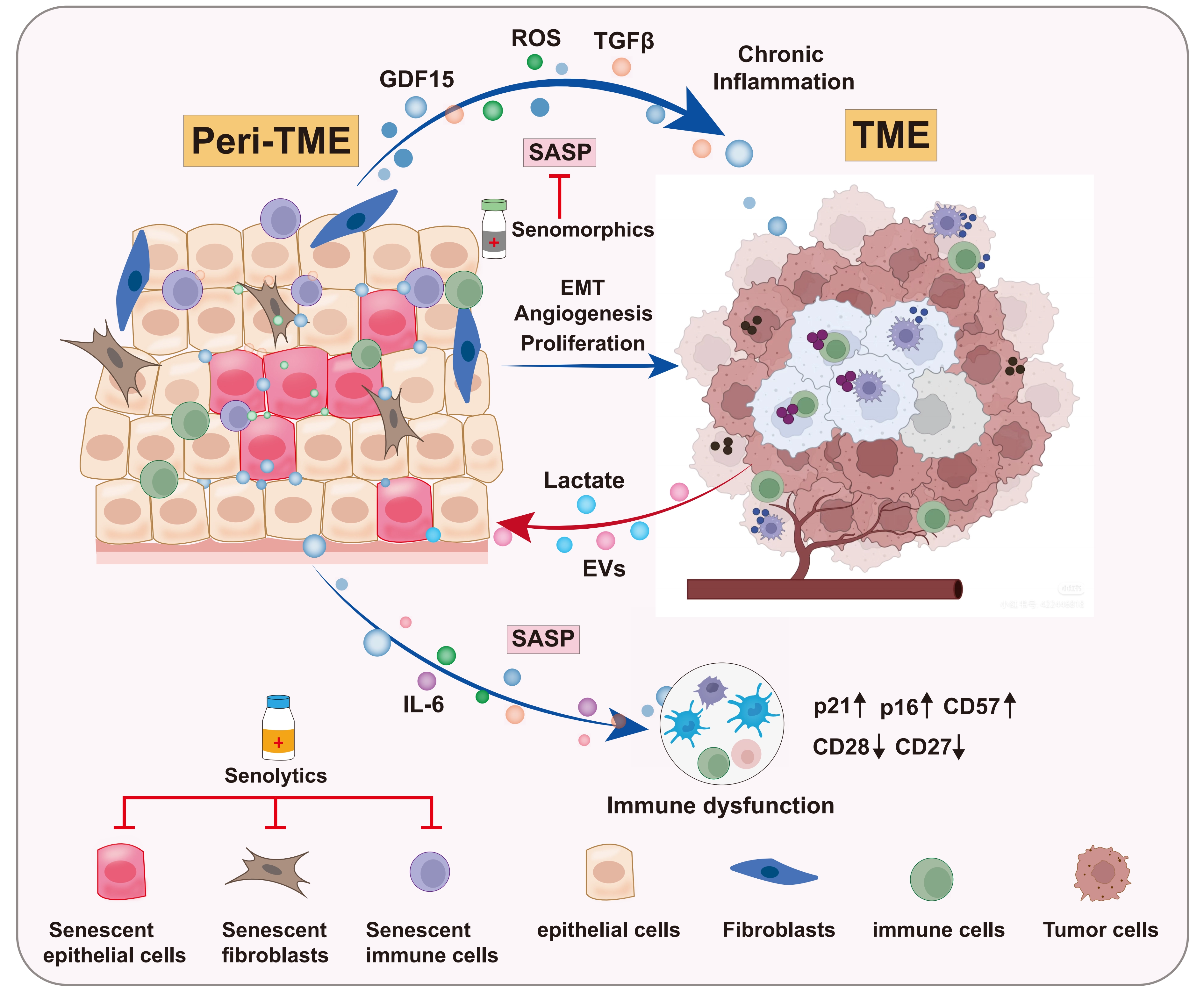
Figure 1. Schematic illustration of the key crosstalk axes between the tumor and the Peri-TME in CRC. TME: tumor microenvironment; GDF15: growth differentiation factor 15; CRC: colorectal cancer; Peri-TME: peritumoral microenvironment; SASP: senescence-associated secretory phenotype.
Axis 1: Peritumoral CEC-derived growth differentiation factor 15 (GDF15)-driven metabolic feedback loop fueling tumor growth[13]
The diverse SASP factors, including cytokines, growth factors, and matrix-remodeling enzymes, orchestrate a pro-tumor microenvironment by driving epithelial-mesenchymal transition (EMT) in nearby epithelial cells, stimulating angiogenesis, recruiting immunosuppressive cells, or increasing metastasis formation[16,20,21]. Our recent findings demonstrated that tumor-derived lactate, a pro-senescence factor[22], induced senescence in peritumoral CECs and triggered the secretion of large amounts of GDF15, a key SASP component. Subsequently, GDF15 secreted by peritumoral CECs induced activation of the JNK1-STAT3-ENO1 signaling pathway in CRC cells, thereby enhancing glycolysis to support tumor survival. This increased glycolysis further triggered extracellular lactate release from CRC cells, which in turn acted on the peritumoral CECs by increasing the lactylation on H4 lysine 8 (H4K8la) and upregulating GDF15 transcription and expression, thereby establishing a metabolic feedback loop between the Peri-TME and the tumor.
Axis 2: Tumor-derived extracellular vesicles as a major driver of metabolic crosstalk between CRC cells and senescent CECs within the Peri-TME[14]
In this study, we demonstrated that peritumoral CECs took up intact mitochondrial DNA (mtDNA) delivered via tumor-derived extracellular vesicles (EVs). This uptake triggered metabolic reprogramming by upregulating the production of reactive oxygen species (ROS) and the secretion of transforming growth factor beta 1 (TGFβ1), two key components of the SASP. The TGFβ1 secreted by peritumoral CECs, in turn, acted on CRC cells to promote tumor progression. Thus, our work identified EV-mtDNA as a major driver of paracrine metabolic crosstalk between colorectal cancer cells and adjacent CECs, making it another example of the crosstalk between the Peri-TME and the tumor.
While the detailed mechanistic axes described above were elucidated in the context of CRC, emerging evidence suggests that NAT tissues promote cancer progression across multiple cancer types[1-3,11,12]. For instance, in lung adenocarcinoma, early-stage tumors cause epigenetic and transcriptional changes in adjacent tissues[12], and an inflammatory gene signature in NAT has been identified as a strong predictor of disease progression[2]. Similarly, the NAT tissues exhibit distinct transcriptomic features in hepatocellular carcinoma (HCC), including dysregulation of pathways such as Hippo signaling, which are associated with a high risk of recurrence[4]. Even in breast cancer, histologically normal tissue adjacent to tumors displays markers of fibrosis and epithelial-mesenchymal transition, hallmarks often associated with senescent stroma[11]. These observations suggest that the presence of a senescence-associated, pro-inflammatory microenvironment in NAT may be a shared response across malignancies and potentially impacts tumor progression. However, it is important to note that most of these studies are correlative, and direct causal evidence linking senescence within the Peri-TME to tumor progression remains limited.
Moreover, senescence within the Peri-TME is a heterogeneous and context-dependent process. In the early stages of tumor development, senescence in NAT tissues may act as a transient “tumor-suppressive barrier” by enforcing cell cycle arrest and limiting the expansion of damaged cells. However, as the tumor progresses, this senescent state often fails to be cleared by the immune system. The persistence of senescent cells leads to a phenotypic switch. Through chronic secretion of SASP factors (e.g., GDF15 or TGFβ1), they turn the Peri-TME into a pro-inflammatory, pro-fibrotic, and immunosuppressive niche that potentially fuels tumor invasion and immune evasion.
Collectively, the above findings suggest that tumors could reshape the Peri-TME into a senescent state by secreting metabolites (e.g., lactate) or molecules (e.g., EVs), which in turn promotes tumor progression, particularly in the context of colorectal cancer. While the CRC-derived model provides a valuable mechanistic framework, the generalizability of these findings warrants further investigation across diverse tumor types and experimental systems. The Peri-TME framework we propose will facilitate clearer research across other cancer types.
4. Senescence of other Cells in Peri-TME
Within the Peri-TME, senescence is not only observed in epithelial cells but may also occur in stromal cells, particularly fibroblasts. Based on extensive literature from tumor contexts, it is plausible that these cells undergo senescence in response to specific stress signals within the Peri-TME and could thereby profoundly influence tumor progression. In the colon, the accumulation of senescent fibroblasts creates a tumor-promoting microenvironment mediated by GDF15[23]. Moreover, senescent fibroblasts secrete SASP and reprogram the extracellular matrix (ECM) to drive infiltration of immunosuppressive cells into the tumor microenvironment[24]. While direct evidence for such senescent stromal populations within the Peri-TME remains lacking, findings in tumors hint that senescent fibroblasts within the Peri-TME exhibit a strong potential to promote tumor cell growth.
The immune system plays a critical role in the anti-tumor response, yet the immune status of the Peri-TME remains poorly understood. Limited data suggest that immune aging, particularly T cell senescence, may occur within this niche. Insights from the broader SASP field suggest a potential link: factors such as TGFβ and prostaglandin E2 (PGE2), known to suppress systemic immunity, may drive local immunosuppression in this niche[19,25]. Under persistent antigen stimulation or inhibitory signals, immune cells, particularly T cells, enter a state of “senescence” characterized by proliferation arrest and functional dysregulation without cell death[26,27]. Unlike anergy and exhaustion, T cell senescence is irreversible[28]. These cells exhibit an abnormal phenotype with downregulation of CD27, CD28, and cytotoxic molecules, alongside upregulation of CD57, p21, p16, and CD45RA, which are closely associated with malignant tumor development[29,30]. Moreover, senescent T cells show a deficiency in both canonical and alternative p38 signaling activation pathways, rendering them unable to proliferate upon TCR stimulation[31]. Given the potentially senescent state within the Peri-TME, it is plausible that “immunosenescence”, particularly T cell senescence[28-31], could occur and potentially contribute to tumor progression. However, direct evidence for such processes within the Peri-TME is currently lacking, and this hypothesis awaits investigation in future studies.
5. Targeting Cellular Senescence within Peri-TME for Cancer Therapy
As described above, cellular senescence within the Peri-TME is emerging as a dynamic and pro-tumorigenic state that drives tumor progression. Thus, there is a compelling rationale for developing “senotherapeutics (senolytics and SASP inhibitors)” to target peritumoral senescent cells or SASP, or integrating the Peri-TME modulation as a neoadjuvant treatment strategy to enhance therapeutic efficacy against tumors.
5.1 Senolytic agents
Senolytics have been used as a promising adjuvant tumor therapy, which specifically target and eliminate senescent cells[32,33]. Thus, we propose that using small-molecule senolytic drugs to deplete senescent cells within the Peri-TME is a promising strategy for cancer treatment. Studies have shown that navitoclax (ABT263) and the related ABT737, selective inhibitors of BCL-2, BCL-XL, and BCL-W, can exert antitumor effects by selectively eliminating senescent cancer cells[34]. As the first and most extensively studied senolytic combination in preclinical models, dasatinib and quercetin (D+Q) treatment has been shown to extend both lifespan and healthspan in aged mice, and significantly enhance the antitumor efficacy of immune checkpoint inhibitors (ICIs) while minimizing adverse events[35,36]. The FOXO4-DRI peptide functions as a senolytic by disrupting the p53-FOXO4 interaction, which leads to the nuclear exclusion of p53, thereby selectively inducing apoptosis in senescent cancer cells[37,38]. Moreover, research has demonstrated that CAR-T cells targeting the senescent cell surface protein urokinase-type plasminogen activator receptor (uPAR) can effectively clear SnCs. In mouse models of lung adenocarcinoma and liver fibrosis, this novel senolytic therapy has shown therapeutic potential[39].
However, a critical challenge for senolytics in clinical studies lies in achieving precision senotherapy, which distinguishes and selectively targets the chronic, pro-tumorigenic senescence while sparing the transient, tumor-suppressive senescence within the Peri-TME. Therefore, the identification of specific senescence biomarkers for SnCs in peritumoral tissue holds great promise for enabling early cancer diagnosis, treatment, and monitoring therapeutic response.
5.2 SASP inhibitors
As an alternative therapeutic approach, SASP inhibitors (senomorphics) can directly or indirectly suppress the SASP without eliminating senescent cells, thereby preserving their anti-tumor function. Senomorphics function by attenuating SASP production through the inhibition of key signaling pathways, including nuclear factor kappa B (NF-κB), Janus kinase-signal transducer and activator of transcription (JAK-STAT), or mitochondrial complex I/IV targets[40,41]. Metformin acts as a senostatic agent to enhance the anticancer efficacy of cyclin-dependent kinase 4 and 6 (CDK4/6) inhibitor LY2835219 by reprogramming the profiles of the SASP[42]. Moreover, compounds such as JQ1, rapamycin, and simvastatin target senescent fibroblasts to inhibit the release of key pro-inflammatory SASP factors, weakening their tumor-promoting capacity[21,43]. Preclinical studies have confirmed that using a cAMP response element-binding protein (CREB) inhibitor or simvastatin (a lipid-lowering drug) can block cholesterol synthesis and lipid droplet formation, thereby averting the tumor-derived extracellular vesicles (tEV)-mediated T cell senescence and synergizing with programmed cell death ligand 1 (PD-L1) treatment[44].
Collectively, senomorphics represent a promising approach in cancer therapy by reprogramming the SASP within the Peri-TME to attenuate its pro-tumorigenic effects and potentially enhance anti-tumor immunity. However, despite the enthusiasm for senolytics and senomorphics, no studies have specifically evaluated their effects on the Peri-TME, and the feasibility of spatially selective targeting remains unproven.
5.3 Combination therapeutic strategies
Given the heterogeneity of senescent cells and the complexity of the SASP within the Peri-TME, future therapeutic strategies are needed to shift towards combination strategies, integrating senotherapeutics with conventional therapies. A study found that the plant compound Rutin is an emerging natural senomorphic agent; when combined with chemotherapy, it can significantly slow tumor progression[45]. Moreover, combining immunotherapy with anti-senescence treatments is a promising strategy. A phase 2 clinical trial of head and neck squamous cell carcinoma (COIS-01) demonstrated that combining senolytics with anti-PD-1 therapy achieved a 33.3% major pathological response rate (95% CI 16.6-54.7%) with a low 4.2% incidence of grade 3-4 adverse events[35]. In another clinical trial, neutralizing GDF15 with visugromab can overcome resistance to anti-PD-1 therapy in lung cancer and urothelial cancer[46]. These findings suggest that combining conventional therapies with agents that target the senescent Peri-TME represents a promising strategy for overcoming treatment resistance in cancer.
6. Challenges and Future Perspectives
While the role of cellular senescence in tumorigenesis or tumor progression has been extensively studied, we are just beginning to reveal its role in the Peri-TME, as exemplified by our recent studies in CRC[13,14]. The Peri-TME is complex and may vary across tumor types. A key challenge is to dissect which features of senescence-driven crosstalk between Peri-TME and tumor are conserved across cancers, and which are tumor-specific. Systematic comparative studies across multiple tumor types will be essential for distinguishing universal principles from context-dependent mechanisms. We propose that cellular senescence is a key feature of the Peri-TME. However, we must acknowledge that the Peri-TME niche has a complex molecular landscape shaped by many overlapping processes, including reactive changes to the tumor burden, active immune cell infiltration, or the “field cancerization” effect, which may not be exclusively driven by senescence. This heterogeneity underscores the need for precise biomarkers to distinguish senescence-specific signatures from other reactive states in future studies. Another challenge is to clearly define the cellular and molecular composition of the Peri-TME and to assess the extent of senescence across different tumor types. Single-cell multi-omics, spatial technologies, and functional studies will be employed for this purpose.
Systemic senotherapeutics demonstrate efficacy in preclinical models, yet most current agents lack the spatial specificity to selectively target the Peri-TME without disrupting systemic homeostasis or transient tumor-suppressive senescence elsewhere. Therefore, identifying Peri-TME-specific senescence biomarkers is critical for developing precision senotherapeutics. While we have identified the GDF15-lactate axis between Peri-TME and tumor in CRC, many other interaction modes are still unknown. Through systematic screening of senescence-associated pathways, we may identify novel crosstalk mechanisms with therapeutic potential.
Clinically, a critical question is whether selective targeting of senescent cells within the Peri-TME can serve as a neoadjuvant approach to enhance surgical outcomes in locally advanced cancer. Although the combination of senotherapeutics with standard anti-tumor treatments is under active exploration, the efficacy of targeting the senescent Peri-TME still requires extensive validation, alongside the identification of reliable biomarkers to select patients most likely to benefit.
Authors contribution
Zhou M, Han A: Writing-original draft, visualization.
Zhao S, Zhang X, Yan D: Conceptualization.
Guan B: Writing-original draft, visualization, conceptualization.
Conflicts of interest
Xiaoming Zhang is an Editorial Board member of Geromedicine. The other authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 82372643 to S.Z. and 82503175 to B.G.), the Strategic Priority Research Program of the Chinese Academy of Sciences (Grant No. XDB0940000 to X.Z.), and the Natural Science Foundation of Shanghai (Grant Nos. 25ZR1402438 to B.G. and 25ZR1401303 to D.Y.).
Copyright
© The Author(s) 2026.
References
-
2. Dolgalev I, Zhou H, Murrell N, Le H, Sakellaropoulos T, Coudray N, et al. Inflammation in the tumor-adjacent lung as a predictor of clinical outcome in lung adenocarcinoma. Nat Commun. 2023;14:6764.[DOI]
-
3. Kim J, Kim H, Lee MS, Lee H, Kim YJ, Lee WY, et al. Transcriptomes of the tumor-adjacent normal tissues are more informative than tumors in predicting recurrence in colorectal cancer patients. J Transl Med. 2023;21(1):209.[DOI]
-
8. Curtius K, Wright NA, Graham TA. An evolutionary perspective on field cancerization. Nat Rev Cancer. 2018;18(1):19-32.[DOI]
-
15. de Magalhães JP. Cellular senescence in normal physiology. Science. 2024;384(6702):1300-1301.[DOI]
-
16. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186(2):243-278.[DOI]
-
17. López-Otín C, Pietrocola F, Roiz-Valle D, Galluzzi L, Kroemer G. Meta-hallmarks of aging and cancer. Cell Metab. 2023;35(1):12-35.[DOI]
-
19. Wang B, Han J, Elisseeff JH, Demaria M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol. 2024;25(12):958-978.[DOI]
-
20. McHugh D, Durán I, Gil J. Senescence as a therapeutic target in cancer and age-related diseases. Nat Rev Drug Discov. 2025;24(1):57-71.[DOI]
-
21. Colucci M, Sarill M, Maddalena M, Valdata A, Troiani M, Massarotti M, et al. Senescence in cancer. Cancer Cell. 2025;43(7):1204-1226.[DOI]
-
23. Guo Y, Ayers JL, Carter KT, Wang T, Maden SK, Edmond D, et al. Senescence-associated tissue microenvironment promotes colon cancer formation through the secretory factor GDF15. Aging Cell. 2019;18(6):e13013.[DOI]
-
24. Hoi XP, Stangis MM, Glass SE, Kim JH, Kang SW, Brennen WN, et al. Cellular senescence in precancer lesions and early-stage cancers. Cancer Cell. 2026;44(1):6-11.[DOI]
-
28. Zhao Y, Shao Q, Peng G. Exhaustion and senescence: Two crucial dysfunctional states of T cells in the tumor microenvironment. Cell Mol Immunol. 2020;17(1):27-35.[DOI]
-
29. Lian J, Yue Y, Yu W, Zhang Y. Immunosenescence: A key player in cancer development. J Hematol Oncol. 2020;13(1):151.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite




