Abstract
Traditional cancer treatment methodologies, including chemotherapy, surgery, radiotherapy, and immunotherapy, commonly lead to severe adverse reactions. In recent years, innovative treatment modalities, particularly photodynamic therapy (PDT) and photothermal therapy (PTT), have garnered increasing attention due to their enhanced therapeutic levels. Against this background, black TiO2, a new kind of functional material, has become a research emphasis in the domain of cancer treatment. This material exhibits broad spectral absorption and strong near-infrared light penetration, enabling it to efficiently satisfy the optical requirements of both PDT and PTT. This article presents a mini review of PDT, PTT, and their synergistic combination strategies based on black TiO2.
Keywords
1. Introduction
Cancer remains a major threat to human health in the 21st century. Fortunately, a multitude of cancer treatment strategies are clinically accessible, including chemotherapy and radiotherapy. Nevertheless, conventional treatment approaches frequently result in acute adverse effects on normal tissues, imposing additional suffering on patients. Moreover, these methods typically trigger the development of drug resistance in cancer cells, giving rise to tumor cell deterioration, recurrence, and metastasis. One major cause of drug resistance is the development of genetic mutations in cancer cells. These mutations can affect genes that are targeted by drugs, such as enzymes or receptors that control cell growth or survival. Also, changes in genes that repair DNA damage can make cancer cells better at fixing drug-induced damage, allowing them to survive and grow even with drug treatment. Furthermore, mutations in genes related to cell death pathways can render cancer cells resistant to drug-induced apoptosis, thereby promoting their survival[1]. Consequently, there is a pressing requirement for novel therapeutic modalities to address cancer and mitigate these side effects.
In the past few decades, photodynamic therapy (PDT) and photothermal therapy (PTT) have emerged as novel cancer treatment methods[2,3]. PDT relies on the combined effects of excitation light, photosensitizer (PS), and molecular oxygen[3]. Photosensitizers are photosensitized to decompose H2O/O2 in tissues, generating reactive oxygen species (ROS) to damage tumor cells[4]. Unlike PDT, the main mechanism of PTT is that photothermal agents absorb near-infrared (NIR) light and convert it into heat energy (raising the local temperature of tumors to 40-45 ℃) to kill tumor cells[3]. In fact, TiO2 nanoparticles (NPs) simultaneously possess PDT and PTT effects and can be potentially used for cancer treatment.
Recently, titanium dioxide (TiO2), one of the most vital and classical metal oxides, has been thoroughly investigated and extensively used in many fields of science and technology owing to its outstanding photoactivity[5]. Notably, with high absorption in the ultraviolet (UV) light region, TiO2 can act not only as a PS to generate cytotoxic ROS, but also as a photothermal agent (PTA) to convert light energy into hyperthermia[6]. However, the relatively wide energy band gap of TiO2 enables it to absorb energy only in the ultraviolet light region. In practical applications, it still has limitations such as insufficient utilization of visible light and low quantum efficiency[7]. In 2011, Chen et al. constructed a disordered structure on the surface of the TiO2 lattice through a hydrogenation method and successfully prepared black TiO2 nanomaterials for the first time (Figure 1a). The band gap is significantly reduced (Figure 1b), which enhanced the visible-light response performance and quantum efficiency of the materials. In PDT/PTT, “high UV absorption” underpins the UV-driven PDT performance of conventional TiO2, and yet UV suffers from poor tissue penetration. The primary merit of black TiO2 is that it preserves this feature while extending spectral absorption into the NIR region, thus achieving efficient therapy by utilizing the superior tissue penetration of NIR. Since then, a research upsurge on black TiO2 nanomaterials has been sparked[7].

Figure 1. (a) Photographs of white TiO₂ and black TiO₂ (B-TiO₂); (b) Comparison of the band structures and band gaps of black TiO₂ and white TiO₂[8]. TiO2: Titanium dioxide.
Numerous studies have been conducted on the use of black TiO2 in cancer treatment. Therefore, a comprehensive review is required to systematically summarize its applications in PDT, PTT, and their combined treatment strategies with other methods. This review first introduces the characteristics and preparation of black TiO2. It then presents the current research on black TiO2 in PDT and PTT. Finally, it discusses current advances in combined treatment strategies involving black TiO2 and other methods, and provides a prospect.
2. Characteristics and Preparation of Black TiO2
2.1 Characteristics of black TiO2
Similar to TiO2, black TiO2 mainly exists in three crystalline structures: rutile[9], anatase[10], and brookite[11]. The rutile phase, belonging to the hexagonal crystal system, is the most stable crystal form of black TiO2, exhibiting high catalytic activity and stability[9]. The anatase phase, also a member of the hexagonal crystal system, has a relatively unstable crystal structure yet exhibits a large specific surface area and photocatalytic activity, making it one of the most commonly observed crystal forms of black TiO2[10]. The brookite phase of black TiO2 belongs to the orthorhombic crystal system with relatively low structural stability; however, it features high electron mobility and is often blended with the anatase phase for photocatalytic applications[11]. Black TiO2 is similar to titanium dioxide in terms of crystal structure, but differs in composition, optical properties, electronic properties, and surface properties.
Black TiO2 actively incorporates defects, such as oxygen vacancies and low-valence ions, resulting in marked differences from ordinary TiO2 in optical, electronic, and surface properties[12]. In terms of optical properties, the light absorption of ordinary TiO2 is limited to the ultraviolet region, while black TiO2, through defect engineering, enables efficient absorption in the near-infrared region. Regarding electronic properties, the energy band structure of ordinary TiO2 is fixed. However, the intrinsic defects in black TiO2 reduce the band gap from 3.2 eV to approximately 1.5 eV, extending the photoresponse to the near-infrared region. Meanwhile, a core-shell structure with a disordered surface and an ordered interior is formed, and oxygen vacancies are introduced simultaneously[7].
In terms of surface properties, ordinary TiO2 has a regular surface with relatively low reactivity. The defects in black TiO2 decrease the adsorption energy an activation energy of reactants. Moreover, broadening the light response range improves the efficiency of active sites. For instance, low-valence ions and oxygen vacancies can trap photogenerated charge carriers, enhance the separation of photogenerated electrons (e-) and holes (h+), and thus achieve efficient catalytic activity under visible or near-infrared light[13].
2.2 Preparation of black TiO2
Currently, the methods for preparing black TiO2 are mainly classified into atmosphere roasting method[14], metal reduction method[13], chemical reduction method[15], femtosecond pulse method[15], and sol-gel method[16]. The preparation methods and synthesis conditions can induce changes in physical and chemical properties of black TiO2, including morphology, structure, optical properties, and electrical properties, which are summarized in Table 1.
| Method | Feature | Limitation | Ref. |
| Atmosphere calcination | Precise control over the type and content of defects | The hydrogen-atmosphere method suffers from high costs, high risks, and is not suitable for large-scale production. | [7] |
| Metallothermic reduction | Self-sustaining exothermic reaction enabling facile preparation of black TiO2 with high defect density | Excessive doping or the generated defects can influence the light-absorption and photocatalytic properties of the material. | [13] |
| Chemical reduction | Mild reaction conditions and simple operation, facilitating easy regulation of product particle size and dispersibility | The reduction ability is limited, and the reduction reaction is mostly restricted to the surface of TiO2. | [17] |
| Femtosecond laser pulse | High processing precision and localized modification capability for fabricating black TiO2 with specific microstructures | It is costly and not applicable to large-scale production. | [18] |
| Sol-gel | Easy doping and modification, allowing the preparation of nanoscale black TiO2 powders or thin films. | It has a long cycle and poor process stability. | [19] |
2.2.1 Atmosphere roasting method
The atmosphere roasting method involves roasting the TiO2 precursor under specific gas atmospheres (either non-air or air-controlled) and heating conditions to ultimately prepare black TiO2. The commonly used gases are primarily hydrogen and nitrogen. The hydrogen atmosphere method has the drawbacks of high cost, great safety risks, and limited suitability for large-scale production[7]. For example, Chen et al.[7] synthesized black TiO2 via calcination in a hydrogen atmosphere. The high-temperature, high-pressure conditions and H2 environment induce lattice disorder on the surface of white TiO2, leading to the formation of a core-shell structure with a crystalline core and an amorphous shell. This structural transformation alters the band gap width of TiO2, reducing it to an initial absorption band gap of 1.0 eV, while hydrogen further stabilizes the lattice disorder by passivating dangling bonds. Currently, hydrogenation remains the mainstream synthesis method for black TiO2; however, this method still suffers from the mentioned-above drawbacks.
2.2.2 Metallothermic reduction method
The synthesis of black TiO2 by the metallothermic reduction method makes use of the strong reducibility of active metals[20]. Under specific conditions (high temperature and/or an inert atmosphere), an oxidation-reduction reaction occurs between the active metals and the lattice oxygen in TiO2. By “seizing” the lattice oxygen in TiO2, a large number of oxygen vacancies and low-valence ion defects are directionally introduced. For example, Song et al.[13] synthesized black TiO2 nanostructures with an ultrathin hollow sphere morphology, high crystallization quality, small grain size, and ultra-high surface area through the aluminothermic reduction method. Different from the atmosphere roasting method, the reaction by this method is more intense, making it easier to achieve multi-metal doping during the reduction process. In 2015, Sinhamahapatra et al.[21] first reported the synthesis of TiO2 via magnesiothermic reduction in the coexistence of H2/Ar, followed by acid washing to prepare black TiO2 nanoparticles. The study indicated that in the high-resolution X-ray photoelectron spectroscopy (XPS) spectra, the peak intensity corresponding to black TiO2 increased with the increase in the dosage of Mg reductant. Specifically, as the amount of Mg increased, more oxygen vacancies were formed on the surface of black TiO2, which further enhanced the visible light absorption capacity and photocatalytic activity of the samples. Therefore, the key to this method lies in the reasonable control of the reducing agent quantity during the reaction.
2.2.3 Chemical reduction method
The chemical reduction method involves the use of reductive agents to generate the reductive condition to produce black TiO2[22]. Under mild temperature conditions, the oxidation-reduction reaction can occur with TiO2. By injecting electrons into the TiO2 lattice, low-valence ion and oxygen vacancies are induced. Jiang et al.[15] synthesized black TiO2 with a mesoporous hollow shell structure by the evaporation-induced self-assembly method. In this work, the authors fabricated silica (SiO2) colloidal particles via a modification experiment. These SiO2 colloidal particles were repeatedly dispersed and coated onto the surface of TiO2 followed by high-temperature calcination to prepare SiO2@TiO2@SiO2 core-shell-shell particles. Subsequently, the particles were subjected to NaOH etching and NaBH4 reduction to obtain mesoporous black TiO2 hollow spheres. It was found that this unique hollow mesoporous structure endows black TiO2 with a higher specific surface area and more active reaction sites, significantly enhancing its photocatalytic activity and light absorption capacity. Compared with the metallothermic reduction method, the mild temperature condition results in the limited reducing ability, confining the reduction reaction mostly to the particle surface[15].
2.2.4 Femtosecond laser pulses
Recent studies have demonstrated that a femtosecond laser filament-based far-field technique can remotely and rapidly fabricate nano/microstructures on crystalline silicon, as well as on irregularly shaped organic and metals surfaces for improved light absorption[23]. Su et al.[18] irradiated TiO2 particle using femtosecond laser pulses, leading to the a large number of oxygen vacancies generated in the particle surface. However, compared with other methods, this method is costly and unsuitable for large-scale production.
2.2.5 Sol-gel method
The sol-gel method uses precursors containing highly chemically active components and hydrolysis (as well as other reactions) in the liquid phase to form a stable sol system[19]. After aging, the colloidal particles slowly polymerize and transform into a gel state with a three-dimensional network structure. The gel can be further processed through drying, calcination, and other techniques to obtain functional materials with a porous structure. Currently, this method has also been applied to the construction of black TiO2[19]. Patil et al.[19] prepared S, C, Ti3+ co-doped black TiO2 nanoparticles using the sol-gel method. However, the synthesis of black TiO2 by the sol-gel method has a long cycle and poor process stability.
3. BT in Cancer Treatments
Before being applied to cancer treatment, black TiO2 was widely used in various fields, such as photocatalytic hydrogen production, air purification, water treatment, and photoelectrochemical cells[24]. Based on excellent broad-spectrum properties and biocompatibility of black TiO2, in 2015, Ren et al.[17] first used black TiO2 to treat cancer under near-infrared light irradiation. Since then, endeavor on the application of black TiO2 in cancer treatment has been boosted. In this section, the applications of black TiO2 in PDT, PTT, and the combined PDT and PTT are introduced (Figure 2).
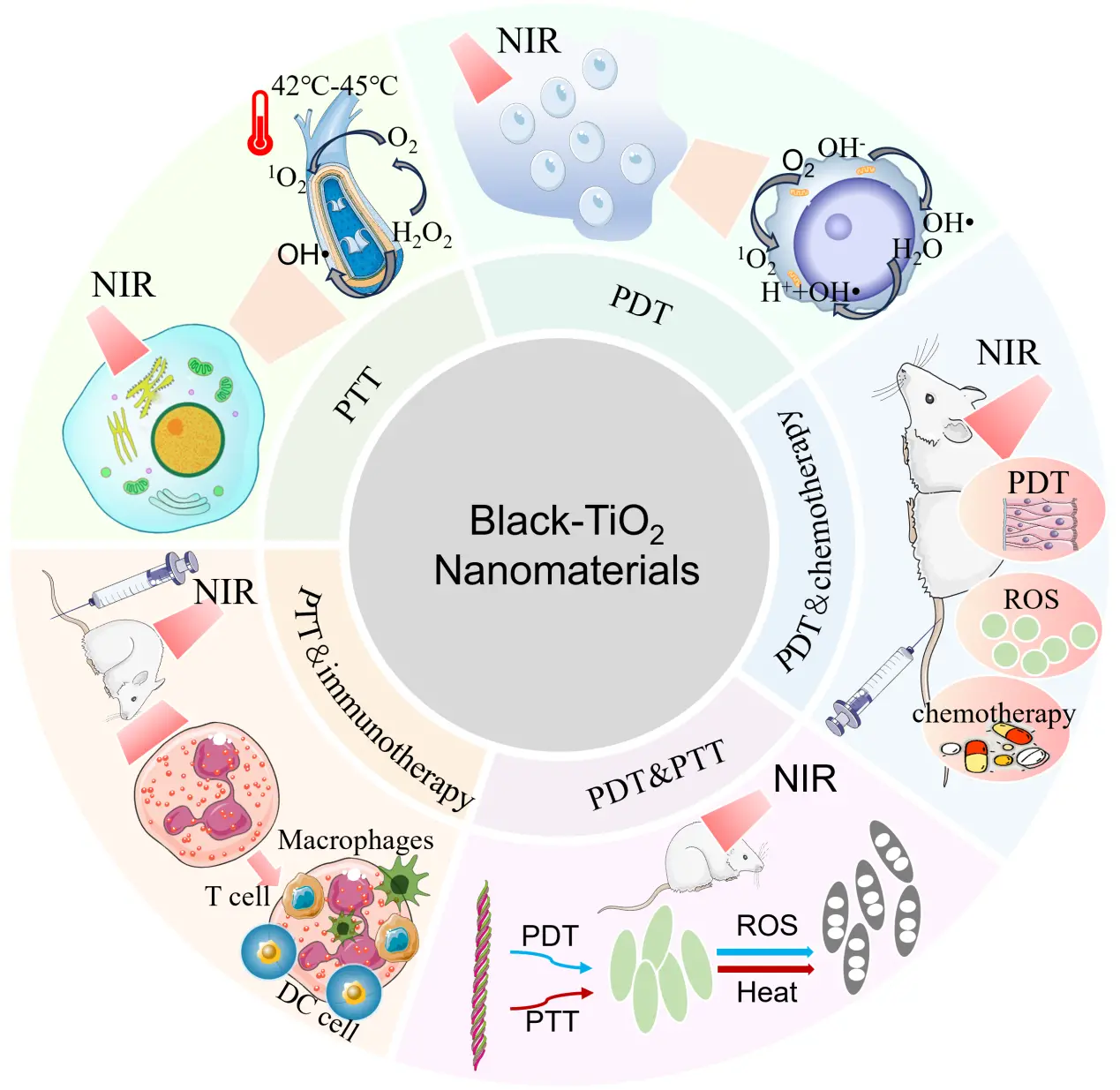
Figure 2. Therapeutic approaches for cancer using black TiO2. PTT: photothermal therapy; PDT: photodynamic therapy; NIR: near-infrared; ROS: reactive oxygen species.
3.1 BT in PDT therapy
As early as 2017, Ni et al.[25] first reported that black TiO2 could be used as a photosensitizer for the phototherapy of bladder cancer cells at 808 nm, which established a basis for further research on PDT anti-cancer treatment. With the development of PDT for anti-cancer treatment, Estefanía et al.[26] developed photoactive nanomaterials with drug-encapsulating properties. This study employs cervical cancer HeLa cells as the model, and mesoporous black TiO2 nanorods prepared via sodium borohydride (NaBH4) reduction as both drug carriers and photodynamic platforms. The Ti3+ sites and oxygen vacancies within the nanorods enable visible light-responsive generation of hydroxyl radicals (HO•). The material efficiently loads the chemotherapeutic drug doxorubicin (encapsulation efficiency: 92%). Through the synergistic effect of chemotherapy and photodynamic therapy, it also inhibits cancer cells via DNA/RNA damage, lipid peroxidation, and apoptotic pathways, reducing the cell viability to 30% at a concentration of 400 μg/mL. This work provides a novel strategy for the combined therapy of cervical cancer.
3.2 BT in PTT therapy
In 2015, Ren et al.[17] pioneered the application of black TiO2 for tumor PTT, with polyethylene glycol (PEG) modification to enhance biocompatibility and stability. In this study, black TiO2-PEG was intravenously administered to mice bearing 4T1 breast cancer xenografts, followed by irradiation of the tumor region with an 808 nm NIR laser. Tumor ablation was achieved through local hyperthermia, and the material exhibited low toxicity and high photothermal conversion efficiency, which were fully validated by experimental results. Building on this foundational work, subsequent studies have continued to innovate while retaining these core advantages. In recent years, mesoporous silica-coated black TiO2 photothermal nanoparticles have not only been shown to enhance the anti-cancer effect of PTT, but also been found to elicit tumor-associated antigens after PTT, which can stimulate the body to generate a tumor-specific immune response[27]. This provides a typical example for photothermal immunotherapy.
3.3 BT in combined PDT/PTT therapy
Based on the excellent performance of black TiO2 in PDT and PTT, combined PDT and PTT have attracted significant attention. In 2019, Saeed et al.[28] designed an Fe3O4-black TiO2 nanocomposites, which exhibited strong broadband light absorption, high photothermal conversion efficiency, and good magnetic resonance imaging properties. In 2021, Guo et al.[29] designed a novel titanium-based targeting agent and proposed a nano-theranostic system with strong near-infrared-II absorption. In this study, black TiO2 was subjected to hydrogenation reduction with NaBH4 to introduce oxygen vacancies and boron doping, narrowing its band gap for NIR-II responsiveness. Its surface was coated with SiO2 and grafted with hyaluronic acid (HA) to obtain black TiO2@SiO2–HA, which possesses both the functions of targeting CD44-overexpressing tumors and NIR-II photoacoustic imaging guidance. Under 1,064 nm laser irradiation, this system synchronously mediates PDT and PTT, achieving tumor ablation through a synergistic effect in vitro (4T1 and HeLa cells) and in vivo (4T1 tumor-bearing mice). Guided by real-time NIR-II photoacoustic imaging, combined PTT-PDT effects act on the tumor cells apoptosis, showing great potential in targeted PTT-PDT.
3.4 Comparison of BT with conventional PDT/PTT agents
Traditional PDT requires the synergistic use of photosensitizers (PSs) and light of specific wavelengths. Porphyrin-based compounds are the mainstay of PSs, such as hematoporphyrin derivative (HpD, trade name: Photofrin) and 5-aminolevulinic acid (5-ALA), with excitation wavelengths mostly ranging from 630 to 689 nm. Conventional PTT primarily relies on noble metal nanomaterials, including gold nanospheres, gold nanorods, gold nanoshells, and gold nanocages, all of which can efficiently generate heat through the localized surface plasmon resonance effect, with excitation wavelengths adapted to theNIR region[30].
Compared with traditional agents, black TiO2 features excitation wavelengths precisely matched to the NIR region, achieving a tissue penetration depth of 1-2 cm. This effectively overcomes the limitation of conventional PDT photosensitizers (excited by red light with a penetration depth < 1 cm). Furthermore, in contrast to gold-based photothermal agents that suffer from high preparation costs and potential in vivo accumulation toxicity, black TiO2 boasts more universal preparation processes and lower risks of clinical translation. However, its photothermal conversion efficiency, ranging from 32%-40%[26] and sufficient for clinical therapeutic needs, is lower than that of morphologically optimized traditional photothermal agents (e.g., gold nanoshells and gold nanocages with a photothermal efficiency of 35%-50%)[31].
In addition, although traditional inorganic phosphorus materials (such as violet phosphorus quantum dots (VPQDs) and black phosphorus nanosheets (BPNSs)) exhibit excellent photoresponsive activity, they possess inherent drawbacks: the weak van der Waals forces between layers render them prone to degradation in oxygen- or water-containing environments, leading to diminished photoresponsive performance. Despite the production of metabolites (e.g., phosphate ions) capable of inducing calcification during degradation, their insufficient short-term stability restricts the long-term synergy of PDT/PTT. Moreover, the photothermal conversion efficiency of traditional inorganic phosphorus materials depends on specific morphological regulation (e.g., the two-dimensional sheet structure of BPNSs and the quantum size effect of VPQDs)[32-35]. In contrast, the defect state structure of black TiO2 exhibits low dependence on morphology, endowing it with more universal preparation processes.
In terms of therapeutic safety, the biocompatibility of black TiO2 has been validated in preclinical studies, with its degradation product (titanium ions) showing extremely low toxicity. In contrast, the degradation of phosphorus materials may be accompanied by an abnormal local increase in phosphate ion concentration, necessitating strict control of the administration dose to avoid potential tissue damage[27]. In summary, black TiO2 demonstrates a balanced advantage between stability and photoresponsive efficiency in synergistic PDT/PTT therapy. To facilitate its further development and clinical translation, Table 2 summarizes the key technical parameters of the black TiO2 system.
| nanomaterials | Wavelength | power density | Irradiation time | ROS detection method | PTT conversion efficiency | Model system | Ref |
| black TiO2 (PEG-modified) | 808 nm | 2 W·cm-2 | In vitro experiments: 0-5 min (the optimal killing effect is achieved at 5 min); In vivo experiments: 5 min per irradiation, with consecutive irradiation for 3 days | Not specifically performed | 40.8% | In vitro: human breast cancer MCF-7 cells, murine breast cancer 4T1 cells; In vivo: 4T1 breast cancer-bearing Balb/C mice | [17] |
| Mesoporous black TiO2 nanorods | LED | 150 W·cm-2 | 15 min (In vitro) | 1.•OH: HPF probe + fluorescence detection; 2. EPR + DMPO spin trapping | Not specifically performed | HeLa cervical cancer cells | [26] |
| Fe3O4-Black TiO2 | 808 nm, 671 nm | 808 nm: 0.7-1.3 W·cm-2; 671 nm: 0.5-1.0 W·cm-2 | In vitro:5-8 min; In vivo: 5 min | 1. 1O2: DPBF probe + UV-Vis spectroscopy detection; 2. Intracellular ROS: 2',7'- DCFH-DA probe + laser confocal microscopy observation | 808 nm:61.8%; 671 nm:75.4% | In vitro: MCF-7 human breast cancer cells; In vivo: MCF-7 breast cancer-bearing nude mice | [28] |
| Black TiO2@mSiO2@Gd/R848 | 808 nm | 0.5-2.0 W·cm-2 | In vitro: 5-10 min; In vivo: 5 min | Not specifically performed | Not specifically performed | In vitro: Panc02 pancreatic cancer cells; In vivo: Panc02 pancreatic cancer-bearing C57BL/6 mice | [27] |
ROS: reactive oxygen species; PTT: photothermal therapy; EPR: electron paramagnetic resonance; DMPO: 5,5-dimethyl-1-pyrroline N-oxide; HPF: hydroxyphenyl fluorescein; DPBF: 1,3 diphenylisobenzofuran; UV: ultraviolet; LED: light emitting diode.
4. Summary and Prospects
This review systematically elaborates on the research progress of black TiO2 in the field of cancer treatment. As a novel functional material, it possesses unique crystal core-disordered shell structure, which endows it with excellent broadband absorption, and notably enhances its light absorption in the near-infrared region. Black TiO2 has become increasingly popular in the medical field due to its unique broadband absorption, high near-infrared light penetration, and tunable defect structure characteristics. Its unique properties underpin nearly all its current applications. Black TiO2 can act as an efficient photosensitizer to generate reactive oxygen species, and a photothermal agent to achieve local thermal ablation. Moreover, by compounding with other functional materials (such as Au nanoclusters, Fe3O4, mesoporous silica, etc.), it shows great potential for applications in integrated strategies, such as imaging-guided therapy and immunotherapy. However, our understanding of the complexity of black TiO2 are still incomplete. At the defect level, the existence forms, stability of low-valence ions and oxygen vacancies, as well as their regulatory mechanisms on PDT/PTT efficiency, remain unclear. The limitations in detection techniques further hinder precise defect optimization. In terms of biosafety, the issues of long-term in vivo accumulation risk and insufficient biodegradability have not been fully resolved, which may induce chronic inflammation. Regarding therapeutic efficacy, improvements in visible-light activity are limited, and deep-tissue therapeutic effects relying on near-infrared light still requires optimization. Future research should focus on the practical needs of cancer treatment. It is necessary to further deepen the research on the defect regulation mechanism and biological interactions of black TiO2. Specifically, advanced characterization techniques should be integrated to clarify the defect-efficacy relationship, stimuli-responsive systems need to be designed to enhance biosafety, and standardized synthesis and evaluation systems should be established. Additionally, efforts should be made to promote the synergistic integration of black TiO2 with immunotherapy and gene therapy, while strengthening the complementary advantages with other phototherapeutic materials. Ultimately, these strategies will facilitate the translation of black TiO2 from basic research to clinical applications in precise, combined cancer therapy.
Authors contribution
Pei YF: Writing-original draft, investigation, formal analysis.
ao XY, Wen XM: Methodology, data curation, investigation
Chen LY: Writing-review & editing, validation.
Conflicts of interest
Liangyu Chen is an Editorial Board Member of BME Horizon. The other authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
None.
Copyright
© The Author(s) 2026.
References
-
1. Garg P, Malhotra J, Kulkarni P, Horne D, Salgia R, Singhal SS. Emerging therapeutic strategies to overcome drug resistance in cancer cells. Cancers. 2024;16(13):2478.[DOI]
-
2. Alvarez N, Sevilla A. Current advances in photodynamic therapy (PDT) and the future potential of PDT-combinatorial cancer therapies. Int J Mol Sci. 2024;25(2):1023.[DOI]
-
3. Dai T, Ren W, Wu A. Cancer theranostics of black TiO2 nanoparticles. In: Wu R, Ren W, editors. TiO2 Nanoparticles: Applications in Nanobiotechnology and Nanomedicine. Weinheim: Wiley-VCH; 2020. p. 185-215.[DOI]
-
4. Wan H, Zhang Y, Zhang W, Zou H. Robust two-photon visualized nanocarrier with dual targeting ability for controlled chemo-photodynamic synergistic treatment of cancer. ACS Appl Mater Interfaces. 2015;7(18):9608-9618.[DOI]
-
5. Shi J, Li J, Wang Y, Zhang CY. TiO2-based nanosystem for cancer therapy and antimicrobial treatment: A review. Chem Eng J. 2022;431:133714.[DOI]
-
6. Zhang H, Wang J, Zhang W, Han D, Zhang A. TiO2/SiO2-NHOC-FA nanocomposite as a photosensitizer with targeting ability for photocatalytic killing MCF-7 cells in vitro and its mechanism exploration. Photochem Photobiol. 2021;97(2):398-407.[DOI]
-
7. Chen X, Liu L, Yu PY, Mao SS. Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science. 2011;331(6018):746-750.[DOI]
-
8. Yan X, Li Y, Xia T. Black titanium dioxide nanomaterials in photocatalysis. Int J Photoenergy. 2017;2017:8529851.[DOI]
-
9. Yang C, Wang Z, Lin T, Yin H, Lü X, Wan D, et al. Core-shell nanostructured “black” rutile titania as excellent catalyst for hydrogen production enhanced by sulfur doping. J Am Chem Soc. 2013;135(47):17831-17838.[DOI]
-
10. Wu T, Fan J, Li Q, Shi P, Xu Q, Min Y. Palladium nanoparticles anchored on anatase titanium dioxide-black phosphorus hybrids with heterointerfaces: Highly electroactive and durable catalysts for ethanol electrooxidation. Adv Energy Mater. 2018;8:1701799.[DOI]
-
11. An HR, Park SY, Huh JY, Kim H, Lee YC, Lee YB, et al. Nanoporous hydrogenated TiO2 photocatalysts generated by underwater discharge plasma treatment for solar photocatalytic applications. Appl Catal B Environ. 2017;211:126-136.[DOI]
-
12. Hou X, Li Y, Zhang H, Lund PD, Kwan J, Tsang SCE. Black titanium oxide: Synthesis, modification, characterization, physiochemical properties, and emerging applications for energy conversion and storage, and environmental sustainability. Chem Soc Rev. 2024;53(21):10660-10708.[DOI]
-
13. Song H, Li C, Lou Z, Ye Z, Zhu L. Effective formation of oxygen vacancies in black TiO2 nanostructures with efficient solar-driven water splitting. ACS Sustainable Chem Eng. 2017;5(10):8982-8987.[DOI]
-
14. Zhou L, Cai M, Zhang X, Cui N, Chen G, Zou GY. In-situ nitrogen-doped black TiO2 with enhanced visible-light-driven photocatalytic inactivation of Microcystis aeruginosa cells: Synthesization, performance and mechanism. Appl Catal B Environ. 2020;272:119019.[DOI]
-
15. Jiang X, Yan Z, Zhang J, Gao J, Huang W, Shi Q, et al. Mesoporous hollow black TiO2 with controlled lattice disorder degrees for highly efficient visible-light-driven photocatalysis. RSC Adv. 2019;9(63):36907-36914.[DOI]
-
16. Haider S, Nawaz R, Anjum M, Haneef T, Oad VK, Uddinkhan S, et al. Property-performance relationship of core-shell structured black TiO2 photocatalyst for environmental remediation. Front Environ Sci Eng. 2023;17(9):111.[DOI]
-
17. Ren W, Yan Y, Zeng L, Shi Z, Gong A, Schaaf P, et al. A near infrared light triggered hydrogenated black TiO2 for cancer photothermal therapy. Adv Healthc Mater. 2015;4(10):1526-1536.[DOI]
-
18. Su Y, Zhang W, Chen S, Yao D, Xu J, Chen X, et al. Engineering black titanium dioxide by femtosecond laser filament. Appl Surf Sci. 2020;520:146298.[DOI]
-
19. Patil SB, Phattepur H, Nagaraju G, Gowrishankar BS. Highly distorted mesoporous S/C/Ti3+ doped black TiO2 for simultaneous visible light degradation of multiple dyes. New J Chem. 2020;44(23):9830-9836.[DOI]
-
20. Andronic L, Enesca A. Black TiO2 synthesis by chemical reduction methods for photocatalysis applications. Front Chem. 2020;8:565489.[DOI]
-
21. Sinhamahapatra A, Jeon JP, Yu JS. A new approach to prepare highly active and stable black titania for visible light-assisted hydrogen production. Energy Environ Sci. 2015;8(12):3539-3544.[DOI]
-
22. Shang M, Hu H, Lu G, Bi Y. Synergistic effects of SrTiO3 nanocubes and Ti3+ dual-doping for highly improved photoelectrochemical performance of TiO2 nanotube arrays under visible light. J Mater Chem A. 2016;4(16):5849-5853.[DOI]
-
23. Zhan XP, Wang YJ, Su Y, Li MT, Zang HW, Xia H, et al. Micro–nano-texturing inner surfaces of small-caliber high aspect ratio and superhydrophobic artificial vessels using femtosecond laser filamenting pulses. Adv Materials Inter. 2018;5(23):1801148.[DOI]
-
24. Rajaraman TS, Parikh SP, Gandhi VG. Black TiO2: A review of its properties and conflicting trends. Chem Eng J. 2020;389:123918.[DOI]
-
25. Ni W, Li M, Cui J, Xing Z, Li Z, Wu X, et al. 808nm light triggered black TiO2 nanoparticles for killing of bladder cancer cells. Mater Sci Eng C. 2017;81:252-260.[DOI]
-
26. Vélez-Peña E, Jiménez VA, Manzo-Merino J, Melin V, Contreras D, Alderete JB, et al. Visible light-activated mesoporous black titania nanorods for enhanced chemo-photodynamic cancer therapy. J Drug Deliv Sci Technol. 2025;106:106713.[DOI]
-
27. Xu L, Wu R, Ni J, Jin L, Xu K, Zhu Y, et al. Black TiO2-based nanoparticles as Toll-like receptor stimulator delivery system for enhanced photothermal-immunotherapy of pancreatic cancer. Cancer Nano. 2024;15:27.[DOI]
-
28. Saeed M, Iqbal MZ, Ren W, Xia Y, Khan WS, Wu A. Tunable fabrication of new theranostic Fe3O4-black TiO2 nanocomposites: Dual wavelength stimulated synergistic imaging-guided phototherapy in cancer. J Mater Chem B. 2019;7(2):210-223.[DOI]
-
29. Guo X, Wen C, Xu Q, Ruan C, Shen XC, Liang H. A full-spectrum responsive B-TiO2@SiO2–HA nanotheranostic system for NIR-II photoacoustic imaging-guided cancer phototherapy. J Mater Chem B. 2021;9(8):2042-2053.[DOI]
-
30. Bhole R, Bonde C, Kadam P, Wavwale R. A comprehensive review on photodynamic therapy (PDT) and photothermal therapy (PTT) for cancer treatment. Turk J Oncol. 2021;36(1).[DOI]
-
31. Vines JB, Yoon JH, Ryu NE, Lim DJ, Park H. Gold nanoparticles for photothermal cancer therapy. Front Chem. 2019;7:167.[DOI]
-
32. Ou M, Lin C, Wang Y, Lu Y, Wang W, Li Z, et al. Heterojunction engineered bioactive chlorella for cascade promoted cancer therapy. J Control Release. 2022;345:755-769.[DOI]
-
33. Zhang H, Zhang Y, Zhang Y, Li H, Ou M, Yu Y, et al. Catalytic activity of violet phosphorus-based nanosystems and the role of metabolites in tumor therapy. Nat Commun. 2024;15:6783.[DOI]
-
34. Tan CF, Azmansah SAB, Zhu H, Xu QH, Ho GW. Photoelectrocatalysis: Spontaneous electroless galvanic cell deposition of 3D hierarchical and interlaced S–M–S heterostructures. Adv Mater. 2017;29:adma.201770001.[DOI]
-
35. Hua D, Wu X, Wu Z, Fan C, Wang J, He W, et al. Nanoparticles, a promising treatment for gastric cancer. Smart Mater Med. 2025;6(1):23-55.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite




