Yuanmin Zhang, Key Laboratory of Cell and Biomedical Technology of Shandong Province, Jining 272000, Shandong, China; Affiliated Hospital of Jining Medical University, Jining Medical University, Jining 272000, Shandong, China. E-mail: yuanminzhang@mail.jnmc.edu.cn
Yujie Liang, Key Laboratory of Cell and Biomedical Technology of Shandong Province, Jining 272000, Shandong, China; Affiliated Hospital of Jining Medical University, Jining Medical University, Jining 272000, Shandong, China. E-mail: liangyjie@126.com
Abstract
Adipose-derived mesenchymal stem cells (ADSCs) are increasingly recognized as a promising therapeutic tool in regenerative medicine, particularly for skin wound repair. However, achieving consistent and effective healing remains a clinical challenge, especially in chronic or complex wounds. ADSC-derived exosomes (ADSC-Exos) have emerged as a key cell-free alternative, functioning as nanoscale messengers that transfer bioactive molecules to coordinate tissue regeneration. Unlike cell-based therapies, ADSC-Exos offer enhanced safety, stability, and logistical advantages while retaining potent regulatory capacity. They orchestrate multiple stages of wound healing by promoting angiogenesis, modulating inflammation, and accelerating re-epithelialization. This review first evaluates the relative benefits and limitations of various mesenchymal stem cells-derived cell populations for wound therapy. We then systematically examine the molecular mechanisms and functional roles of ADSC-Exos in skin repair, highlighting recent advances in their isolation, engineering, and delivery. Furthermore, we discuss current challenges and strategic insights for clinical translation. By integrating existing evidence with future perspectives, this review aims to guide the development of ADSC-Exos-based acellular therapies toward improved wound regeneration outcomes.
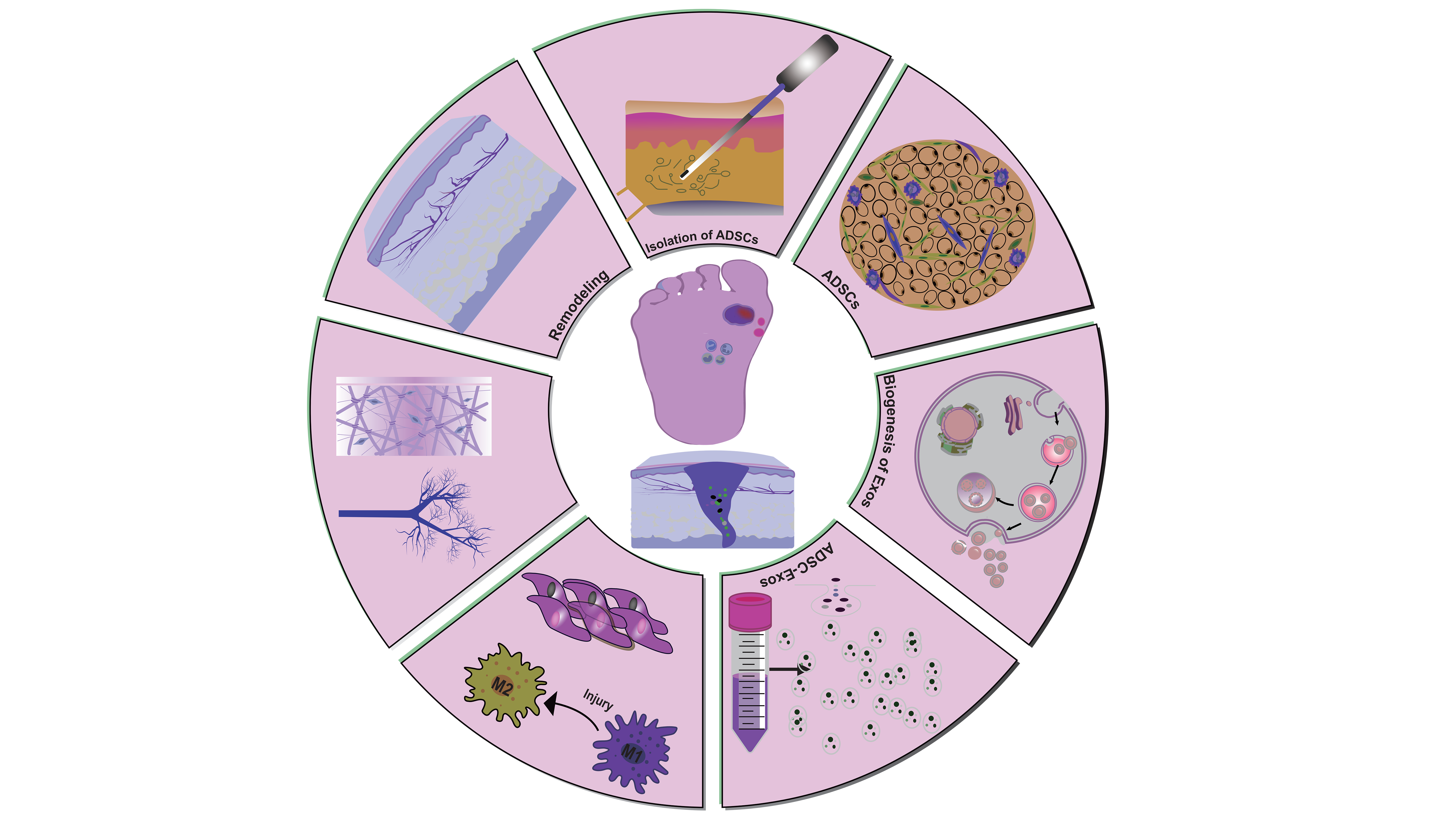
Graphical Abstract
Keywords
1. Introduction
Chronic wounds are generally defined as skin injuries that fail to achieve anatomical repair and functional reconstruction after a healing process exceeding 90 days, with a high incidence rate among diabetic patients, patients with vascular diseases, the elderly, and immunocompromised populations. Such wounds not only severely affect patients’ quality of life but also are prone to complications such as local infection, cellulitis, and osteomyelitis, in severe cases, they can lead to amputation and even endanger life through sepsis[1].Statistical data show that chronic wounds affect approximately 2% of the global population and cause an estimated 300,000 deaths each year, among which burns and diabetic foot ulcers (DFUs) are the most clinically challenging manifestations[2-4]. The healing ability of skin wounds is closely related to the defect area: small- to medium-sized defects can be repaired through local epidermal cell proliferation and migration or autologous skin transplantation, while large-area wounds require surgical intervention. Although autologous skin transplantation is the preferred treatment, its clinical application is still restricted by issues such as insufficient donor sites, postoperative scar hyperplasia, and mismatched cosmetic outcomes.
Wound healing is a complex cascade that involves multiple cell types, including inflammatory cells, fibroblasts, and keratinocytes, as well as diverse bioactive factors such as growth factors, cytokines, and matrix metalloproteinases. This process progresses through three sequential and interdependent phases: inflammation, proliferation, and remodeling, each governed by tightly regulated molecular networks essential for successful tissue repair. Numerous factors, including wound size, anatomical location, local blood supply, and the intensity of the inflammatory response, can substantially influence healing outcomes. Although advanced technologies such as growth factor therapy and engineered skin substitutes have been gradually introduced into clinical practice, current treatments still face limitations. These include the short half-life and low bioavailability of growth factors, immune rejection risks associated with engineered grafts, and poor adaptability, which collectively contribute to low healing rates in many clinical settings[5,6]. In this context, the rapid advancement of regenerative medicine has opened new avenues for treating chronic wounds. Over the past decade, accumulating preclinical and clinical evidence has demonstrated that ADSCs, owing to their abundant availability, accessibility, and potent paracrine activity, hold considerable potential to accelerate wound healing and improve outcomes in refractory chronic wounds[7]. Recent mechanistic studies further indicate that the therapeutic benefits of ADSCs rely not on direct cellular engraftment, but rather on their secretion of ADSC-Exos. These nanosized vesicles function as key intercellular carriers, delivering bioactive molecules that modulate cellular behavior and molecular networks across all stages of wound repair. Importantly, ADSC-Exos bypass several risks linked to whole-cell therapies, such as tumorigenicity, immune rejection, and poor cell survival, thereby offering a safer and more effective strategy for chronic wounds that respond inadequately to conventional treatments[8,9].
2. Advantages of ADSC-Exos for Skin Wound Healing
Despite their therapeutic potential, ADSC present several drawbacks in the treatment of skin injuries, such as infusion-related toxicity[10], immune reactions[11], tumorigenic risks[11,12], and ethical concerns[13]. The therapeutic utility of ADSCs is limited by their short retention time at the target site and a dramatic reduction in cell number following transplantation[14,15]. Studies report that the survival rate of human ADSCs falls below 1% one week post-transplantation, and concerns regarding their potential oncogenic properties further restrict their biomedical application[16,17]. These critical limitations have severely hindered the clinical translation and practical application of ADSC-based therapies for skin injuries, driving the urgent need to explore safer, more stable, and equally effective alternative therapeutic modalities that circumvent the aforementioned drawbacks. To overcome these limitations, exosomes have emerged as a promising alternative to stem cell-based therapies. Exosomes are nanoscale extracellular vesicles secreted by cells, characterized by a lipid bilayer membrane and often considered functional replicas of their parental cells. These exosomes not only lack tumor-promoting activity but also effectively encapsulate and protect key soluble molecules, such as inflammatory factors, growth factors, transcription factors, and RNAs, essential for tissue repair. This encapsulation slows molecular degradation and extends the duration of therapeutic activity[18]. Furthermore, ADSC-Exos demonstrate favorable storage stability, retaining their protein content without significant loss when kept in phosphate-buffered saline (PBS) at 4 °C for at least 24 hours[19]. Consequently, exosome-based cell-free therapy has emerged as a promising new direction in regenerative medicine. Table 1 summarizes the key advantages of ADSC-Exos over traditional stem cell therapies.
| Advantages | MSCs | ADSCs | ADSC-Exos | BMSC-Exos | References |
| Risk of tumorigenicity | Risk of tumorigenicity; genetic mutations; dual tumor-promoting/suppressing roles | Non-tumorigenic; promote tumor progression via microenvironment modulation | Non-tumorigenic; carry regenerative cargo; enhanced therapeutic safety profile | Non-tumorigenic; if parental BMSCs are passaged long-term, the level of miR-155 in exosomes increases, which may slightly promote tumor cell migration | [20-25] |
| Stability | Donor-dependent variability; functional decay with passaging; challenging long-term preservation | Subject to source cell heterogeneity; superior storage stability compared to whole MSCs | Inherently stable; retains activity after 6 months; lyophilized formulations can stable for long time | Storage-stable, but requires full cold chain transportation; activity is significantly lost after more than 72 hours | [22,23,25-28,29,30] |
| Immune rejection | Low immunogenicity; induce rejection under allogeneic settings | Low immunogenicity; exhibit potent immunomodulatory capacity | Minimizes risks of immune rejection, target homing, and blood-brain barrier crossing | Low immunogenicity; as donor age increases, the level of TNF-α in exosomes slightly rises, causing local inflammatory responses | [23,25,29,31-33] |
| Manufacturing cost | High cost due to invasive extraction, limited donor availability, and low yield | Lower cost; readily accessible source from adipose tissue with significantly higher cell yield per harvest | Reduced donor dependence through scalable culture and isolation | Affected by donors, unstable source of raw materials, and higher culture and manufacturing costs than ADSC-Exos | [23,30,34-38] |
ADSCs: adipose-derived mesenchymal stem cells; BMSCs: bone marrow mesenchymal stem cells; ADSC-Exos: ADSC-derived exosomes; BMSC-Exos: bone marrow MSC-derived exosomes.
In skin wound healing, the therapeutic value and safety profiles of different adipose-derived cell populations vary considerably. While mesenchymal stem cells (MSCs) possess regenerative potential, they carry a recognized risk of tumorigenicity, which may be exacerbated under conditions of long-term culture or within immunosuppressive microenvironments. Their stability is further compromised by donor variability and functional decline during serial passaging, and their activity is often diminished after cryopreservation. Bone marrow-derived MSCs present additional challenges, including complex isolation and culture procedures, along with the risk of immune rejection in allogeneic settings, despite having some immunomodulatory capacity[20-23,35]. ADSCs, in contrast, are obtained through minimally invasive procedures and lack direct tumorigenic potential, while still exhibiting strong reparative properties. However, they also display significant heterogeneity, donor age and adipose tissue site influence their functional activity, and they remain susceptible to functional decline during preservation and expansion. Although ADSCs exhibit lower immunogenicity than MSCs, the risk of immune rejection cannot be entirely excluded[26-28,36]. As a key cell-free therapeutic alternative, bone marrow MSC-derived exosomes (BMSC-Exos) offer distinct repair capabilities, yet their clinical applicability and safety features differ notably from those of ADSC-Exos. BMSC-Exos promote angiogenesis, fibroblast proliferation, and collagen deposition largely through the delivery of molecules such as miR-126-3p and miR-29a, which activate PI3K/Akt and Ras/Erk signaling pathways. Moreover, their cargo is enriched with bone metabolism-related molecules like RUNX2 and OCN, providing a synergistic repair advantage in complex wounds involving bone exposure, an area where ADSC-Exos show relative limitation[23,30,32]. Although BMSC-Exos lack a complete cellular structure and are not directly tumorigenic in long-term animal studies, yet, after extended passaging of the parent BMSCs, elevated levels of exosomal miR-155 have been shown to slightly promote tumor cell migration in vitro, suggesting a need for vigilance in patients with a history of malignancy[23]. By comparison, ADSC-Exos present several irreplaceable advantages: their acellular nature eliminates tumorigenic risk, and they accelerate wound healing through paracrine delivery of bioactive molecules. They exhibit particular promise in refractory diabetic wounds, especially when combined with biomaterial scaffolds[24,29]. Their lipid bilayer structure confers considerable stability to the encapsulated cargo, and lyophilization coupled with standardized production protocols enables reliable long-term storage while reducing batch-to-batch variability[30,37]. Low immunogenicity minimizes rejection risk, and they can modulate immune cell functions to suppress excessive inflammation. Targeted homing ability further enhances local efficacy and safety[33]. Moreover, ADSC-Exos can be efficiently harvested from large-scale ADSC cultures without the need for viable cell preservation, thereby simplifying production, lowering costs, reducing donor dependence, and improving suitability for broad clinical application[37].
3. Biogenesis and Uptake Mechanism of ADSC-Exos
3.1 Biogenesis of ADSC-Exos
The biogenesis of ADSC-Exos primarily follows the endosomal pathway. The process initiates with the endocytic invagination of the plasma membrane in ADSCs, forming early endosomes. These then mature into multivesicular bodies (MVBs) through intraluminal budding, encapsulating intraluminal vesicles (ILVs), which are the direct precursors of ADSC-Exos[39-41]. Subsequently, the transport of mature MVBs to the cell periphery is regulated by Rab GTPases, notably with Rab27a. Their final fusion with the plasma membrane is mediated by SNARE proteins, including VAMP8 and Syntaxin-1, leading to the release of ILVs into the extracellular space as functional ADSC-Exos[42-44]. Furthermore, this biogenesis process can be modulated by external signals. For instance, adiponectin secreted by adipocytes binds to T-cadherin on ADSCs and promotes MVB accumulation, thereby enhancing the production of ADSCs-Exos[45].
3.2 Uptake mechanism of ADSC-Exos
The cellular uptake of ADSC-Exos primarily occurs through endocytosis, which comprises several temperature-sensitive subtypes dependent on cytoskeletal remodeling. Clathrin-mediated endocytosis relies on the coordinated function of clathrin heavy chain and dynamin-2. The specific interaction between CD63 on ADSC-Exos and the transferrin receptor on target cells can significantly enhance uptake efficiency via this pathway[46-48]. Lipid raft-mediated endocytosis depends on cholesterol and sphingolipid enrichment in the membrane and is positively regulated by Annexin A2 and flotillin proteins[49,50]. Additionally, transmembrane ligands on ADSC-Exos, such as MHC molecules, TNF-related factors, and tetraspanins CD63/CD81, can directly bind to corresponding receptors (e.g., integrin αvβ3 and transferrin receptor) on target cells, including skin fibroblasts and immune cells. These interactions can trigger downstream signals involved in immune regulation, proliferation, or apoptosis without requiring internalization[51]. Alternatively, ADSC-Exos may directly fuse with the target-cell plasma membrane through the coordinated action of SNARE family proteins (e.g., VAMP8 and Syntaxin-1) and Rab GTPases, facilitated by lipid-raft structures and integrin-mediated membrane contacts. This fusion enables the direct delivery of cargo, such as microRNAs (miRNAs) and functional proteins into the cytosol (Figure 1)[52].
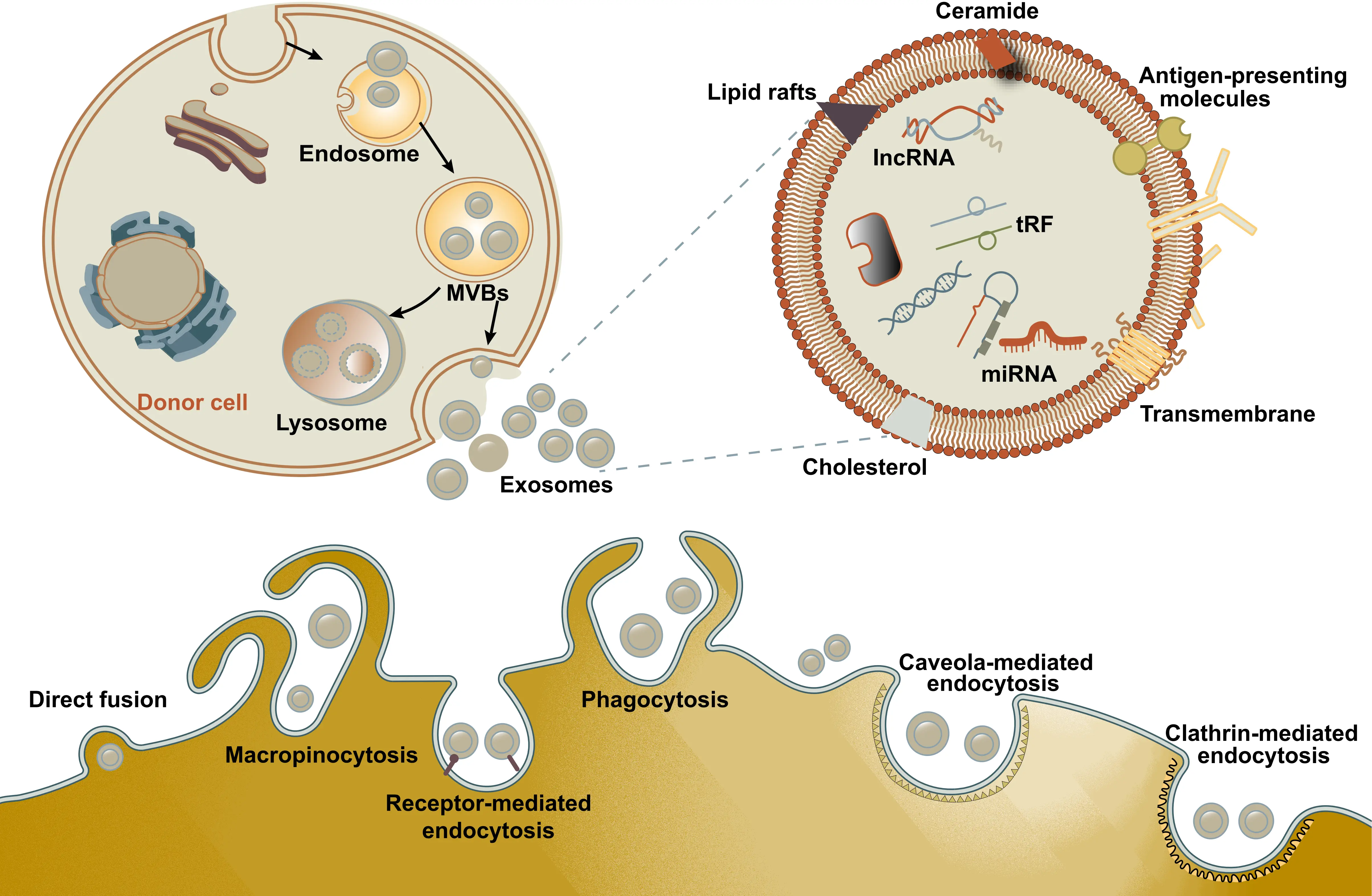
Figure 1. Schematic representation of EVs biogenesis, structure, and their uptake mechanism. EVs biogenesis encompasses cargo sorting, formation and maturation of MVBs, transport of MVBs, and fusion of MVBs with the plasma membrane. Upon release, the generated exosomes enter target cells through mechanisms such as receptor-mediated endocytosis, clathrin-coated pits, lipid rafts, phagocytosis, caveolae-mediated uptake, and macropinocytosis. EVs: extracellular vesicles; MVBs: multivesicular bodies.
3.3 The differences in exosome isolation methods and research limitations
In current ADSC-Exos-related studies, the diversity of isolation methods is a key factor leading to result heterogeneity and a major bottleneck restricting clinical translation. Commonly used isolation methods include differential centrifugation, ExoQuick solution precipitation, density gradient centrifugation, and membrane filtration, with significant differences in yield, purity, and retention of biological activity among different methods.
Differential centrifugation is the most commonly used method, but it is cumbersome and time-consuming, and prone to contamination by protein aggregates and cell debris, resulting in insufficient exosome purity[37]. ExoQuick precipitation is simple and time-efficient, but the precipitation reagents may affect the biological activity of exosomes, and it is difficult to remove high-abundance plasma proteins[22]. Density gradient centrifugation can obtain high-purity exosomes, but the yield is low, and the high centrifugal speed during the process may damage the exosome membrane structure and affect its function[40]. A 2025 methodological study confirmed that the combined use of differential centrifugation and 80 kDa membrane filtration can significantly improve exosome purity while ensuring yield, with its pro-angiogenic activity increased by 30% compared to single differential centrifugation[53].
In addition, the quantitative standards for exosomes are inconsistent across different studies (e.g., protein concentration, particle number, RNA content), and there is a lack of unified quality control indicators, making it difficult to conduct horizontal comparisons of research results[39]. For example, some studies use protein concentration as the dosage standard, ignoring the integrity of exosome particles and the content of cargo, which may lead to bias in actual therapeutic effects[50]. These methodological limitations suggest that future efforts should focus on establishing standardized ADSC-Exos isolation, purification, and quantification processes to ensure the reliability and reproducibility of research results.
4. Therapeutic Effects of ADSC-Exos in Wound Repair
Numerous preclinical studies on the application of natural ADSC-Exos in wound repair (Table 2) have clearly demonstrated their therapeutic value and related mechanisms. However, the wound healing process itself is extremely complex, regulated by multiple factors such as the quantity and function of growth factors, macrophages, keratinocytes, fibroblasts, and collagen deposition. ADSC-Exos participate in wound repair precisely through multiple pathways, including regulating inflammatory responses, accelerating cell proliferation and differentiation, promoting cell migration and angiogenesis, influencing extracellular matrix (ECM) remodeling, and inhibiting scar formation (Figure 2).
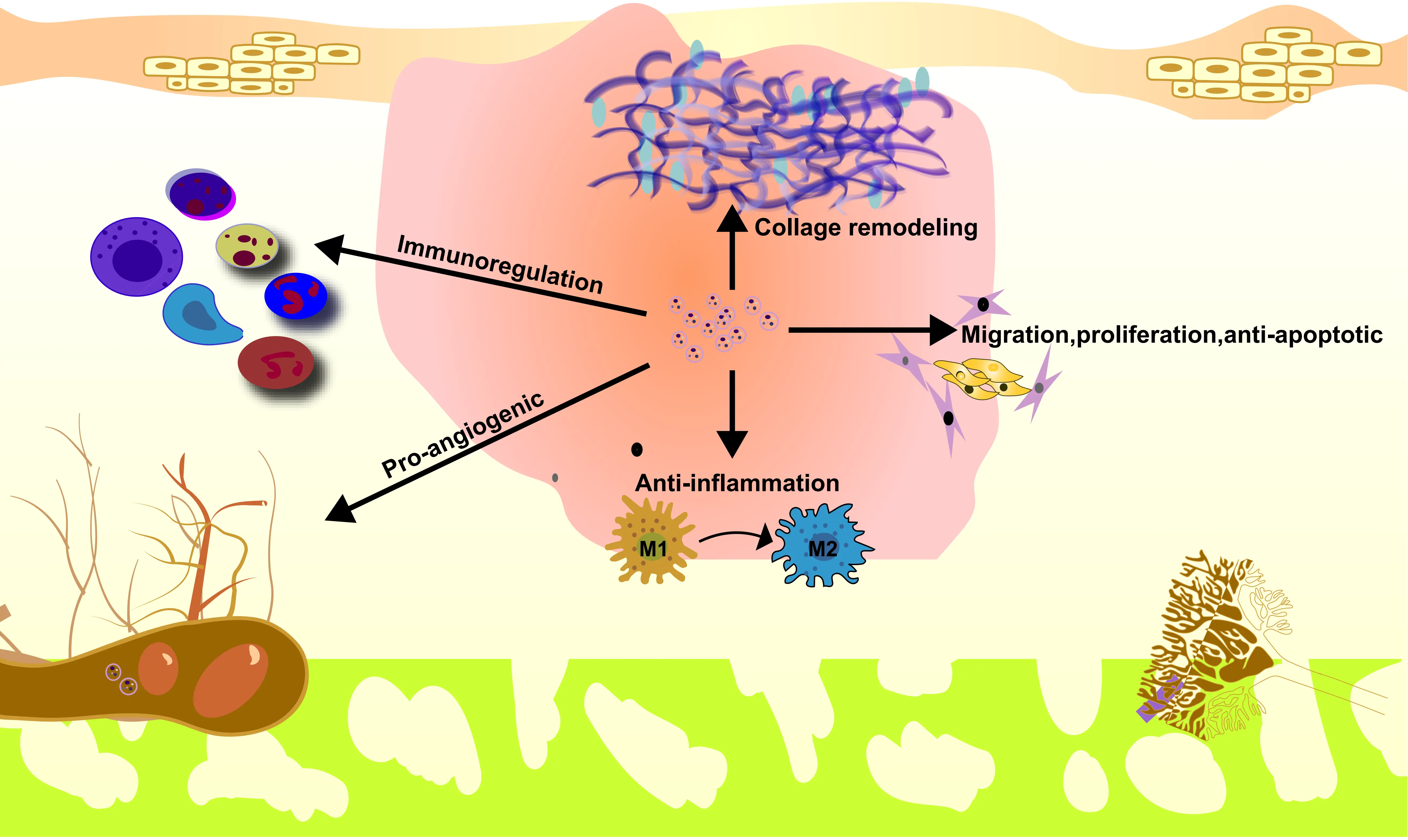
Figure 2. ADSC-Exos have shown to be more effective in promoting cell proliferation and migration, reducing oxidative stress levels, enhancing collagen expression, and suppressing the expression of inflammatory factors, thereby more effectively enhancing skin wound repair. ADSC: adipose-derived mesenchymal stem cell; ADSC-Exos: ADSC-derived exosomes.
| Cell source | Isolation method | Wound models | EV dosages/Administration | Time | Effect | Possible mechanism | Reference |
| Human | Differential centrifugation filtration, centrifugation with 80 kDa membrane | 10 mm × 10 mm full-thickness square wounds on the back of C57BL/6 mice | 100 μL per dose (concentration 25 μg/mL or 50 μg/mL), perilesional injection on days 1, 3, 7, and 10 post-wounding | 14 days | Promoted wound healing and angiogenesis; increased CD31 expression in wound granulation tissue | miR-125a-3p inhibits PTEN and activates the PI3K/AKT signaling pathway | [54] |
| Human | Differential centrifugation | 4 mm full-thickness excisional wounds on the back of C57BL/6 diabetic mice | 200 μg/100 μL PBS, subcutaneous injection at 4 perilesional sites | 15 days | Inhibited the expression of pro-inflammatory factors (TNF-α, IL-6, etc.); promoted angiogenesis and M2-like macrophage polarization | circ-Snhg11 upregulates HIF-1α expression by sponging miR-144-3p | [55] |
| Human | Differential centrifugation | In vivo angiogenesis model with Matrigel plug in BALB/c nude mice | 100 μg/mL, subcutaneous injection mixed with HUVECs | 7 days | Promoted angiogenesis in vitro and in vivo; increased the number and tube length of vascular structures | miR-125a targets DLL4, inhibits its expression, and promotes endothelial tip cell formation | [56] |
| Human | Differential centrifugation | 10 mm circular full-thickness skin defects on the back of SD rats | 0.2 mL ADSC-Exos, injection around the wound bed | 5 weeks | Accelerated wound healing; increased collagen deposition and the number of new blood vessels | miR-126-3p targets PIK3R2, promotes fibroblast proliferation and migration, and HUVEC angiogenesis | [57] |
| Human | Differential centrifugation | 1 × 1 cm2 full-thickness skin defects on the back of BALB/c mice | 70 μg/100 μL PBS, subcutaneous injection for 5 consecutive days | 14 days | Significantly promotes wound healing in mouse skin | miR-192-5p targets IL-17RA and regulates the Smad signaling pathway | [58] |
| Human | Precipitation techniques by ExoQuick solution | 1 × 1 cm2 Circular wounds on C57BL/6 mice | 21 days | Significantly enhanced skin wound healing in mice | LncRNA Neat1 activates Ulk1-mediated autophagy by sponge-mediated adsorption of miR-17-5p | [59] | |
| Human | Precipitation techniques by ExoQuick solution | 1 × 1 cm size Full-thickness skin wound on the shaved dorsum of C57BL/6 mice | 5 μg in 100 μL PBS | 14 days | Promotes proliferation/migration of HSFs and accelerates wound closure | LncRNA MALAT1 upregulates FGF2 expression by inhibiting miR-378a | [60] |
| Human | Precipitation techniques by exosome extraction reagent | full-thickness skin wounds in male BALB/c mice | 200 μg in PBS | 12 days | Enhances proliferation of MDFs, reduces inflammation, and decreases collagen deposition | LncRNA XIST restores DDR2 expression by silencing miR-96-5p | [61] |
ADSC-Exos: ADSC-derived exosomes; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; HUVECs: human umbilical vein endothelial cells; PBS: phosphate-buffered saline; HSFs: human skin fibroblasts; MDFs: mouse dermal fibroblasts.
ADSC therapy primarily accelerates wound healing through combined application with dermal regeneration templates (DRT). DRT can significantly enhance the survival and colonization efficiency of ADSCs in the wound area, preventing cell loss[15]. ADSCs secrete growth factors such as CTGF, FGF, and HGF, as well as matrix metalloproteinases such as MMP3, through paracrine signaling[37,62]. On one hand, they promote keratinocyte proliferation, granulation tissue maturation, and re-epithelialization[37]; on the other hand, they precisely regulate extracellular matrix remodeling, inducing type III collagen deposition in the early stage to provide structural support, which gradually transitions to type I collagen subsequently[37]. Meanwhile, they inhibit the transformation of fibroblasts into myofibroblasts, reducing excessive collagen deposition. Ultimately, this achieves rapid healing of full-thickness wounds and significantly alleviates scar formation[37,62]. Compared with therapy using ADSCs, the application of ADSC-Exos as an autologous cell-free therapy not only exhibits effects similar to those of the parent cells but also effectively avoids issues such as immune rejection, limited survival of transplanted cells, ethical concerns, loss of function due to genetic mutations, and tumorigenic risks.
4.1 The effects of different components in ADSC-Exos on the process of skin wound healing
The abundant bioactive components in ADSC-Exos are the core mechanism underlying their promotion of skin wound healing. Different components form a synergistic repair network by targeting and regulating key cells and signaling pathways at various stages of wound healing. Among them, miRNAs serve as major functional carriers and exert precise regulatory effects: overexpressed miR-21-5p can promote the proliferation and migration of keratinocytes through the Wnt/β-catenin signaling pathway, accelerating re-epithelialization and vascular maturation in diabetic wounds[42]; miR-146a enhances fibroblast activity and promotes neovascularization by upregulating the expression of SERPINH1 and p-ERK, optimizing wound repair efficiency[43]; while miR-378 alleviates oxidative stress-induced damage in HaCaT cells by targeting and inhibiting caspase-3, creating a stable microenvironment for wound healing[44]. Long non-coding RNAs (lncRNAs) participate in regulation through the ceRNA mechanism: for example, lncRNA H19 upregulates SOX9 expression by sponging miR-19b, activating the Wnt/β-catenin pathway to promote the proliferation, migration, and extracellular matrix remodeling of skin fibroblasts[52]; lncRNA MALAT1 not only binds to miR-124 to activate the Wnt/β-catenin pathway but also inhibits cell apoptosis through the miR-378a/FGF2 axis, thereby dually promoting skin wound healing[57,58]. Although circular RNAs have been less studied, they have been confirmed to function by sponging target miRNAs: mmu_circ_0000250 upregulates SIRT1 expression by sponging miR-128-3p, activating autophagy and inhibiting cell apoptosis, thereby significantly accelerating diabetic wound healing[60]; mmu_circ_0001052 promotes angiogenesis in DFUs through the miR-106a-5p/FGF4/p38MAPK pathway[61]. Protein components are also indispensable: ADSC-Exos overexpressing Nrf2 can reduce oxidative stress damage in a high-glucose environment by inhibiting the secretion of reactive oxygen species and inflammatory factors, promoting the vascularization and healing of DFUs[63]; EGR-1 directly promotes the expression of vascular endothelial growth factor (VEGF-A) by regulating the lncRNA-SENCR/DKC1/VEGF-A axis, enhancing wound angiogenesis[64]. These components cooperate with each other, exerting effects in key processes such as inflammation regulation, cell proliferation, angiogenesis, and matrix remodeling, collectively forming the molecular basis for ADSC-Exos to efficiently promote skin wound healing[31,34].
4.2 ADSC-Exos promote diabetic wound repair
ADSC-Exos have shown considerable therapeutic potential in diabetic wound repair by promoting tissue repair through multiple synergistic mechanisms. Initially, they modulate the inflammatory microenvironment by downregulating pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) and promoting M2 macrophage polarization, thereby alleviating chronic inflammation[24]. Additionally, ADSC-Exos deliver bioactive molecules including miR-132 and miR-21, which activate signaling pathways such as PI3K/AKT, subsequently enhancing vascular endothelial cell proliferation, fibroblast migration, and collagen deposition. Loading ADSC-Exos onto hydrogel carriers such as ECM to achieve sustained release can maintain a high local concentration at the wound site, further enhancing their repair efficiency in refractory diabetic wounds, significantly increasing the wound closure rate and improving the quality of skin regeneration[63]. A Phase I clinical trial conducted in 2025 demonstrated promising clinical outcomes with ADSC-Exos in DFU treatment. In this study, eight patients received intralesional injections of ADSC-Exos (200 μg per dose) administered every two weeks. After 12 weeks of treatment, complete wound closure was achieved in six cases. No serious adverse events were reported, supporting the preliminary safety and efficacy of this exosome-based therapy[53]. Additionally, ADSC-Exos can reduce oxidative stress damage in diabetic wounds by upregulating the expression of antioxidant receptors such as Nrf2, providing new ideas for the clinical treatment of DFUs and other conditions[64]. These effects collectively address impaired angiogenesis and delayed tissue regeneration in diabetic wounds.
4.3 ADSC-Exos for cutaneous wound inflammation
ADSC-Exos regulate cutaneous wound inflammation at multiple levels to accelerate healing. Firstly, they inhibit the activation of skin T cells, as evidenced by reduced expression of the activation marker CD25, and decrease the secretion of pro-inflammatory cytokines such as IL-2 and IL-17A. Simultaneously, ADSC-Exos mitigate inflammation-induced T cell apoptosis by activating the PI3K/Akt pathway[60]. Secondly, they promote macrophage polarization toward the anti-inflammatory M2 phenotype, thereby reducing the release of inflammatory mediators including TNF-α and IL-6 and alleviating chronic inflammatory states[24,29]. Thirdly, ADSC-Exos suppress the excessive recruitment of dendritic epidermal T cells following acute injury, preventing uncontrolled inflammatory responses[60]. Furthermore, when delivered via carriers such as extracellular matrix hydrogels, ADSC-Exos achieve sustained local release, which effectively downregulates inflammation in both normal and diabetic chronic wounds. This creates a favorable microenvironment conducive to angiogenesis, cell proliferation, and tissue repair.
4.4 ADSC-Exos for skin burn wounds
Burns can be caused by thermal, electrical, or chemical agents, initiating a complex wound healing process shortly after injury. If deep burn wounds are not promptly repaired, excessive granulation tissue formation can occur, leading to impaired healing and increased susceptibility to infection. Studies indicate that ADSC-Exos regulate the proliferation and migration of fibroblasts derived from wound granulation tissue, thereby influencing the outcome and quality of wound repair[10]. ADSC-Exos modulate the inflammatory response in burn wounds by downregulating the expression of pro-inflammatory factors such as TNF-α and IL-6. For instance, human umbilical cord mesenchymal stem cell exosomes overexpressing miR-181c have been shown to effectively inhibit the TLR4 signaling pathway in burned rats, alleviating inflammation and promoting wound healing[65]. Furthermore, ADSC-Exos help mitigate excessive inflammation by regulating immune cell functions, such as modulating macrophage polarization, creating a microenvironment more conducive to burn wound repair[24,29]. In addition to inflammation modulation, ADSC-Exos promote several key repair processes in burn wounds. They accelerate fibroblast proliferation and migration to facilitate granulation tissue formation, enhance neovascularization by increasing the number of cells positive for vascular endothelial markers (e.g., CD31 and CD34), and improve local ischemia, collectively accelerating wound closure[24]. Moreover, ADSC-Exos optimize collagen deposition and arrangement through the regulation of collagen remodeling pathways, reducing the risk of hypertrophic scarring. When delivered via carriers such as ECM hydrogels, ADSC-Exos achieve sustained local release, maintaining high concentrations at the wound site and further enhancing their therapeutic efficacy in burn repair[24].
4.5 ADSC-Exos for cutaneous scar formation
ADSC-Exos exert multifaceted effects on skin wound healing and scar regulation. They reduce the expression of pro-inflammatory factors such as TNF-α and IL-6 at the wound site, thereby attenuating excessive inflammation and creating a microenvironment conducive to healing. In addition, ADSC-Exos concentration-dependently promote the proliferation and migration of keratinocytes (HaCaT cells), fibroblasts, and human umbilical vein endothelial cells (HUVECs). The pro-angiogenic effect on HUVECs is associated with exosomal microRNAs such as miR-128-3p and miR-125a-3p, which activate the PI3K/AKT pathway[24,54,65]. Moreover, ADSC-Exos enhance neovascularization in burn wounds by delivering miR-125a and miR-31 to endothelial cells, improving local ischemia and accelerating wound closure[56,66].
Scar formation involves multiple factors, including genetic predisposition, dysregulated cytokine secretion, excessive ECM deposition, impaired collagen degradation, abnormal fibroblast activity, and local hypoxia[67]. In regulating scar formation, ADSC-Exos act through several mechanisms: they inhibit fibroblast-to-myofibroblast differentiation, upregulate the ratio of transforming growth factor-β3 (TGF-β3) to TGF-β1, and increase the matrix metalloproteinase 3 (MMP3) to tissue inhibitor of metalloproteinase 1 (TIMP1) ratio in dermal fibroblasts. These changes promote the degradation and organized remodeling of aberrant collagen, reducing excessive scar hyperplasia in burn wounds. Studies have confirmed that ADSC-Exos significantly decrease incision scar size in mice and improve the collagen III/I ratio[68]. When loaded into ECM hydrogels for sustained local release, ADSC-Exos maintain high local concentrations, further enhance collagen alignment, and lower the risk of scar formation. Further research is needed to clarify the role of ADSC-Exos in scar suppression and their potential clinical application, with the aim of advancing toward scar-free wound healing.
5. Perspective and Conclusion
ADSC-Exos, as the key paracrine mediators of ADSCs, have emerged as a highly promising cell-free therapeutic strategy in skin wound repair. Their advantages, including high biocompatibility, low immunogenicity, ease of storage and transport, and the ability to circumvent the tumorigenic risks and ethical constraints associated with whole-cell therapies, position them as a compelling alternative to traditional stem cell-based approaches.
The therapeutic efficacy of ADSC-Exos is mediated through a coordinated multi-mechanistic network rather than a single pathway, encompassing anti-inflammation, pro-regeneration, and anti-scarring effects. Specifically, ADSC-Exos modulate the immune microenvironment by suppressing excessive T-cell activation, promoting M2 macrophage polarization, and reducing pro-inflammatory cytokines such as TNF-α and IL-6. Concurrently, they enhance tissue repair via the delivery of bioactive molecules (e.g., miR-125a, miR-31) that activate pro-angiogenic and proliferative pathways such as PI3K/AKT, thereby accelerating endothelial cell and fibroblast activity, collagen deposition, and neovascularization. Furthermore, ADSC-Exos mitigate pathological scarring by regulating the TGF-β3/TGF-β1 ratio, inhibiting fibroblast-to-myofibroblast differentiation, and promoting extracellular matrix remodeling through balanced MMP3/TIMP1 expression.
Despite promising preclinical outcomes and the growing integration of ADSC-Exos with advanced biomaterial carriers for complex wound management, significant scientific and translational challenges must be overcome to realize their full clinical potential. The path forward requires a concerted effort to address fundamental biological questions alongside rigorous standardization of manufacturing and clinical evaluation. Regarding core scientific issues, a deeper mechanistic understanding is imperative. First, the specific functional molecules (e.g., miRNAs, proteins, lncRNAs) and their synergistic or antagonistic regulatory networks within ADSC-Exos that mediate healing in distinct pathological microenvironments, such as the hyperglycemic, oxidative stress milieu of diabetic ulcers versus the necroinflammatory setting of burns, remain inadequately mapped. Crucially, direct in vivo validation of these interactions and their conservation across donor populations is lacking. Second, breakthroughs in targeted and adaptive delivery are needed. Current carrier systems primarily enable passive release, failing to dynamically respond to phase-specific wound cues (e.g., pH, protease activity). Furthermore, the limited targeting specificity of native exosomes leads to off-target uptake and efficacy loss. Future strategies must focus on engineering “smart” responsive biomaterials and exosome surface modifications to achieve spatiotemporally controlled delivery. Concerning key translational challenges, a multi-faceted approach is essential. Defining critical quality attributes (size, concentration, purity, bioactive cargo) and establishing robust, scalable production processes are foundational. The inherent heterogeneity of ADSC-Exos due to donor variability and culture conditions necessitates stringent quality control and traceability systems to ensure batch-to-batch consistency and clinical safety. Clinical trial design faces the hurdle of wound heterogeneity, complicating patient stratification and endpoint selection. Moreover, long-term safety data regarding potential immunogenicity or tissue accumulation are insufficient. Future trials must adopt optimized, stratified designs to generate high-level evidence for specific wound indications. The entire pipeline, from large-scale exosome production and isolation to functionalized carrier fabrication, must be optimized for cost-effectiveness to ensure clinical accessibility.
In summary, ADSC-Exos represent a versatile, cell-free platform with significant potential to improve wound healing outcomes across a spectrum of cutaneous injuries. With continued research into their mechanisms, delivery systems, and scalable production, ADSC-Exos are poised to transition from promising preclinical candidates to clinically viable therapies, ultimately supporting the broader adoption of exosome-based regenerative medicine in dermatology and wound care.
Acknowledgments
We confirm that Gemini-2.5-Pro was used for language polishing and grammar checking during the preparation of this manuscript. No AI tools were used to generate, interpret, or analyze any academic content, data, or conclusions.
Authors contribution
Ji H: Writing-original draft.
Liang Y: Conceptualization, project administration, writing-original draft, writing-review & editing.
Yu N, Shi Y: Visualization.
Xu L, Wen C: Investigation, formal analysis.
Xu X: Investigation, formal analysis, funding acquisition.
Zhang Y: Funding acquisition.
Conflicts of interest
Yujie Liang is a Youth Editorial Board Member of BME Horizon. The other authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by the Undergraduate Training Program for Innovation and Entrepreneurship (Grant No. 202410443001 and No. 202410443025), Taishan Scholar Youth Expert Program of Shandong Province (Grant No. tsqnz20231256), the National Natural Science Foundation of China (Grant No. 82401860), Shandong Provincial Natural Science Foundation (Grant No. ZR2025MS1471 and No. ZR2023QH148), the PhD Research Foundation of Affiliated Hospital of Jining Medical University (Grant No. 2022-BS-03 and No. 2022-BS-04).
Copyright
© The Author(s) 2026.
References
-
1. Reid IR, Billington EO. Drug therapy for osteoporosis in older adults. Lancet. 2022;399(10329):1080-1092.[DOI]
-
2. Martinengo L, Olsson M, Bajpai R, Soljak M, Upton Z, Schmidtchen A, et al. Prevalence of chronic wounds in the general population: Systematic review and meta-analysis of observational studies. Ann Epidemiol. 2019;29:8-15.[DOI]
-
3. Armstrong DG, Tan TW, Boulton AJM, Bus SA. Diabetic foot ulcers: A review. JAMA. 2023;330(1):62.[DOI]
-
4. Rubio JA, Jiménez S, Lázaro-Martínez JL. Mortality in patients with diabetic foot ulcers: Causes, risk factors, and their association with evolution and severity of ulcer. J Clin Med. 2020;9(9):3009.[DOI]
-
5. Lei C, Song JH, Li S, Zhu YN, Liu MY, Wan MC, et al. Advances in materials-based therapeutic strategies against osteoporosis. Biomaterials. 2023;296:122066.[DOI]
-
6. Gentile P, Garcovich S. Systematic review: Adipose-derived mesenchymal stem cells, platelet-rich plasma and biomaterials as new regenerative strategies in chronic skin wounds and soft tissue defects. Int J Mol Sci. 2021;22(4):1538.[DOI]
-
8. Hwang JH, Park YS, Kim HS, Kim DH, Lee SH, Lee CH, et al. Yam-derived exosome-like nanovesicles stimulate osteoblast formation and prevent osteoporosis in mice. J Control Release. 2023;355:184-198.[DOI]
-
9. Liu H, Zhang H, Wang S, Cui J, Weng W, Liu X, et al. Bone-targeted bioengineered bacterial extracellular vesicles delivering siRNA to ameliorate osteoporosis. Compos Part B Eng. 2023;255:110610.[DOI]
-
10. Mazini L, Rochette L, Admou B, Amal S, Malka G. Hopes and limits of adipose-derived stem cells (ADSCs) and mesenchymal stem cells (MSCs) in wound healing. Int J Mol Sci. 2020;21(4):1306.[DOI]
-
11. Fang W, Wang G, Lin L, Ajoolabady A, Ren J. Extracellular vesicles in skin health and diseases. Life Sci. 2025;378:123813.[DOI]
-
13. Czop JK, Jałowska M. Stem cells in plastic surgery and aesthetic medicine. Adv Dermatol Allergol. 2023;40(4):504-509.[DOI]
-
14. Yoshizumi K, Saito N, Wu Y, Shirado T, Asahi R, Mori M, et al. Adipose-derived stem cells and wound healing are progressively impaired long-term after radiotherapy in mice. Plast Reconstr Surg Glob Open. 2025;13(1):e6419.[DOI]
-
15. Xu J, Chen X, Wang J, Zhang B, Ge W, Wang J, et al. An ADSC-loaded dermal regeneration template promotes full-thickness wound healing. Regen Ther. 2024;26:800-810.[DOI]
-
16. Agrawal H, Shang H, Sattah AP, Yang N, Peirce SM, Katz AJ. Human adipose-derived stromal/stem cells demonstrate short-lived persistence after implantation in both an immunocompetent and an immunocompromised murine model. Stem Cell Res Ther. 2014;5(6):142.[DOI]
-
17. De Pieri A, Rochev Y, Zeugolis DI. Scaffold-free cell-based tissue engineering therapies: Advances, shortfalls and forecast. npj Regen Med. 2021;6:18.[DOI]
-
18. Cai Y, Li J, Jia C, He Y, Deng C. Therapeutic applications of adipose cell-free derivatives: A review. Stem Cell Res Ther. 2020;11:312.[DOI]
-
19. Evtushenko EG, Bagrov DV, Lazarev VN, Livshits MA, Khomyakova E. Adsorption of extracellular vesicles onto the tube walls during storage in solution. PLoS One. 2020;15(12):e0243738.[DOI]
-
20. Jeung S, Kim S, Ah J, Seo S, Jan U, Lee H, et al. Exploring the tumor-associated risk of mesenchymal stem cell therapy in veterinary medicine. Animals. 2024;14(7):994.[DOI]
-
22. Chen J, Wang Z, Yi M, Yang Y, Tian M, Liu Y, et al. Regenerative properties of bone marrow mesenchymal stem cell derived exosomes in rotator cuff tears. J Transl Med. 2025;23:47.[DOI]
-
23. Li Y, Zhu Z, Li S, Xie X, Qin L, Zhang Q, et al. Exosomes: Compositions, biogenesis, and mechanisms in diabetic wound healing. J Nanobiotechnol. 2024;22:398.[DOI]
-
24. Song Y, You Y, Xu X, Lu J, Huang X, Zhang J, et al. Adipose-derived mesenchymal stem cell-derived exosomes biopotentiated extracellular matrix hydrogels accelerate diabetic wound healing and skin regeneration. Adv Sci. 2023;10(30):2304023.[DOI]
-
25. Ding Y, Luo Q, Que H, Wang N, Gong P, Gu J. Mesenchymal stem cell-derived exosomes: A promising therapeutic agent for the treatment of liver diseases. Int J Mol Sci. 2022;23(18):10972.[DOI]
-
27. Prieto González EA. Heterogeneity in adipose stem cells. In: Birbrair A, editor. Stem cells heterogeneity-Novel concepts. Cham: Springer; 2019. p. 119-150.[DOI]
-
28. Abu-El-Rub E, Khaswaneh RR, Almahasneh FA, Almazari R, Alzu’bi A. Adipose tissue and bone marrow-derived mesenchymal stem cells are not really the same: Investigating the differences in their immunomodulatory, migratory, and adhesive profile. Biochem Genet. 2025;63(1):378-392.
-
29. An Y, Lin S, Tan X, Zhu S, Nie F, Zhen Y, et al. Exosomes from adipose-derived stem cells and application to skin wound healing. Cell Prolif. 2021;54(3):e12993.[DOI]
-
31. Chen W, Lv L, Chen N, Cui E. Immunogenicity of mesenchymal stromal/stem cells. Scand J Immunol. 2023;97(6):e13267.[DOI]
-
32. Tang Y, Zhou Y, Li HJ. Advances in mesenchymal stem cell exosomes: A review. Stem Cell Res Ther. 2021;12:71.[DOI]
-
33. Yoshida S, Miyagawa S, Toyofuku T, Fukushima S, Kawamura T, Kawamura A, et al. Syngeneic mesenchymal stem cells reduce immune rejection after induced pluripotent stem cell-derived allogeneic cardiomyocyte transplantation. Sci Rep. 2020;10:4593.[DOI]
-
34. Li H, Ghazanfari R, Zacharaki D, Lim HC, Scheding S. Isolation and characterization of primary bone marrow mesenchymal stromal cells. Ann N Y Acad Sci. 2016;1370(1):109-118.[DOI]
-
36. Ong WK, Sugii S. Adipose-derived stem cells: Fatty potentials for therapy. Int J Biochem Cell Biol. 2013;45(6):1083-1086.[DOI]
-
38. Zhang Y, Khan D, Delling J, Tobiasch E. Mechanisms underlying the osteo- and adipo-differentiation of human mesenchymal stem cells. Sci World J. 2012;2012:793823.[DOI]
-
39. Simon B, Bolumar D, Amadoz A, Jimenez-Almazán J, Valbuena D, Vilella F, et al. Identification and characterization of extracellular vesicles and its DNA cargo secreted during murine embryo development. Genes. 2020;11(2):203.[DOI]
-
40. Doyle L, Wang M. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells. 2019;8(7):727.[DOI]
-
42. Solinger JA, Ott DP, Spang A. ESCRTing the RABs through conversion. Biochem Soc Trans. 2025;53(2):431-445.[DOI]
-
43. Liu C, Liu D, Wang S, Gan L, Yang X, Ma C. Identification of the SNARE complex that mediates the fusion of multivesicular bodies with the plasma membrane in exosome secretion. J Extracellular Vesicle. 2023;12(9):12356.[DOI]
-
44. Zhang Y, Liu Y, Liu H, Tang WH. Exosomes: Biogenesis, biologic function and clinical potential. Cell Biosci. 2019;9:19.[DOI]
-
45. Kita S, Shimomura I. Stimulation of exosome biogenesis by adiponectin, a circulating factor secreted from adipocytes. J Biochem. 2021;169(2):173-179.[DOI]
-
47. Mettlen M, Chen PH, Srinivasan S, Danuser G, Schmid SL. Regulation of clathrin-mediated endocytosis. Annu Rev Biochem. 2018;87:871-896.[DOI]
-
50. Delenclos M, Trendafilova T, Mahesh D, Baine AM, Moussaud S, Yan IK, et al. Investigation of endocytic pathways for the internalization of exosome-associated oligomeric alpha-synuclein. Front Neurosci. 2017;11:172.[DOI]
-
52. Mulcahy LA, Pink RC, Carter DRF. Routes and mechanisms of extracellular vesicle uptake. J Extracellular Vesicle. 2014;3:24641.[DOI]
-
53. Li H, Chen C, Wang Y, Yi W, Guo P, Yao C, et al. A meta-analysis on application and prospect of cell therapy in the treatment of diabetes mellitus. Stem Cell Res Ther. 2025;16:249.[DOI]
-
55. Shi R, Jin Y, Zhao S, Yuan H, Shi J, Zhao H. Hypoxic ADSC-derived exosomes enhance wound healing in diabetic mice via delivery of circ-Snhg11 and induction of M2-like macrophage polarization. Biomed Pharmacother. 2022;153:113463.[DOI]
-
58. Li Y, Zhang J, Shi J, Liu K, Wang X, Jia Y, et al. Exosomes derived from human adipose mesenchymal stem cells attenuate hypertrophic scar fibrosis by miR-192-5p/IL-17RA/Smad axis. Stem Cell Res Ther. 2021;12:221.[DOI]
-
61. Zhu J, Quan H. Adipose-derived stem cells-derived exosomes facilitate cutaneous wound healing by delivering XIST and restoring discoidin domain receptor 2. Cytokine. 2022;158:155981.[DOI]
-
62. Yeon E, Kang D, Kang T, Song H, Kim J. The adipose-derived stem cell secretome: Composition, fibroblast modulation, and clinical translation. Biomed Sci Lett. 2025;31(4):279-288.[DOI]
-
63. Ji H, Li H, Wang J, Wang K, Wen C, Xu L, et al. Multifunctional hydrogels-based dressings for diabetic chronic wounds: Mechanisms and clinical translation. Int J Biol Macromol. 2026;336:149373.[DOI]
-
64. Li X, Xie X, Lian W, Shi R, Han S, Zhang H, et al. Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med. 2018;50(4):58.[DOI]
-
65. Li X, Liu L, Yang J, Yu Y, Chai J, Wang L, et al. Exosome derived from human umbilical cord mesenchymal stem cell mediates miR-181c attenuating burn-induced excessive inflammation. EBioMedicine. 2016;8:72-82.[DOI]
-
66. Kang T, Jones TM, Naddell C, Bacanamwo M, Calvert JW, Thompson WE, et al. Adipose-derived stem cells induce angiogenesis via microvesicle transport of miRNA-31. Stem Cells Transl Med. 2016;5(4):440-450.[DOI]
-
67. Kohlhauser M, Mayrhofer M, Kamolz LP, Smolle C. An update on molecular mechanisms of scarring: A narrative review. Int J Mol Sci. 2024;25(21):11579.[DOI]
-
68. Wang L, Hu L, Zhou X, Xiong Z, Zhang C, Shehada HMA, et al. Exosomes secreted by human adipose mesenchymal stem cells promote scarless cutaneous repair by regulating extracellular matrix remodelling. Sci Rep. 2017;7:13321.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite