Abstract
Medical catheters constitute indispensable components of contemporary healthcare, yet they are confronted with persistent limitations in biocompatibility and functionality, such as thrombosis, infection, and tissue injury, which adversely affect patient safety and therapeutic outcomes. Surface coating technology has consequently arisen as a critical approach to reengineer the catheter-biological interface, thereby augmenting functional performance while preserving the intrinsic properties of the bulk materials. This review systematically outlines recent advances in coating technologies for medical catheters. It begins by analyzing mainstream coating methods, such as layer-by-layer self-assembly, surface grafting, and biomimetic adhesion, followed by the introduction of coatings with anticoagulant, antibacterial, lubricant, and multifunctional properties. The effectiveness and challenges of these coatings in clinical applications, such as cardiovascular intervention, long-term indwelling urinary catheters, and respiratory management are critically examined. Finally, the review discusses current translational bottlenecks and future trends toward intelligent, durable, and cost-effective coating solutions, providing a comprehensive reference for developing next-generation high-performance medical catheters.
Keywords
1. Introduction
Medical catheters, as indispensable tools in modern healthcare, have become core devices for clinical diagnosis, treatment, and life support[1,2]. Their applications span multiple critical fields, including cardiovascular intervention, urinary drainage, respiratory support, and nutritional delivery. The performance of catheters, particularly the characteristics of their interfaces with human tissues and blood, directly determines the treatment success rates, risk of complications, and patient long-term prognosis and safety[3,4]. However, despite advances in materials science, traditional catheter materials (such as silicone, polyurethane (PU), and polyvinyl chloride (PVC)) still face significant biological challenges during prolonged or short-term contact with the human environments[5]. These challenges primarily include thrombosis triggered by non-specific protein adsorption, catheter-related infections caused by persistent biofilms formed after bacterial adhesion, tissue and mucosal damage resulting from surface friction, and the subsequent inflammatory responses[6-8]. Medical catheter-related infections cause more than 300, 000 deaths worldwide every year, and the incidence of hospital catheter-related infections accounts for 20%-30% of nosocomial infections[9,10]. Among them, respiratory intubation has the highest infection rate, up to 11.5%, with a high risk of death, followed by intravenous catheters (6.9%) and urinary catheters (3.9%)[11-14]. These complications not only severely compromise the safety and efficacy of catheter clinical applications, prolong patient hospital stays, but also impose a substantial healthcare burden.
To address these challenges, optimizing the catheter body material alone is no longer enough to meet the complex clinical demands. Consequently, precise regulation and modification of the material-biological interfaces through surface engineering has emerged as a frontier and hotspot in biomedical engineering research[15-17]. Surface coating technology enables the construction of a functionalized thin layer on the catheter substrate, endowing it with superior interfacial biological properties without altering the mechanical characteristics of the catheter body. The core of this strategy lies in achieving precise control over the material’s surface chemical composition, topological structure, charge distribution, and energy state[18,19]. This targeted approach enhances the key performance indicators such as biocompatibility, anticoagulant properties, antimicrobial activity, and/or lubricity[20-22].
In recent years, the coating modification technology has made significant progress in layer-by-layer (LBL) self-assembly, surface grafting, and biomimetic adhesive hydrogel coating[23-25]. These methods enable the flexible integration of anticoagulants, antibacterial agents, lubricating polymers, and bioactive molecules, forming a series of coating systems with anticoagulant and antifouling properties, antibacterial and anti-biofilm capabilities, hydrophilic lubrication, and multifunctional composites[26,27]. These systems provide tailored solutions for diverse clinical scenarios through passive repulsion, active killing, or smart responses[28-30].
This paper aims to provide a systematic review of the latest research advances in coating modifications for medical catheters (Figure 1). First, it will thoroughly analyze the principles, characteristics, and application examples of mainstream coating technologies, including LBL self-assembly, surface grafting, and biomimetic adhesion. Second, coatings will be categorized based on their core functions, covering anticoagulant, antibacterial, lubricant, and multifunctional composite coatings. Subsequently, the effectiveness and challenges of these coating technologies in specific clinical applications will be explored, such as cardiovascular interventional devices, long-term indwelling urinary catheters, and respiratory anesthesia management. Finally, this paper will objectively analyze the technical bottlenecks encountered in the clinical translation of current coating technologies and project future development trends toward intelligent, long-lasting, and green, low-cost solutions. It aims to provide theoretical references and technical guidance for the research, development, and clinical translation of next-generation high-performance medical catheters.
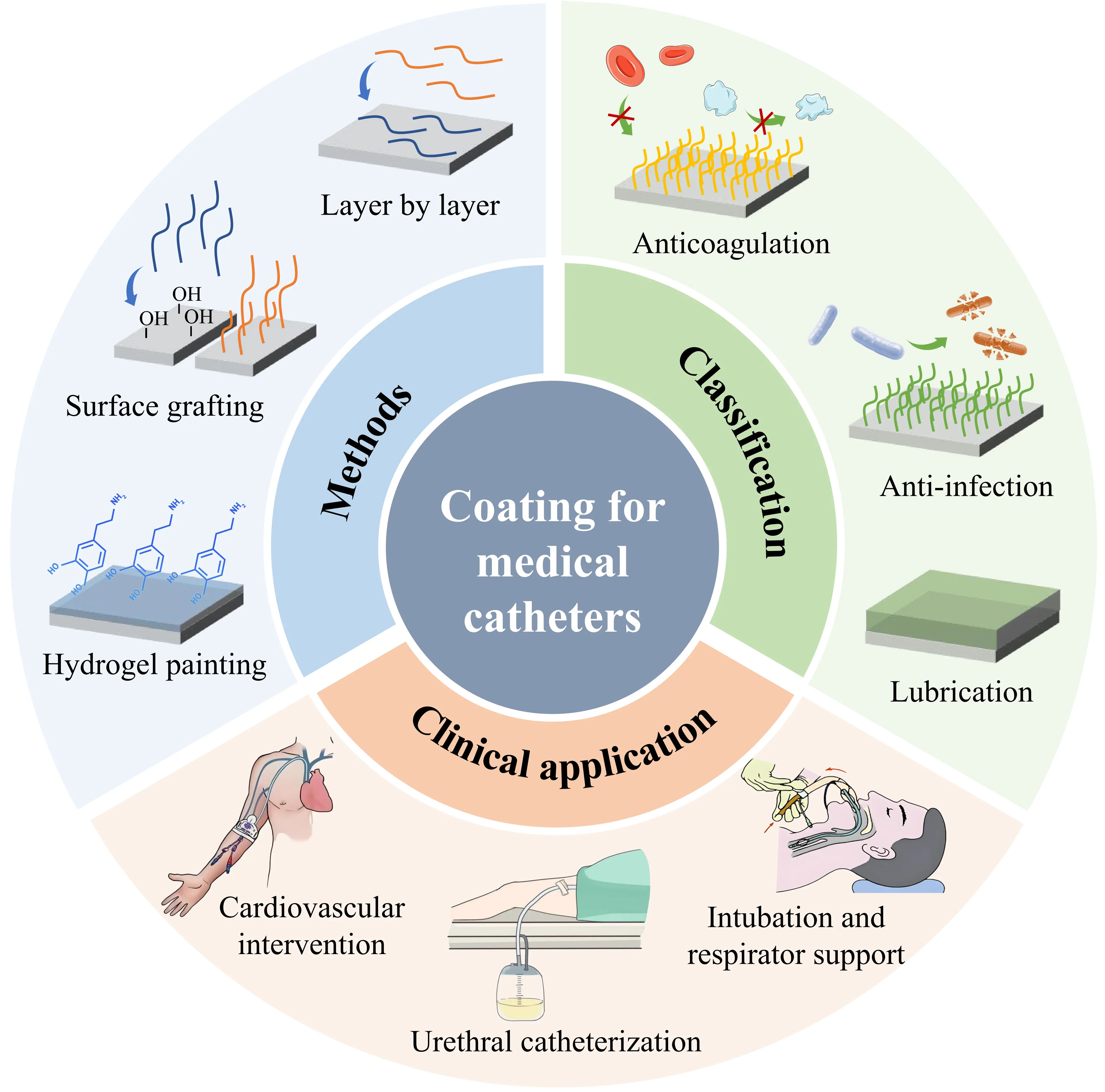
Figure 1. Technical methods, functional classification, and clinical application of medical catheter coatings.
2. Coating Modification Methods for Medical Catheters
With the cross-fertilization of materials science and surface modification technology, the coating building strategy has developed from simple physical adsorption to multi-scale and multifunctional design, covering chemical grafting, biomimetic modification, and other cutting-edge directions[31,32]. According to the principle of coating-substrate interaction and the principle of coating preparation, the current coating construction strategies are classified into LBL self-assembly technology, surface graft modification, surface-initiated polymerization, and biomimetic adhesion strategies.
2.1 LBL self-assembly surface construction strategy
The LBL self-assembly is a technology to construct multifunctional thin films with nanoscale accuracy at the liquid-solid interface[33]. The most classic LBL model involves the alternating adsorption of polycations (e.g., polyallylamine, chitosan, and polylysine) and polyanions (e.g., sodium polystyrene sulfonate, sodium alginate, and heparin)[34]. For example, Chen et al. employed positively charged polyethyleneimine (PEI) and negatively charged hyaluronic acid (HA) to self-assemble a bioinspired endothelial coating on vascular stents in a LBL manner, utilizing a metal-phenol network (epigallocatechin gallate and Cu) to enhance coating-stent adhesion[35]. Liu et al. employed LBL to combine dendritic polyglycerol amine, dendritic polyglycerol aldehyde, and linear polyglycerol sulfate (lPGS) onto a polystyrene surface[36]. Fixing dendritic polyglycerol enhanced surface coverage, enabling a higher density of heparin mimicking lPGS. Pires et al. employed alternating layers of poly(L-arginine) and poly(L-glutamic acid) on drug carriers to significantly prolong their blood half-life[37].
In some instances, LBL is reinforced by hydrogen bonding, host-guest interactions, and covalent bonding in addition to electrostatic attraction. Electrostatic forces can form a coating between PEI and HA, and the addition of epigallocatechin gallate and Cu can improve the coating’s stability through interactions such as π-π stacking, hydrogen bonding, and covalent crosslinking between phenols, aldehydes, and amines[35]. Polyethylenimine-β-cyclodextrin (PEI-β-CD) and ferrocene-modified chitosan (Fc-CHT) can be assembled through host-guest interactions to form PEI-β-CD/Fc-CHT multilayers. Polydopamine can be used to deposit the PEI-β-CD/Fc-CHT multilayers onto substrate surfaces to obtain antimicrobial coatings[38]. Adamantane (Ada) is one of the guest molecules for β-CD. Biotin-, mannose-, or cell adhesion peptides-modified β-CD can be coated onto polymer surfaces containing Ada through host-guest interactions (Figure 2a), enabling streptomycin modification, lectin modification, or enhanced cell adhesion on the coated surface[42]. It is well-known that replaceable deposition building blocks provide a straightforward method for preparing advanced multilayer films with tailored membrane structures and functions. In short, the LBL technology offers low equipment requirements, mild processing conditions, controllable film thickness, and adaptability to complex substrate geometries, establishing it as a powerful platform for constructing bioactive surfaces.
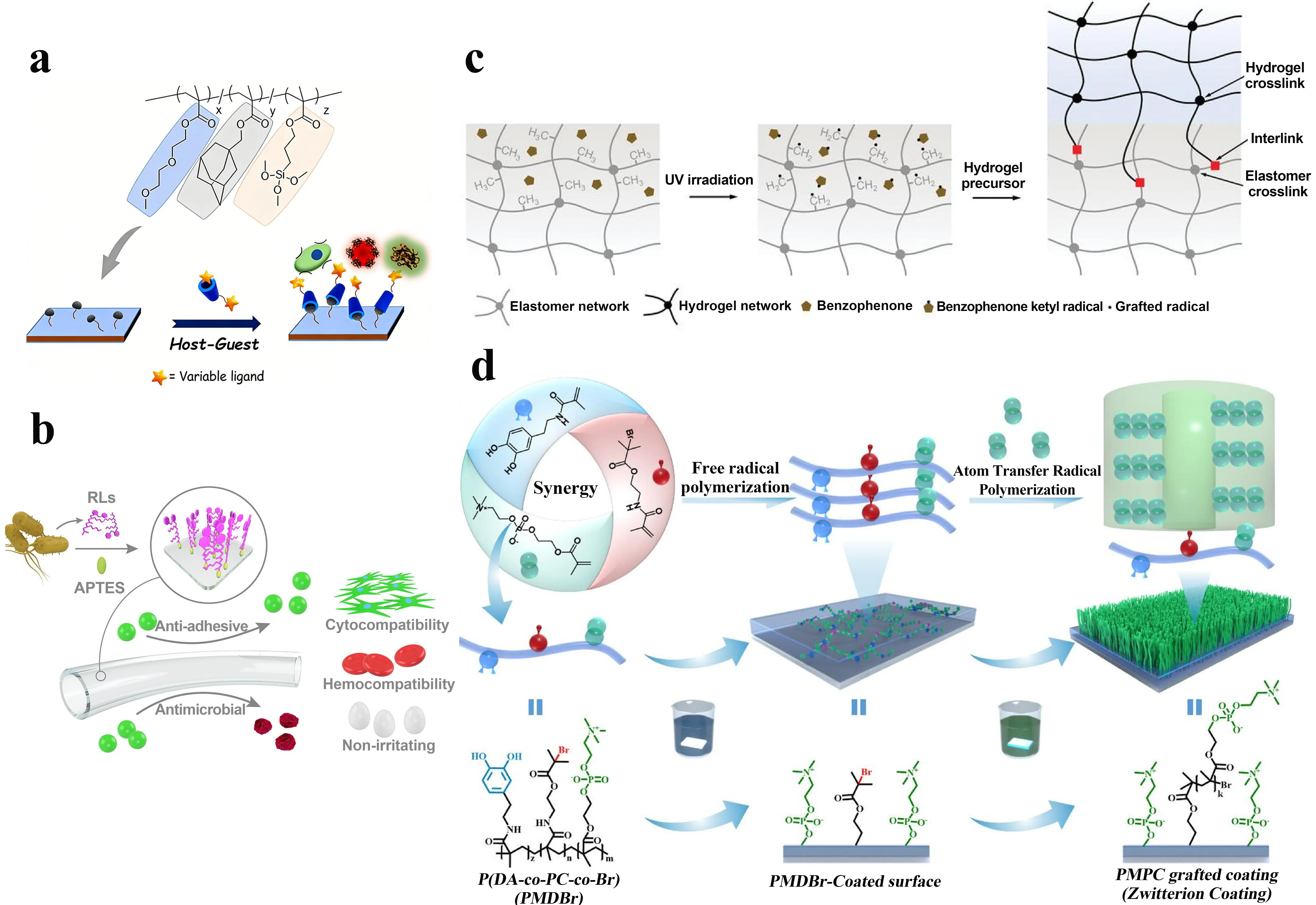
Figure 2. Coating modification methods for medical devices. (a) Schematic representation of the layer-by-layer self-assembly strategy based on host-guest interactions. Republished with permission from[38]; (b) Schematic representation of the surface grafting strategy based on silane coupling agents[39]; (c) Schematic diagram of surface grafting technology based on surface-initiated polymerization. Republished with permission from[40]; (d) Dopa-assisted coating technique. The polymer coating has phosphorylcholine, dopa, and bromoisobutyrate side groups, which enhance the polymer-substrate interaction and make the assembly process more rapid in aqueous environment[41].
2.2 Grafting to
The surface grafting technology firmly anchors functional polymer chains to material surfaces via covalent bonds, forming high-density, highly stable “polymer brush” interfaces. This requires introducing bridging molecules whose ends can form strong interactions with both the hydrogel and the substrate, establishing covalent bonds at the substrate-coating interface. The commonly used bridging molecules for coatings are silane coupling agents, such as γ-methacryloxypropyl trimethoxy silane (KH570) and 3-triethoxysilylpropylamine (KH550)[43]. The alkoxy group at one end of the bridging molecule is hydrolyzed in an aqueous environment to form a silanol group, which then condenses with a hydroxyl group on the target surface to form a siloxane bond. This process creates covalent bonding between the target substrate and the bridging molecules. KH550 contains an amino group with strong polarity and reactivity, enabling it to form chemical bonds with active groups in resins such as epoxy and phenolic. For KH570, the vinyl group at the other end can participate in the copolymerization of hydrogel coating precursors, thereby achieving covalent bonding between the hydrogel coating and KH570[44].
In principle, this method is effective when the substrate surface is rich in hydroxyl groups. Most inorganic materials used in medical devices, such as glass, metals, and ceramics, naturally possess hydroxyl groups on their clean surfaces, making them generally easier to be modified. For certain medical polymeric materials, such as elastomers and plastics, hydroxyl groups can be introduced through surface treatments such as oxygen plasma and ultraviolet (UV) ozone. Dardouri et al. subjected medical-grade polydimethylsiloxane (PDMS) surfaces to plasma treatment and utilized KH550 as a bridging molecule to covalently bind rhamnolipids to the silicone rubber (SR) surface (Figure 2b)[39]. The rhamnolipid coating rendered the PDMS surface more hydrophilic, reducing Gram-positive bacterial colonization and biofilm formation while exhibiting favorable hemocompatibility. Soyhan et al. activated silicone catheter surfaces via UV/ozone treatment, followed by alkenyl group functionalization. They then employed thiol-alkene click chemistry to bind free thiol groups in antimicrobial peptides (AMPs)[45]. The peptide-immobilized surfaces demonstrated enhanced resistance to bacterial adhesion while maintaining biocompatibility with mammalian cells.
2.3 Grafting from
The surface-initiated polymerization stands as the universal surface grafting technology. By precisely anchoring initiators onto the substrate surfaces, it enables the “growth” of polymer brushes with controllable molecular weight, structure, and functionality starting from the interfaces. This “bottom-up” precision synthesis strategy provides an unprecedented platform for constructing next-generation high-performance, intelligent medical coatings. Benzophenone (BP), a classic surface initiator, has seen widespread application in this technique. Under UV irradiation, BP abstracts hydrogen atoms from the substrate surface to generate free radicals suitable for chemical grafting. The modification process relies on swelling BP into the surface of the catheter substrate using a suitable solvent, where it serves as the initiator for the grafting reaction[46]. Moreover, water-based precursor solutions are often employed for the grafting. This approach reduces the solubility of BP in the liquid phase, thereby promoting its localization and retention within the substrate. This ensures sufficient photoinitiation efficiency during the UV irradiation stage and guarantees a robust bond between the coating and the substrate interface (Figure 2c)[40]. Yu et al. prepared a dual-network hydrogel composed of poly(2-acrylamido-2-methylpropane sulfonic acid) and chitosan (CS) on catheter surfaces made of various substrates (thermoplastic polyurethane (TPU), nylon, PVC, and SR)[47]. This coating endowed the catheters with an extremely low friction coefficient (as low as 0.03) and high anticoagulant properties. Similarly, You et al. successfully constructed an anionic hydrogel coating of poly(2-acrylamido-2-methylpropane sulfonic acid sodium salt) on catheter surfaces[48]. Leveraging the synergistic interactions between sulfonate groups and rifampicin molecules via electrostatic attraction and hydrogen bonding, this coating enables efficient loading of this hydrophobic antibiotic. In contrast to previous approaches, Cao et al. chose to swell BP into the PDMS elastomer surface alongside monomers, resulting in a hydrogel coating having an interpenetrating network with the substrate surface[49]. The highly entangled zwitterionic polymer chains form a high toughness, low coefficient of friction, and antithrombotic gel coating that dissolves in water upon exfoliation, providing an insurance mechanism that eliminates the risk of embolism.
Similar to the BP UV-induced grafting, electron beam and gamma radiation irradiation can also generate graftable active sites on substrate surfaces. Costoya et al. introduced glycidyl methacrylate onto PVC catheter surfaces using a 60Co gamma source, successfully immobilizing lysozyme and acyltransferase[50]. The functionalized catheter exhibited significant dual antimicrobial efficacy: lysozyme effectively hydrolyzed bacterial cell walls, reducing Staphylococcus aureus adhesion by 2-3 orders of magnitude; while acyltransferase effectively disrupted bacterial quorum sensing by degrading multiple acylated histidine lactones, including signaling molecules produced by Pseudomonas aeruginosa and Acinetobacter baumannii, thereby inhibiting biofilm formation. Valencia-Mora et al. grafted thermosensitive poly(N-vinylcaprolactam) onto SR surfaces via direct γ-irradiation[51]. The functionalized SR-g-NVCL material exhibited pronounced temperature-responsive properties, demonstrating reversible hydrophilic-hydrophobic transitions near physiological temperatures. Zuñiga-Zamorano et al. grafted pH-responsive polymethacrylate onto the surface of medical PVC catheters[52]. The functionalized PVC-g-PMAA catheter exhibited good pH-responsive swelling behavior (critical pH of 8.5), and efficiently loaded the cationic antimicrobial benzalkonium chloride and the antibiotic ciprofloxacin. Overall, the surface-initiated techniques based on radical-active polymerization enable precise control over surface polymer chain structures, serving as a powerful tool for preparing high-performance, long-lasting medical coatings.
2.4 Hydrogel paint based on biomimetic adhesion strategy
Inspired by the robust adhesion achieved by natural organisms such as mussels and barnacles in wet, dynamic environments, biomimetic adhesion strategies, particularly those centered on dopamine (DA) chemistry, have transformed the landscape of hydrogel coating applications in biomedicine. DA undergoes oxidative self-polymerization under weakly alkaline conditions, forming poly(dopamine) (PDA) films rich in catechol/quinone groups and amino groups. Catechol groups firmly anchor to nearly all substrate surfaces through diverse interactions including covalent bonds (-SH and -NH2), hydrogen bonds, coordination bonds, and π-π stacking. Leveraging this mechanism, Bai et al. synergistically co-deposited quaternized polyethyleneimine, polycarboxylic acid betaine copolymer with an amphoteric structure, and DA to construct a uniform, stable, and highly hydrophilic anti-adhesion coating on commercial silicone-based urinary catheters[53]. Zhou et al. coated PDA onto PDMS catheter surfaces, followed by deposition of atom transfer radical polymerization (ATRP) initiators for direct growth of antimicrobial hydrogels from the initiator sites to form covalently bonded, robust coatings[54]. Additionally, Zhang et al. copolymerized DOPA (wet-adhesive group), phosphatidylcholine (hydrophilic and anti-fouling group), and bromoisobutyric ester (initiator group) to synthesize a novel water-soluble polymer (PMDBr) bearing three functional side groups (Figure 2d)[55]. The synergistic action of these groups enables the polymers to rapidly and firmly self-assemble onto various catheter inner walls within 3 min in aqueous solution. The active groups incorporated into the molecules facilitate subsequent grafting, enabling the customization of biological functions to meet diverse clinical needs. The bionic coatings based on mussel adhesive proteins, with their universal adhesion, biocompatibility, and strong functional integrability, provide an innovative solution for constructing highly effective antibacterial and antifouling medical catheters.
In summary, these approaches have evolved from simple physical adsorption to multi-mechanism integrated design, driven by the convergence of surface chemistry, materials science, and medical requirements. Each of these four approaches has distinct pros and cons with respect to stability, difficulty of preparation, cost, and durability, and clinical applicability (Table 1). Specifically, LBL self-assembly is characterized by low cost, straightforward operation, and strong adaptability to complex substrates, but its stability and durability are limited by non-covalent interactions. Both the Grafting to and Grafting from strategies rely on covalent bonds to achieve high interface stability and durability. The Grafting to is limited by substrate surface reactive groups such as hydroxyls and higher preparation complexity, while the Grafting from has better controllability and longer service life. The Hydrogel paint based on the biomimetic adhesion strategy of DA chemistry strikes a balance between practicality and performance with a straightforward immersion process, universal substrate compatibility, and moderate stability. The trade-offs among the four strategies provide a clear reference for the selection of coating methods in different situations for healthcare scenarios.
| Strategy | Stability | Complexity | Cost | Durability | Clinical applicability |
| Layer-by-layer self-assembly | Low; mainly relies on non-covalent interactions, easy to peel off under dynamic friction. | Medium; requires alternate deposition of multiple layers. | Low; uses common polyelectrolytes, simple equipment requirements. | Low; the non-covalent bond is easy to dissociate in long-term physiological environment. | Suitable for short-term indwelling devices. |
| Grafting to | High; relies on covalent bonds between bridge molecules and substrate/coating, strong interface binding. | High; requires surface activation, introduction of bridge molecules, and covalent reaction steps. | High; bridge molecules and surface activation equipment increases costs. | High; covalent bond is stable, and the polymer brush is not easy to fall off. | Suitable for catheters with hydroxyl-rich surfaces (glass, metal, modified polymers), long-term indwelling devices. |
| Grafting from | High; polymer chains grow directly from the substrate surface, forming covalent bonds. | High; requires precise fixation of initiators, strict control of polymerization conditions. | Medium; special polymerization equipment (UV lamp or heater) is usually required. | High; the polymer brush has high density and stable structure, suitable for long-term indwelling | mainly used for high-performance demand scenarios (e.g. cardiovascular interventional catheters). |
| Hydrogel paint | Medium; polydopamine forms multiple interactions with the substrate. | Low; simple immersion process, no surface activation required. | Medium; dopamine and its derivatives are moderately priced. | Medium; the hydrogel network is stable, and the biomimetic adhesion layer is not easy to wear. | Suitable for various short-term and long-term indwelling catheters. |
UV: ultraviolet.
3. Functional Classification of Coatings
3.1 Hydrophilicity enhancement and lubrication
Reducing friction resistance during catheter insertion and removal significantly minimizes the mechanical damage to delicate tissues such as the urethra and blood vessels, alleviates patient discomfort, and lowers the risk of secondary infections and strictures caused by injury. When the hydrophilic polymer networks absorb large amounts of water, they form a smooth aqueous film on the surface, providing ideal lubrication[55,56]. The common lubricating polymers include polyvinylpyrrolidone (PVP), polyacrylamide (PAM), polyethylene glycol (PEG), and polymers containing zwitterionic groups along with their derivatives.
The molecular structure of PVP contains a highly polar lactam group, endowing it with strong hydrophilic properties. Upon contact with water, PVP rapidly absorbs moisture, forming a hydration layer through hydrogen-bonded water molecules, which reduces friction between the catheter surface and human tissue during insertion, achieving a lubricating effect. Liu et al. utilized hydrogen bonding and electrostatic interactions to prepare a uniform and robust PVP composite coating on catheter surfaces[57]. The hydroxyl groups were created via plasma activation of the surface. Medicinal PU served as the primer, while PVP and anhydrous citric acid (CA) were used as the topcoat. The coating (PVP/CA-PU) was prepared by dip-coating followed by drying. The PVP molecular chains tightly intertwine with the PU chains, while the CA bonds to PVP via electrostatic interactions. This coating exhibits stability, with peel strength exceeding 600 N/m. Post-modification, the catheter’s hydrophobic surface transforms into a hydrophilic one, reducing the contact angle by 60° and lowering the friction coefficient below 0.1, a fourfold decrease from the base material, demonstrating low friction and superior hydrophilic lubrication. As a typical water-soluble polymer, PAM contains abundant amide groups (-CO-NH2) in its molecular chains. These polar groups form hydrogen bonds with water molecules, conferring hydrophilic properties. Ren et al. utilized DA’s adhesive properties to construct a HA hydrogel coating on polymer substrates, whose mechanical strength was enhanced by incorporating a PAM polymer network (Figure 3)[58]. The resulting DA-hyaluronic acid-polyacrylamide hydrogel coating reduced the substrate’s COF by 93% while maintaining strong shear strength (71.88 kPa). The repeating ether linkages (-O-) and terminal hydroxyl groups (-OH) in PEG’s molecular structure confer dual hydrophilic mechanisms: ether bonds enhance water adsorption via dipole interactions, while hydroxyl groups participate in dynamic hydrogen bonding formation. Chawla et al. synthesized a polyethylene glycol-L-3,4-dihydroxyphenylalanine-lysine copolymer and modified the PDMS surface via heat-dip coating, significantly improving its lubrication properties[59]. The modified PDMS surface exhibited a reduction in contact angle from 109° to below 15° and maintained an extremely low coefficient of friction (COF = 0.03) throughout 200 friction cycles, which is substantially smaller than that of the unmodified PDMS (COF = 0.98).
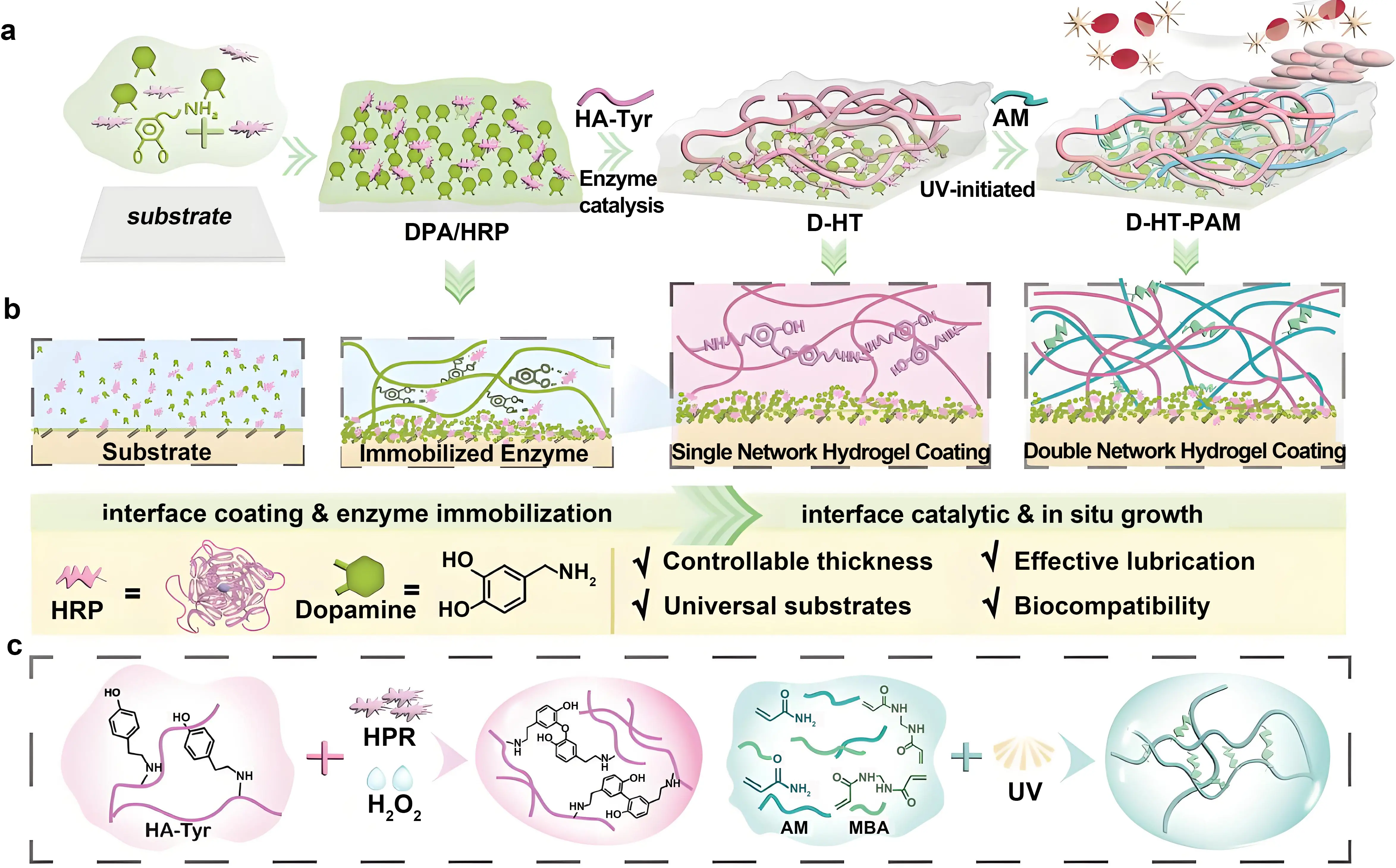
Figure 3. Dual network D-HT-PAM hydrogel coating. (a, b) Schematic diagram of D-HT-PAM; (c) Growth mechanism of the hydrogel coating. Republished with permission from[58]. D-HT-PAM: dopamine-hyaluronic acid-polyacrylamide.
During the preparation of lubricating coatings, the aforementioned hydrophilic polymers are often combined in two or more forms to enhance the lubrication efficacy or network strength. For example, Lai et al. fully leveraged the hydrophilicity of PVP and PEG alongside PDA’s exceptional adhesion to synthesize a PDA-PVP/PEG composite coating[60]. The hydroxyl groups in PEG form hydrogen bonds with the carbonyl groups of amide units in PVP chains, causing intertwining and entanglement between PEG and PVP molecular chains, further enhancing the stability of the coating. The use of both hydrophilic polymers of PEG and PVP, in the polymerization process resulted in a coating that effectively modified the hydrophilic lubrication properties of the TPU material. The water contact angle on the material surface decreased from 91.3° to 28.3°, and the surface friction coefficient in a water environment dropped from 0.4 to approximately 0.03. In summary, all the exemplified hydrophilic polymers provide high lubricity and are suitable for preparing lubricating coatings on medical catheter surfaces to prevent tissue friction damage.
However, it has to be mentioned when hydrophilic polymers are used as lubricant coatings on intravascular devices, their detachment may lead to serious adverse events[61]. Mehta et al. reported fatal complications from iatrogenic hydrophilic polymer embolism (HPE), raising awareness of the incidence and mortality associated with coated catheters[62]. Nevertheless, interventional coatings have been in clinical use for years, and the hazards associated with medical coatings in HPE have remained underappreciated for an extended period. The primary reason is the difficulty in detecting polymer coating embolization in clinical settings. In 2015, the FDA issued a document entitled “Lubricious Coating Separation from Intravascular Medical Devices: FDA Safety Communication”, acknowledging these concerns. It alerts stakeholders to the potential hazards and risks of polymer lubricant coatings on vascular interventional devices, and recommends safe clinical practices[63]. In 2019, the FDA further issued the “Coronary, Peripheral, and Neurovascular Guidewires Performance Tests and Recommended Labeling”, explicitly requiring comprehensive assessments of coating integrity, particulate matter, and lubrication functionality[64]. Specifically, coating integrity testing must be based on data collected from device models simulating clinical conditions such as device bending. Concurrent particle testing during coating integrity assessments must evaluate the source, quantity, and size distribution of particles potentially shed during simulated clinical use. If coating defects are detected, scientific and reasonable justification must be provided to clarify whether such defects pose safety risks. Currently, the evaluation and risk management system for lubricant coatings on vascular interventional products has been progressively refined. Industry-recognized standard assessment methods and acceptable particulate evaluation metrics have been established, forming a closed-loop chain of evidence[65]. Undoubtedly, the development and application of lubricant coatings have significantly advanced the clinical adoption and technological progress of vascular interventional devices. However, effectively mitigating the adverse events associated with these coatings requires collaborative attention and concerted efforts from manufacturers, regulatory authorities, and clinical users.
3.2 Anticoagulated blood and antiprotein adsorption
Anticoagulation and anti-protein adhesion are primary requirements for blood-contacting catheters such as central venous catheters (CVCs) and dialysis catheters. Rapid protein adsorption serves as the “trigger” for thrombosis, which may further activate platelets and the coagulation cascade, leading to catheter occlusion, thromboembolism, and even the death of patients[66]. Anticoagulant and anti-protein adhesion coatings are crucial for preventing catheter-related thrombosis. Common anticoagulant coatings employ active anticoagulant agents (e.g., heparin and hirudin) or passive anticoagulant barriers via fouling-resistant polymers (e.g., polyethylene glycol (PEG), and zwitterionic polymers). Liu et al. developed a multifunctional HS/DAC composite coating using a combination of sodium heparin and quaternary ammonium surfactants (Figure 4)[67]. This coating adheres to medical devices of arbitrary shapes and compositions while simultaneously treating catheter-related bloodstream infections and thrombosis. Zhang et al. created hydrophobic-modified heparin based on electrostatic assembly between the cationic antimicrobial compound ethyl lauroyl sarcosinate and anionic HS[68]. Driven by electrostatic and hydrophobic interactions, it formed a uniform and robust coating on a series of multi-lumen catheters of various shapes, effectively reducing blood cell adhesion and fibrinogen attachment. Although heparin is an ideal anticoagulant bioactive substance, limitations in fixing it to catheter surfaces include covalent bonds restricting heparin bioactivity and protein adsorption, leading to reduced anticoagulant activity, limited drug loading capacity, and increased bleeding risk. Hydrophilic lubricant coatings composed of PEG represent another promising approach to prevent protein and cell adhesion. Wang et al. developed a multifunctional hydrophilic and anti-fouling coating by grafting DA molecules onto the ends of the four-arm polyethylene glycol carboxyl[69]. This coating exhibits high hydrophilicity, anti-protein adsorption properties, and biocompatibility. However, PEG chains are susceptible to oxidative degradation and loss, leading to gradual reductions in chain length and surface density of the PEG layer, which significantly compromises the anti-adhesion properties[70].
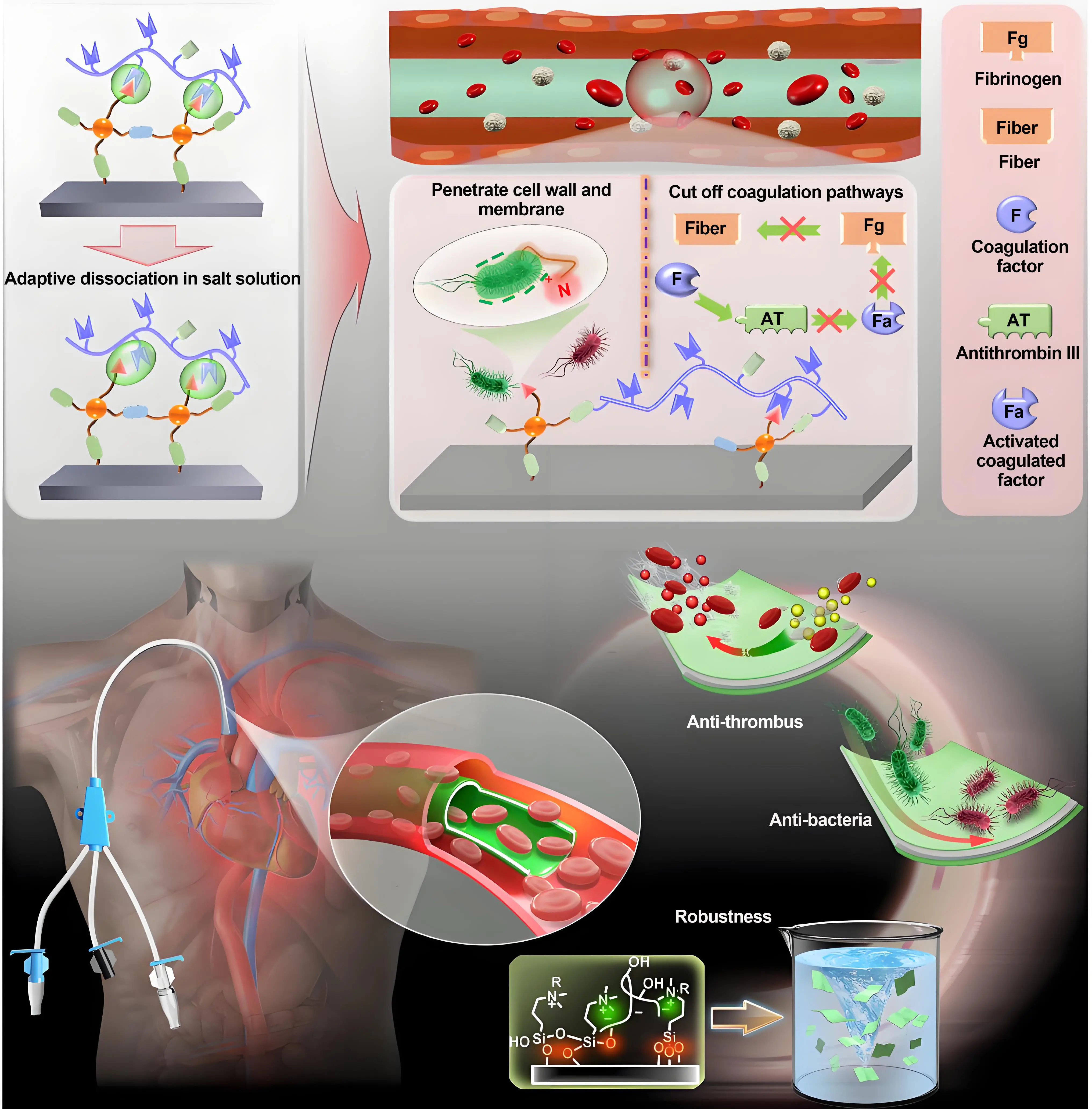
Figure 4. Schematic representation of antithrombotic and anti-infective coatings. The antibacterial and antithrombotic mechanisms and properties of HS/DAC coatings are illustrated[67]. HS: heparin sodium; DAC: dimethyloctadecyl[3-(trimethoxysilyl)propyl]ammonium.
Bionic cell structures represent another promising strategy. Poly(methacryloxyethyl phosphorylcholine) coatings mimic the phosphatidylcholine structure of the outer cell membrane, binding water molecules through electrostatic interactions to form a dense hydration layer. This creates a physical and energetic barrier on the catheter surface, effectively preventing protein and platelet approach and denaturing adsorption[71]. Li et al. developed an endothelial membrane-mimicking coating with the major components of heparin-grafted phosphocholine copolymers (PMPCC/Hep) on the catheter surface after precoating polydopamine[72]. Compared to conventional heparin coatings, the PMPCC/Hep coating could further reduce protein adsorption by 50%. More notably, the coating exhibited superior anticoagulant activity, even significantly surpassing end-point connection heparin coatings. Wang et al. employed a copolymer composed of DMA and MPC to create anticoagulant and lubricant surfaces[73]. This coating enables surface functionalization of various biomedical implants via a soluble dip-coating method, effectively enhancing the anticoagulant and lubricant properties of vascular implant devices.
3.3 Antibacterial and anti-biofilm
Catheter-associated infections rank among the most common hospital-acquired infections, stemming from biofilms formed after bacterial adhesion. Biofilms exhibit exceptional resistance to antibiotics and the host immune system, and are extremely difficult to be eradicated once established. Anti-fouling coatings, which inhibit bacterial adhesion through physical repulsion mechanisms, are a common strategy for preventing catheter biofilm formation. Peng et al. employed reversible addition-fragmentation chain transfer polymerization technology to precisely synthesize an amphoteric sulfosuccinate betaine/active ester block copolymer[74]. By covalently bonding the active ester side chains to amino groups on the surface of activated SR, they successfully constructed a stable anti-fouling coating. In biological performance evaluations, the coated catheters demonstrated significant anti-adhesion effects in the short term, with inhibition rates of 96.5% against S. aureus and 97.3% against E. coli. However, since this approach inhibits bacterial adhesion rather than killing bacteria, a certain risk of infection may still exist.
The antibiotics, metal ions, or other biocides loaded into coatings kill bacteria by releasing antimicrobial agents, thereby reducing bacterial growth and adhesion on biomaterial surfaces, which is a commonly employed antibacterial strategy. Zhang et al. developed a ternary system composed of oxide xanthan gum, gelatin, and gentamicin sulfate (GS), constructing a coating on silicone rubber catheters via Schiff base crosslinking (Figure 5)[75]. Here, GS was covalently loaded via pH-responsive imine bonds. When bacterial metabolism created an acidic microenvironment, the responsive bonds broke, triggering the antibiotic GS release in an on-demand manner. Three consecutive cycles of repeated antimicrobial testing simulating recurrent infections demonstrated over 99% antimicrobial efficacy through this responsive antibiotic release. Metal ions (e.g., silver ions and copper ions) exert bactericidal effects by disrupting microbial metabolic processes and compromising cell membrane integrity[76]. Won et al. employed a dual-layer nanoengineering approach to fabricate a novel catheter utilizing zinc and silver nanoparticles (AgNPs)[77]. The catheter surface was coated with the cationic polymer PDA, followed by sequential deposition of AgNPs onto the PDA layer. Subsequently, Zn was deposited onto the AgNPs layer using radiofrequency sputtering. The inner AgNP layer provides immediate antimicrobial action against pathogens, while the outer porous Zn layer controls zero-order Ag release and generates reactive oxygen species, enhancing long-term bactericidal performance. However, it must be mentioned that metal ion-based antimicrobial agents such as silver and zinc can easily trigger allergic reactions in body. In severe cases, they may lead to postoperative failure of coronary stents, hip and knee prostheses, and other implanted devices[78,79]. Currently, regulatory oversight of metal ion antimicrobial agents in medical coatings is intensifying. Most silver-coated catheter products have been withdrawn from the market, underscoring the need for researchers to shift toward developing and applying safer, more effective antimicrobial materials.
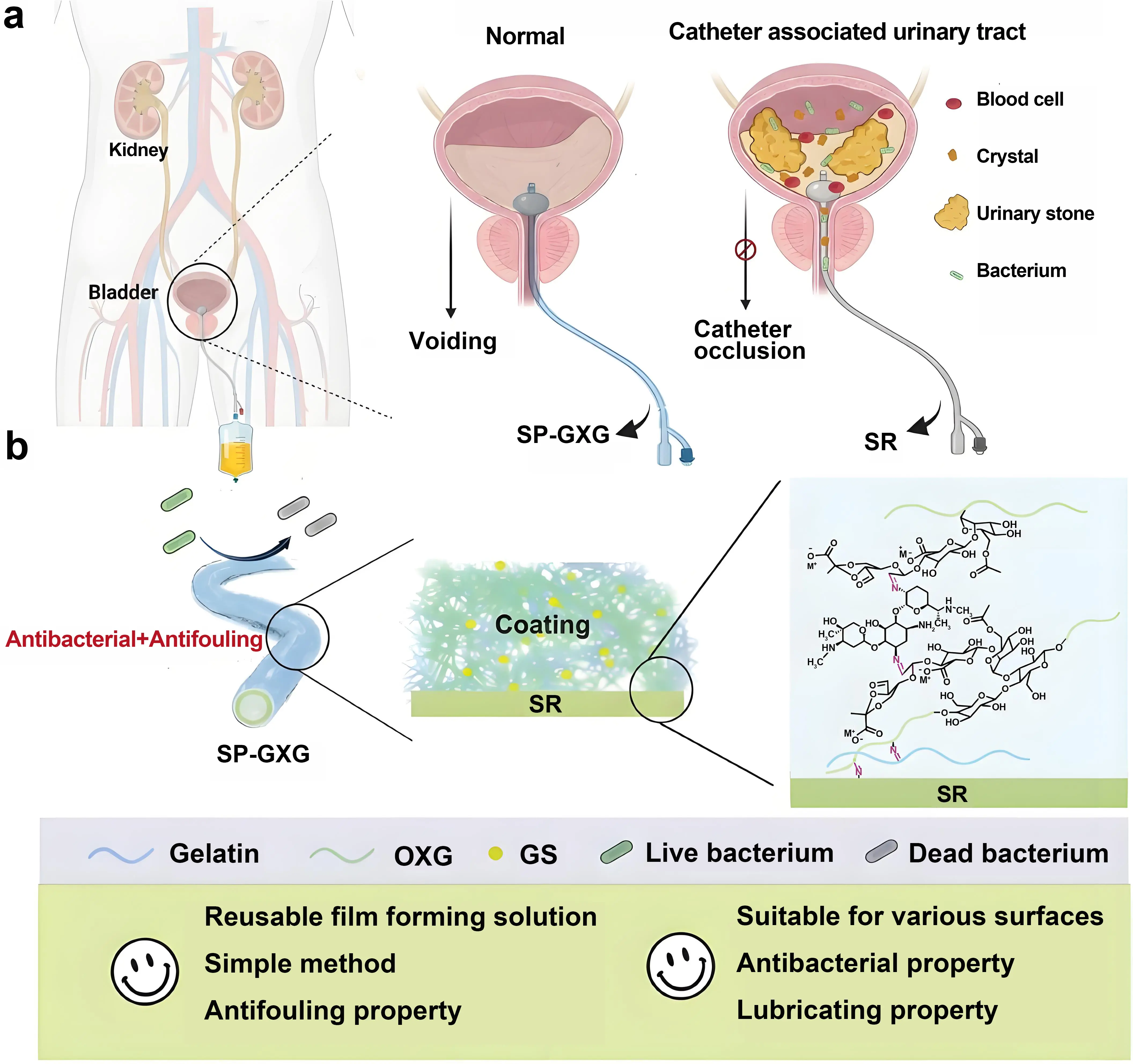
Figure 5. Schematic illustration of the structure and properties of SR-GXG. A ternary system composed of oxide xanthan gum, gelatin, and gentamicin sulfate, constructing a coating on silicone rubber catheters via Schiff base crosslinking. Republished with permission from[75]. SR: silicone rubber.
Covalently anchoring antimicrobial agents to catheter surfaces or coatings can prevent antimicrobial failure caused by rapid release. Commonly used agents include AMPs, quaternary ammonium compounds (QACs), CS derivatives, and other cationic polymers (e.g., polylysine and hyperbranched polylysine). The coating directly contacts bacteria, where the antimicrobial agent disrupts the negatively charged bacterial cell membrane via electrostatic interactions, leading to leakage of cellular contents and cell death. Lin et al. employed a mussel-inspired approach to graft AMP (CWR11 peptide) onto the reactive phenolic groups of DA, achieving AMP fixation on the catheter surface[80]. The coating demonstrated broad-spectrum antibacterial activity against S. aureus and E. coli at approximately 99%, while also inhibiting biofilm formation. Long et al. developed a quaternized coating material (AQTA) by co-substituting tannic acid with alkyl and quaternary ammonium groups[81]. A stable AQTA coating with adjustable content was achieved on polyurethane catheter surfaces via a simple one-step immersion process. The presence of QACs endowed the coating with antibacterial activity exceeding 99% against S. aureus and E. coli in vitro, while reducing inflammation at the implant site in vivo. The contact antimicrobial coatings offer the advantage of sustained antibacterial efficacy without antimicrobial depletion, demonstrating significant potential in the development of anti-infective catheter products.
3.4 Multifunctional composites
Clinical needs are often complex and concurrent, making the single-function coatings insufficient for high-end medical devices. Integrating two or more functions into a single coating is the current research focus and future development direction. For example, a single function-coated catheter may have limited utility in addressing friction damage and infection issues encountered with medical catheters during clinical use. Lubricating coatings can reduce tissue damage and provide antifouling properties, but once bacteria break through the initial barrier and form a biofilm, the antifouling mechanism of the coating may fail, leading to infection. While the single antimicrobial coatings can lower infection risks, a potential drawback is the non-specific adsorption of bacterial debris, proteins, and lipids after the killing process, leading to gradual reduction in antimicrobial activity or functional impairment, along with the risk of recurrent infections. To this end, Shao et al. designed a biocompatible hydrogel coating with strong lubrication and antibacterial properties using hydrophilic PVP, hydroxyethyl methacrylate, and HBPL with an interpenetrating double-network structure (Figure 6)[82]. This coating modifies the surface of polyurethane CVCs, simultaneously reducing friction and wear while inhibiting infection. Pan et al. successfully constructed a dynamically responsive PVEQ hydrogel coating on silicone rubber catheter surfaces using a DA-mediated surface deposition strategy[83]. This coating employs stepwise deposition of β-cyclodextrin-grafted polyvinyl alcohol and 4-arm polyethylene glycol functionalized with adamantane/QACs, achieving molecular-level precise crosslinking through the host-guest recognition between β-CD and adamantane. The QACs intelligently switch between zwitterionic and cationic bactericidal states in response to pH changes in bacterial infection microenvironments (physiological pH = 7.4 to infected microacidic pH = 5.5), conferring on-demand antimicrobial functionality to the coating. The highly hydrophilic surface reduces the friction coefficient by approximately 10-fold compared to the catheter substrate, significantly enhancing the catheter’s tissue interface compatibility. Hu et al. developed a dual-network hydrogel coating (PPM) based on a chemically crosslinked PVP-PEGDA network and a physically crosslinked network inspired by the adhesion mechanism of recombinant mussel protein[84]. Benefiting from the biomimetic adhesion properties and antimicrobial activity conferred by MFP’s 20% lysine and 30% DOPA content, combined with PVP’s hydrophilicity and lubricating effect, the PPM-coated catheters exhibited high antimicrobial and lubricating performance. Li et al. achieved in situ preparation of antimicrobial and lubricating hydrogel coatings on silicone tubing by introducing polyelectrolytes[85]. The first plasma-treatment of the silicone tubes generated surface hydroxyl groups. Subsequently, the tubes were sequentially dipped at room temperature into a PAE binder polyelectrolyte solution containing acrylic acid (AAc) and PEI, followed by an AAP hydrophilic lubricant solution prepared with AAc, acrylamide, and PVP to form an antibacterial lubricating hydrogel coating on the catheter surface. Hu et al. designed a novel bilayer hydrogel coating that mimics the vascular endothelium by integrating BSA/carboxybetaine acrylamide (CBAA) with BSA/CBAA/DA[86]. Unlike conventional monofunctional coatings, this bilayer design exhibited highly integrated structure-function properties, passive antifouling, active inhibition of thrombin generation, and potent interfacial adhesion. More importantly, the hydrogel demonstrates self-adaptive immunomodulatory capabilities that dynamically respond to local inflammatory stimuli.
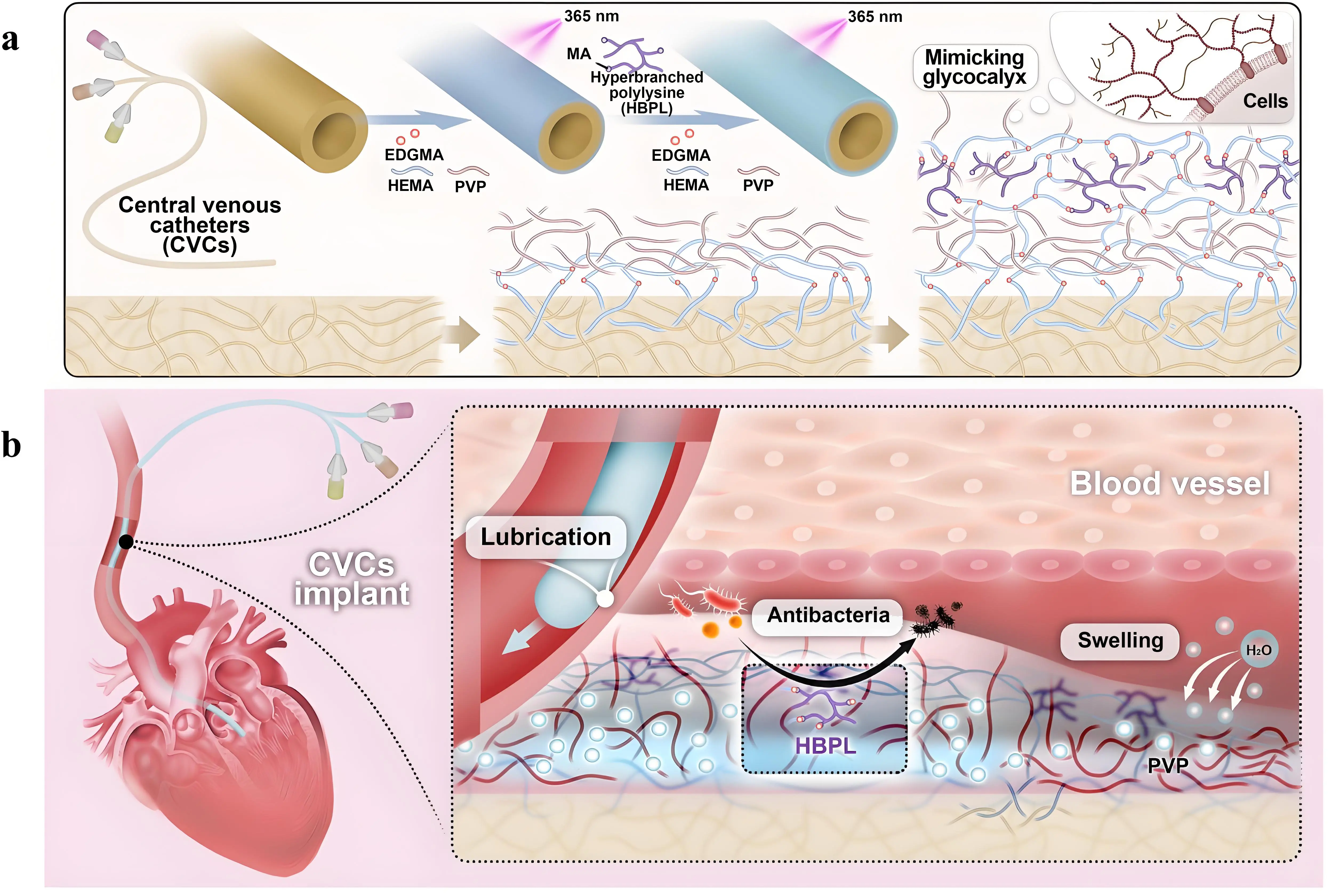
Figure 6. Schematic representation of a biocompatible hydrogel coating with dual lubricating and antibacterial properties. Republished with permission from[82]. (a) Basic composition and process of hydrogel coating. The coatings can be prepared by immersion of CVC catheters in PVP/HEMA and PVP/HEMA/HBPL solutions, respectively, followed by curing under UV radiation; (b) The hydrogel coating can reduce frictional tissue damage and reduce the risk of infection. CVC: central venous catheter; PVP: polyvinylpyrrolidone; HEMA: hydroxyethyl methacrylate; HBPL: hyperbranched poly-L-lysine; UV: ultraviolet.
4. Clinical Application
4.1 Cardiovascular interventional devices
Percutaneous coronary intervention (PCI) is a widely adopted strategy for treating coronary artery disease. However, PCI carries risks of complications, including in-stent restenosis (ISR) and stent thrombosis. Drug-coated balloon (DCB) and drug-eluting stent (DES) implantation are common clinical approaches to prevent ISR. Clinical studies indicate that paclitaxel-coated balloons demonstrate comparable efficacy to DCBs and DES in preventing ISR complications when treating peripheral arterial disease[87]. DCBs exhibit superior late lumen loss in angiographic patterns compared to DES, with comparable clinical outcomes[88]. The paclitaxel DCB demonstrated superior primary patency rates compared with plain balloon angioplasty for treating stenotic lesions in dysfunctional hemodialysis arteriovenous fistulae at 6 and 12 months[89]. So far, DCB angioplasty has become a safe and effective treatment option for arterial lesions[90]. Although the use of polymer coatings to release drugs can prevent restenosis, the long-term presence of these coatings within the body may induce chronic vascular wall inflammation and hypersensitivity reactions[91], leading to delayed endothelialization and significantly increasing the risk of thrombosis one year post-implantation. This situation promotes the clinical shift toward “biodegradable-coated” stents. For example, Boston Scientific’s SYNERGYTM stent coating comprises a PLGA (poly(lactic-co-glycolic acid)) polymer and everolimus. The coating fully degrades into carbon dioxide and water within months post-implantation, coinciding with complete drug release, thus avoiding chronic inflammation caused by prolonged polymer presence. Similarly, Biotronik’s Orsiro Mission DES features a degradable coating made of polylactide. Its ultra-thin design minimizes physical damage to the vessel wall, and the asymmetric coating technology enables exceptionally rapid endothelialization.
4.2 Anti-infection with long-term indwelling catheter
The use of indwelling catheters in intensive care units (ICUs) plays a significant role in determining the incidence and prevalence of hospital-acquired urinary tract infections (UTIs), as well as mortality rates. While implementing evidence-based protocols can substantially reduce catheterization rates and the incidence of hospital-acquired UTIs, catheter-associated urinary tract infections (CAUTIs) remain prevalent among long-term bedridden patients with prolonged catheter use. Therefore, antimicrobial catheters are crucial for preventing urinary tract infections and reducing CAUTI incidence. A clinical trial involving 13,392 patients compared the CAUTI prevention efficacy of two antimicrobial catheters available in the U.S. market (nitrofurazone-coated and silver alloy-coated catheters)[92]. Compared to standard latex or silicone catheters, both the nitrofurazone-coated and silver alloy-coated catheters appeared to reduce the development of asymptomatic bacteriuria during use (< 30 d). Similarly, a prospective study analyzed differences in CAUTI incidence among bladder cancer patients undergoing orthotopic radical cystectomy who received either coated or conventional uncoated catheters[93]. Results indicated that the coated catheters significantly suppressed bacterial colonization on the catheter surface, inhibited biofilm formation, and reduced infections. However, a practical UK multicenter randomized controlled trial comparing three catheter types used in the UK NHS, antimicrobial-impregnated (nitrofurazone) and antimicrobial-coated (silver alloy) catheters versus standard polytetrafluoroethylene-coated catheters, found that the nitrofurazone-coated catheters were more likely to cause discomfort during insertion and removal[94]. The trial excluded the possibility that the silver alloy-coated catheters could achieve the prespecified level of clinical efficacy, and their use was unlikely to be cost-effective. Another study evaluating the safety of a novel silver-impregnated Foley catheter showed that the silver-impregnated Foley catheters appeared effective in preventing catheter-associated bacterial urinary tract infections, particularly those caused by Gram-negative rods[95]. Although multiple studies have evaluated the efficacy and safety of existing antimicrobial catheter products, uncertainties remain regarding these products, including microbial resistance, cost, and duration of use. Research indicates that the coated catheters demonstrate a significantly lower risk of CAUTI compared to the uncoated groups only when used long-term (> 14 d)[96]. Concurrently, issues such as urethral trauma, bladder overdistension, prolonged catheterization, and improper urine bag handling warrant attention, because they may be closely associated with CAUTI development[97].
A large-scale, multicenter randomized controlled trial compared silver-coated catheters, nitrofuran-coated catheters, and standard catheters. No statistically significant difference between silver-coated catheters and standard catheters was found in reducing symptomatic urinary tract infections, while the nitrofuran group exhibited a higher incidence of catheter-related discomfort compared to other groups[98]. Another clinical study reached similar conclusions: silver alloy-coated catheters were not statistically significantly associated with a marked reduction in symptomatic CAUTI, and antibiotic-impregnated catheters were more likely to cause pain than standard catheters[99]. These findings may have influenced national healthcare guidelines, leading to the discontinuation of routine recommendation for expensive silver-coated or antibiotic-impregnated catheters[100]. Furthermore, silver may be an underestimated allergen. A case report described a patient who developed eczema and vesicular lesions with pruritus around a left leg ulcer after using a silver-containing cream, ultimately diagnosed with allergic contact dermatitis[101]. The symptoms were resolved within two weeks after discontinuing silver-containing treatment and administering corticosteroids and antihistamines. Currently, silver-containing coatings face stringent regulation due to potential biosafety risks. These concerns suggest that safer, more effective, and cost-efficient novel antimicrobial catheter products may be urgently needed for patients requiring long-term catheterization.
4.3 Respiratory and anesthetic management
Postoperative sore throat (POST) is a distressing complaint following endotracheal intubation in adults. Topical application of a eutectic mixture of local anesthetics cream on the endotracheal tube (ETT) cuff reduces the incidence of postoperative POST, cough, and hoarseness (HOV) in adults[102]. A clinical study demonstrated that compound lidocaine cream, used as a tracheal tube lubricant, effectively reduces POST, postoperative cough, HOV, and foreign body sensation in the throat[103]. However, ventilator-associated pneumonia (VAP), the second most prevalent hospital-acquired infection in emergency settings, is a potentially fatal consequence that current clinical approaches often ignore. Subglottic suctioning (Taperguard) ETTs represent a clinical approach to reduce VAP risk. In a clinical trial involving 90 patients, no significant difference in VAP incidence or ICU mortality was observed between patients using silver-coated (Bactiguard) and subglottic suctioning (Taperguard) ETTs, suggesting that the coated tubes offer benefits in lowering VAP[104]. Another clinical study demonstrated that the coated Bactiguard infection-protective ETTs, compared to standard uncoated ETTs, reduced the incidence of ventilator days, VAP, and ICU length of stay in patients requiring mechanical ventilation for ≥ 48 h and experiencing drug-induced coma[105]. Clinical studies have also demonstrated that precious metal-coated ETTs effectively reduce VAP incidence without significantly affecting mortality or ICU length of stay[106]. Longer-lasting antimicrobial protection may be a key factor in shortening mechanical ventilation duration or ICU stay. Novel ETTs that balance these benefits while addressing complications such as POST are highly anticipated.
5. Challenges and Outlooks
5.1 Technical difficulties
Most coatings rely on physical adsorption or weak interactions for fixation, making them prone to peeling and wear during long-term implantation or in dynamic physiological environments, leading to functional failure. This issue is particularly pronounced in scenarios requiring repeated friction or prolonged retention, such as vascular interventions and urinary catheters. Covalent grafting methods can overcome this challenge and may offer more practical significance in terms of safety. However, medical catheters typically utilize inert materials such as silicone rubber. Achieving long-term stable surface modification on these highly hydrophobic substrates remains a persistent challenge in medical device research and development. While studies indicate that swelling impregnation can create interpenetrating coating-substrate functional surfaces, applying this technique to small-scale, precision medical catheters appears structurally disruptive. Therefore, achieving long-lasting surface functionalization on inert materials remains a significant challenge in the development of coating technology.
Another critical hurdle in the translation of coating technologies lies in the complexity of laboratory-scale precision modification techniques (e.g., ATRP). First, ATRP is oxygen-sensitive and requires extended polymerization times. Many industrial settings cannot accommodate oxygen-free environments, and the extended polymerization time restricts its application in high-throughput production lines[107]. Second, numerous catalysts are metal salts that cannot be recovered after dissolution or dispersion, posing environmental pollution risks[108]. Furthermore, many initiators readily deactivate in solution, leading to significant monomer waste and increased product costs. Moreover, the high synthesis costs of high-performance coating materials (e.g., customized AMPs and zwitterionic polymers) significantly hinder clinical adoption. Even after market approval, patients may struggle to afford the medical costs associated with these novel catheters. Consequently, cost-effectiveness is a core factor determining market acceptance of novel coated catheters. Additionally, single monomers struggle to meet the multifunctional demands of clinical applications, while the adoption of new monomers faces stringent regulatory oversight. This extends product development and commercialization timelines, further increasing production costs. The industry has long anticipated polymerization technologies that are greener, simpler, and more readily industrialized.
5.2 Development trend of coating modification technology
Intelligent and multifunctional integrated coatings may be urgently needed in clinical settings. Developing stimulus-responsive coatings that respond to pH, temperature, bacterial metabolites, etc., could enable “on-demand” release of antimicrobial agents and “smart activation” of anticoagulant functions. For instance, antibiotics could be released upon triggering by acidic conditions in the infection microenvironment, or anticoagulant activity could be enhanced during hemodynamic changes. Such intelligent coatings, balancing long-lasting efficacy with biosafety, may be highly anticipated by clinicians. Additionally, achieving multifunctional synergy through molecular design and structural regulation, such as blending AMPs with zwitterionic polymers, can simultaneously deliver antibacterial effects, prevent protein adsorption, and enhance biocompatibility. Constructing a “fouling prevention-sterilization-repair” multi-level protection system can inhibit bacterial adhesion at the source, efficiently eliminate residual bacteria, and minimize tissue damage. These aspects may all require simultaneous consideration in novel coating technologies and products.
Regarding application translation, green, low-cost coating technologies are highly desirable to industry. The core demands from industry for coating technologies center on “simplifying processes, reducing costs, and ensuring batch-to-batch consistency”. Only technologies meeting these criteria can truly achieve translation from laboratory to clinical products. Streamlining operational procedures is the primary pathway to cost reduction. The traditional surface grafting techniques demand strict control over reaction temperature, pH, gas conditions, and other parameters. In contrast, the dip-coating techniques and surface-initiation methods significantly simplify the process. For example, the dip-coating method based on polyurethane polymers enables one-step thermal curing to prepare ureteral stent coatings, which retain antibacterial and anti-stone functionality even after one month of implantation in vivo[109]. This coating process, requiring no specialized precision equipment or an oxygen-free environment, reduces capital investment. Moreover, the dip-coating technique enables uniform coating coverage of both inner and outer surfaces without custom fixtures or complex positioning, making it compatible with catheters featuring intricate geometries (e.g., multi-lumen catheters, curved vascular catheters). Surface-initiated strategies can cure catheter coatings within minutes, forming a robust hydrogel skin on the catheter’s outer surface via an interpenetrating network[110]. This simple, high-throughput method enables automated rapid production. Moreover, the dip-and-hang curing approach significantly reduces raw material consumption and waste generation. Raw material accessibility is also crucial for cost control. Overcoming challenges in raw material synthesis or utilizing natural biological materials (such as CS and sodium alginate, etc.) as substitutes for synthetic polymers may be key to cost management. Overall, the advancement of medical catheter coating modification technologies remains centered on clinical needs and industrial transformation. The integrated development of intelligent response systems for precise regulation, multifunctional synergistic approaches for comprehensive protection, and green, low-cost solutions for widespread adoption will provide robust support for enhancing catheter performance and ensuring clinical safety.
6. Conclusion
In conclusion, the surface coating technology represents a transformative approach to overcoming the inherent biocompatible and functional limitations of traditional medical catheters. By engineering the interface between the catheter and the biological environment, the coatings can effectively address critical clinical challenges such as thrombosis, infection, tissue friction, and inflammatory responses. This review has systematically elucidated the principal coating strategies, including LBL self-assembly, surface grafting, and biomimetic adhesion, which enable precise control over surface properties. Furthermore, the functional classification of coatings into lubricant, anticoagulant, antibacterial, and multifunctional composites highlights the tailored solutions available for diverse clinical scenarios. Clinical applications in cardiovascular devices, long-term indwelling catheters, and respiratory management demonstrate the potential of the coated catheters to improve patient outcomes, although variability in efficacy and cost-effectiveness underscores the need for further optimization. Despite significant progress, the clinical translation of advanced coatings faces persistent challenges, particularly regarding long-term stability under physiological conditions, the complexity and cost of manufacturing, and the need for standardized performance evaluation. Future development should focus on intelligent, stimulus-responsive systems that offer on-demand functionality, multifunctional synergistic designs for comprehensive protection, and green, low-cost fabrication processes to enhance industrial scalability and clinical accessibility. Specifically, processes such as one-step preparation of high-performance coatings, rapid low-energy curing techniques, and solvent-free processes that reduce production costs and risks hold greater promise for commercialization. Ultimately, the continued integration of material science, surface engineering, and clinical insights will drive the evolution of catheter coatings, paving the way for safer, more effective, and patient-friendly medical devices.
Authors contribution
Chen H: Investigation, writing-original draft, writing-review & editing, visualization, funding acquisition.
Nie X, Hu Q: Investigation, writing-review & editing.
Ma X, Li J: Data curation, writing-review & editing.
Gao C: Supervision, project administration, methodology, funding acquisition, conceptualization, writing-review & editing.
Conflicts of interest
Changyou Gao is an Editorial Board Member of BME Horizon. The other authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This study was financially supported by the China Postdoctoral Science Foundation (Grant No. 2025M782956), the Zhejiang Association for Science and Technology Young Talent Support Program (Grant No. ZJSKXQT2026098), the Joint Fund of National Natural Science Foundation of China (Grant No. U22A20155), National Natural Science Foundation of China (Grant No. 52573142), the National Key Research and Development Program of China (Grant No. 2023YFE0108700), the State Key Laboratory of Transvascular Implantation Devices (Grant No. 012024004), the 111 Project (Grant No. B16042), and the Overseas Academician Joint Lab for Supramolecular Polymeric Biomaterials, Zhejiang University.
Copyright
© The Author(s) 2026.
References
-
8. Cunegundes PS, Wood K, de Souza JG, Bhangu A, Mao L, Klueh U. Device insertion versus material: Drivers of inflammation in diabetes device interfaces. J Diabetes Sci Technol. 2025.[DOI]
-
14. Viola GM, Darouiche RO. Cardiovascular implantable device infections. Curr Infect Dis Rep. 2011;13(4):333-342.[DOI]
-
15. Xu C, Ivanovski S. Clinical translation of personalized bioengineered implant scaffolds. Nat Rev Bioeng. 2025;3(5):390-407.[DOI]
-
16. Xiong H, Cao Y, Bao Z, Chen S. Strategies for enhancing mechanical performances of long-term zwitterionic biomedical implants. Colloids Surf B Biointerfaces. 2026;258:115232.[DOI]
-
17. Kim HJ, Koo JH, Lee S, Hyeon T, Kim DH. Materials design and integration strategies for soft bioelectronics in digital healthcare. Nat Rev Mater. 2025;10(9):654-673.[DOI]
-
18. Skok K, Bele U, Pintar Š, Peršin Z, Kuzmič K, Bračič M, et al. Urinary catheters: State of the art and future perspectives–a narrative review. Mater Today Bio. 2025;34:102225.[DOI]
-
21. Gregorits BM, Wu Y, Chen C, Yeager E, Parker MC, Martinez I, et al. In Vitro and in Vivo biocompatibility study of fluorinated polyphosphazene coatings for blood-contacting medical devices. Acta Biomater. 2025;203:346-357.[DOI]
-
26. Hu J, Yu Q, Wang L, Shi H, Luan S. Recent progress in antibacterial surfaces for implant catheters. BME Front. 2025;6:63.[DOI]
-
28. Song X, Man J, Qiu Y, Wang J, Liu J, Li R, et al. High-density zwitterionic polymer brushes exhibit robust lubrication properties and high antithrombotic efficacy in blood-contacting medical devices. Acta Biomater. 2024;178:111-123.[DOI]
-
29. Yuwen L, Liu Y, Zhang Q, Xu X, Lu L, Qiusong Y, et al. Magnetic field-targeting and ultrasound-responsive antibiotic delivery for enhanced penetration and eradication of bacterial biofilms. J Control Release. 2025;386:114152.[DOI]
-
30. Tarman AY, Ahmed S, Althumayri MO, Guy M, Lugo DC, Ligler FS, et al. Smart catheters for diagnosis, monitoring, and therapy. Adv Healthc Mater. 2025;e03913.[DOI]
-
35. Chen C, Zhang C, Yu T, Qin Y, Chen Y, Xiong Y, et al. Metal-phenolic networks assisted construction of multi-layered endothelium-mimetic polyelectrolyte coating with nitric oxide generation and glycocalyx functionalization. Biomaterials. 2026;324:123486.[DOI]
-
36. Liu K, Nickl P, Feng J, Haag R. A robust heparin-mimicking polyglycerol-based coating for blood-contacting devices with long-term hemocompatibility and preliminary anti-inflammatory properties. Adv Healthc Mater. 2026;15(2):e02766.[DOI]
-
39. Dardouri M, Bettencourt A, Martin V, Carvalho FA, Santos C, Monge N, et al. Using plasma-mediated covalent functionalization of rhamnolipids on polydimethylsiloxane towards the antimicrobial improvement of catheter surfaces. Biomater Adv. 2022;134:112563.[DOI]
-
40. Yang J, Bai R, Chen B, Suo Z. Hydrogel adhesion: A supramolecular synergy of chemistry, topology, and mechanics. Adv Funct Mater. 2020;30(2):1901693.[DOI]
-
43. Jiang X, Zhou C, Su J, Tang S. Enhanced anti-icing performance of novel superhydrophobic F-L@KH-SiO2/OTMS coating synergistic preparation with bionic micro-nano structures and modified nanoparticles. Chem Eng J. 2024;498:155264.[DOI]
-
44. Zhan H, Cheng W, Liu F, Yang C, Wang Y, Yan K, et al. Resilient and robust mechanoresponsive polydimethylsiloxane/SiO2 composites induced by interfacial enhancement. J Colloid Interface Sci. 2025;690:137361.[DOI]
-
46. Zhang M, Hakobyan K, Cheng C, Xu J. Surface engineering of polymer hydrogels toward functional soft material innovations. Macromol Chem Phys. 2025;226(15):e00511.[DOI]
-
47. Yu Y, Wang J, Wang XW, Huang Y, Li RY, Wang YX, et al. A tough, slippery, and anticoagulant double-network hydrogel coating. ACS Appl Polym Mater. 2022;4(8):5941-5951.[DOI]
-
49. Cao W, Zhou X, Dai W, Zhu Z, Liu Z, Chen K, et al. An embolism-free nonfouling hydrogel coating with high toughness and lubricity for intravascular medical devices via chain-entanglement mediated topological gelation. Bioact Mater. 2026;59:174-185.[DOI]
-
50. Costoya A, Velázquez Becerra LE, Meléndez-Ortiz HI, Díaz-Gómez L, Mayer C, Otero A, et al. Immobilization of antimicrobial and anti-quorum sensing enzymes onto GMA-grafted poly(vinyl chloride) catheters. Int J Pharm. 2019;558:72-81.[DOI]
-
51. Valencia-Mora RA, Zavala-Lagunes E, Bucio E. Grafting of thermo-sensitive N-vinylcaprolactam onto silicone rubber through the direct radiation method. Radiat Phys Chem. 2016;124:155-158.[DOI]
-
52. Zuñiga-Zamorano I, Meléndez-Ortiz HI, Costoya A, Alvarez-Lorenzo C, Concheiro A, Bucio E. Poly(vinyl chloride) catheters modified with pH-responsive poly(methacrylic acid) with affinity for antimicrobial agents. Radiat Phys Chem. 2018;142:107-114.[DOI]
-
53. Bai Y, Li K, Ma L, Wu D, Xiang J, Hu Q, et al. Mussel-inspired surface modification of urinary catheters with both zwitterionic and bactericidal properties for effectively preventing catheter-associated infection. Chem Eng J. 2023;455:140766.[DOI]
-
55. Ma L, Gaisinskaya-Kipnis A, Kampf N, Klein J. Origins of hydration lubrication. Nat Commun. 2015;6:6060.[DOI]
-
56. Liu G, Feng Y, Zhao N, Chen Z, Shi J, Zhou F. Polymer-based lubricating materials for functional hydration lubrication. Chem Eng J. 2022;429:132324.[DOI]
-
57. Liu S, Qian L, Wu Y, Zhou L, Xu X, Yang J, et al. Enhanced long-term performance of ureteral stents with functional coating: Combating bacterial adhesion and inhibiting encrustation. Prog Org Coat. 2024;192:108512.[DOI]
-
58. Ren X, Zhang J, Yang F, Xu H, Guo G, Wang Y. Enzyme-immobilized surface-catalyzed cross-linking: Creating multifunctional double network hydrogel coatings on diverse substrates. Adv Funct Mater. 2024;34(27):2312465.[DOI]
-
60. Lai C, Wang S, Zhong P, Chen Y, Xu J, Li X, et al. A low-friction and high-stability hydrophilic PVP/PEG coated TPU for interventional catheter applications. Tribol Int. 2024;198:109859.[DOI]
-
63. Lubricious coating separation from intravascular medical devices: FDA safety communication [Internet]. Silver Spring: U.S. Food and Drug Administration; 2015. Available from: https://www.moph.gov.lb/userfiles/files.pdf
-
64. Coronary, peripheral, and neurovascular guidewires–performance tests and recommended labeling [Internet]. Silver Spring: U.S. Food and Drug Administration; 2019. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/coronary-peripheral-and-neurovascular-guidewires-performance-tests-and-recommended-labeling
-
65. Xu Y, Ming ZY. Risk assessment and regulatory exploration of the lubrication coating on intravascular medical devices. Front Cardiovasc Med. 2022;9:883406.[DOI]
-
68. Zhang X, Wang M, Liu L, Yu H, Liu Y, Duan X, et al. Hydrophobic-modified heparin coatings with adaptively anti-thrombotic and anti-bacterial functions for interventional cardiovascular catheters. Chem Eng J. 2025;514:163278.[DOI]
-
70. Sun M, Deng J, Tang Z, Wu J, Li D, Chen H, et al. A correlation study of protein adsorption and cell behaviors on substrates with different densities of PEG chains. Colloids Surf B Biointerfaces. 2014;122:134-142.[DOI]
-
71. Ke Y, Meng H, Du Z, Zhang W, Ma Q, Huang Y, et al. Bioinspired super-hydrophilic zwitterionic polymer armor combats thrombosis and infection of vascular catheters. Bioact Mater. 2024;37:493-504.[DOI]
-
72. Li R, Li Y, Bai Y, Yi P, Sun C, Shi S, et al. Achieving superior anticoagulation of endothelial membrane mimetic coating by heparin grafting at zwitterionic biocompatible interfaces. Int J Biol Macromol. 2024;257:128574.[DOI]
-
74. Peng W, Liu P, Zhang X, Peng J, Gu Y, Dong X, et al. Multi-functional zwitterionic coating for silicone-based biomedical devices. Chem Eng J. 2020;398:125663.[DOI]
-
80. Lim K, Chua RRY, Ho B, Tambyah PA, Hadinoto K, Leong SSJ. Development of a catheter functionalized by a polydopamine peptide coating with antimicrobial and antibiofilm properties. Acta Biomater. 2015;15:127-138.[DOI]
-
81. Long L, Fan Y, Yang X, Ding X, Hu Y, Zhang G, et al. A hydrophobic cationic polyphenol coating for versatile antibacterial and hemostatic devices. Chem Eng J. 2022;444:135426.[DOI]
-
82. Shao B, Wang X, Huang L, Liu X, Wang L, Zheng W, et al. A dual lubricating and antibacterial hydrogel coating containing hyperbranched polylysine with excellent biocompatibility for surface modification of central venous catheters. Chem Eng J. 2025;509:161402.[DOI]
-
83. Ran P, Qiu B, Zheng H, Xie S, Zhang G, Cao W, et al. On-demand bactericidal and self-adaptive antifouling hydrogels for self-healing and lubricant coatings of catheters. Acta Biomater. 2024;186:215-228.[DOI]
-
84. Hu Y, Qiao Y, Lei P, Gu Y, Sun L, Qiu Y, et al. Dual network hydrogel coatings based on recombinant mussel protein with enhanced antibacterial and super-lubrication properties for urinary catheter applications. Chem Eng J. 2023;474:145502.[DOI]
-
85. Li S, Yu P, Dong F, Zhang J, Wang T, Zhang P, et al. A lubricated and antibacterial hydrogel coating based on polyelectrolyte adhesion for medical catheters. J Mater Chem A. 2025;13(9):6687-6696.[DOI]
-
86. Hu X, Mou X, Pan G, Liu Y, Song W, Xiao P, et al. Vascular intima-biomimetic bilayer hydrogel coating of central venous catheters with dual-modal anti-coagulation and self-adaptive immunomodulation. Adv Funct Mater. 2026;36(7):e11029.[DOI]
-
90. Gao XF, Ge Z, Kong XQ, Chen X, Han L, Qian XS, et al. Intravascular ultrasound vs angiography-guided drug-coated balloon angioplasty the ULTIMATE Ⅲ trial. JACC Cardiovasc Interv. 2024;17(13):1519-1528.[DOI]
-
93. Tae BS, Oh JJ, Jeong BC, Ku JH. Catheter-associated urinary tract infections in patients who have undergone radical cystectomy for bladder cancer: A prospective randomized clinical study of two silicone catheters (clinical benefit of antibiotic silicone material). Investig Clin Urol. 2022;63(3):334-340.
-
94. Pickard R, Lam T, MacLennan G, Starr K, Kilonzo M, McPherson G, et al. Types of urethral catheter for reducing symptomatic urinary tract infections in hospitalised adults requiring short-term catheterisation: Multicentre randomised controlled trial and economic evaluation of antimicrobial- and antiseptic-impregnated urethral catheters (the CATHETER trial). Health Technol Assess. 2012;16(47):1-197.
-
96. Gauhar V, Castellani D, Teoh JY, Nedbal C, Chiacchio G, Gabrielson AT, et al. Catheter-associated urinary infections and consequences of using coated versus non-coated urethral catheters-outcomes of a systematic review and meta-analysis of randomized trials. J Clin Med. 2022;11(15):4463.
-
98. Pickard R, Lam T, MacLennan G, Starr K, Kilonzo M, McPherson G, et al. Antimicrobial catheters for reduction of symptomatic urinary tract infection in adults requiring short-term catheterisation in hospital: A multicentre randomised controlled trial. Lancet. 2012;380(9857):1927-1935.
-
104. Mahmoodpoor A, Sanaie S, Parthvi R, Shadvar K, Hamishekar H, Iranpour A, et al. A clinical trial of silver-coated and tapered cuff plus supraglottic suctioning endotracheal tubes in preventing ventilator-associated pneumonia. J Crit Care. 2020;56:171-176.[DOI]
-
107. Buhl KB, Agergaard AH, Lillethorup M, Nikolajsen JP, Pedersen SU, Daasbjerg K. Polymer brush coating and adhesion technology at scale. Polymers. 2020;12(7):1475.[DOI]
-
109. Li K, Tang H, Peng J, Gao S, Du Z, Chen G, et al. Smart lubricant coating with urease-responsive antibacterial functions for ureteral stents to inhibit infectious encrustation. Adv Funct Mater. 2024;34(2):2307760.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite




