Abstract
Microwave (MW) medicine has emerged as a distinct interdisciplinary field, predicated on the unique capacity of non-ionizing electromagnetic radiation to penetrate deep-seated tissues and interact efficiently with biological dielectrics for diverse therapeutic and diagnostic applications. Despite its clinical establishment in tumor ablation and hemostasis, conventional MW interventions are largely constrained by non-selective macroscopic heating, leaving the intricate potential of non-thermal biophysical modulation underutilized. The integration of engineered biomaterials provides a transformative framework to bridge this gap, enabling the precise modulation of
Keywords
1. Introduction
Microwaves (MW), a form of non-ionizing electromagnetic radiation in the frequency range of 300 MHz to 300 GHz, exhibit physicochemical properties that enable centimeter-scale tissue penetration and efficient coupling with biological dielectrics. These characteristics have established their critical role in a wide range of biomedical applications such as tumor thermal ablation, physical therapy, and non-invasive imaging[1,2]. In contrast to optical modalities (e.g., photothermal and photodynamic therapies), which are fundamentally confined to superficial depths by strong tissue scattering and absorption[2,3], or ultrasonic therapies often restricted by acoustic impedance mismatches at bone or air interfaces[4,5], MWs maintain superior propagation efficiency and spatial uniformity in deep-seated tissues[1,2,6,7]. Consequently, MW technology has been widely adopted in clinical practice, currently dominated by microwave ablation (MWA) for solid tumors in the liver, lung, and kidney[8-11], as well as rapid hemostasis and deep-tissue
The biological effects of MWs arise from several distinct yet interconnected mechanisms, among which the thermal effect remains the most established[13,14]. Importantly, the thermal effect represents the macroscopic outcome of electromagnetic energy dissipation via dielectric and magnetic loss processes[15]. Dielectric loss primarily originates from dipolar rotation and ionic conduction: under an alternating high-frequency electric field, polar molecules, predominantly water, attempt to realign with the oscillating field, but the rapid field reversal induces a phase lag (dielectric relaxation), converting electromagnetic energy into kinetic energy and ultimately into heat through molecular friction[16]. Magnetic loss, by contrast, involves the conversion of the magnetic component of the MW field into thermal energy via hysteresis loss and eddy current loss, particularly in the presence of magnetic-responsive materials[17]. Together, these mechanisms enable rapid and volumetric heating that is substantially more efficient than the conductive heating characteristic of radiofrequency ablation[18,19]. Beyond the dominant thermal effects, specific biomaterials possessing tailored structures and compositions can be excited by MW irradiation to generate reactive oxygen species (ROS), exhibiting a phenomenon analogous to the photodynamic effect known as the microwave-dynamic effect. Therapeutic strategies predicated on this physicochemical mechanism are defined as microwave-dynamic therapy (MDT)[20,21]. Analogous to photodynamic therapy, MDT utilizes MW energy to activate deep-tissue sensitizers, typically semiconducting heterojunctions or piezoelectric materials, thereby inducing potent non-thermal cytotoxicity[22,23]. Under MW exposure, the oscillating electric field promotes the separation of
To fully harness the diagnostic and therapeutic potential of microwave medicine, a refined system of energy-tissue interaction is essential, particularly for achieving enhanced precision and greater clinical versatility. For instance, in thermal therapies, a critical objective is to circumvent the physiological heat-sink effect caused by blood perfusion[10,36,37], thereby ensuring sufficient heating of large tumor masses while preventing collateral damage to healthy tissues. Similarly, advancing MDT requires overcoming the intrinsic barriers of the hypoxic tumor microenvironment and the rapid recombination of charge carriers inherent to
In this review, we provide a systematic overview of how engineered biomaterials modulate microwave-tissue interactions to expand the horizons of microwave medicine. We specifically focus on the physicochemical mechanisms, ranging from dielectric heating and magnetic loss to catalytic ROS generation and ionic regulation, that enable biomaterials to amplify or redirect microwave energy for targeted interventions. The discussion is categorized into major therapeutic domains, including antitumor, antibacterial, and
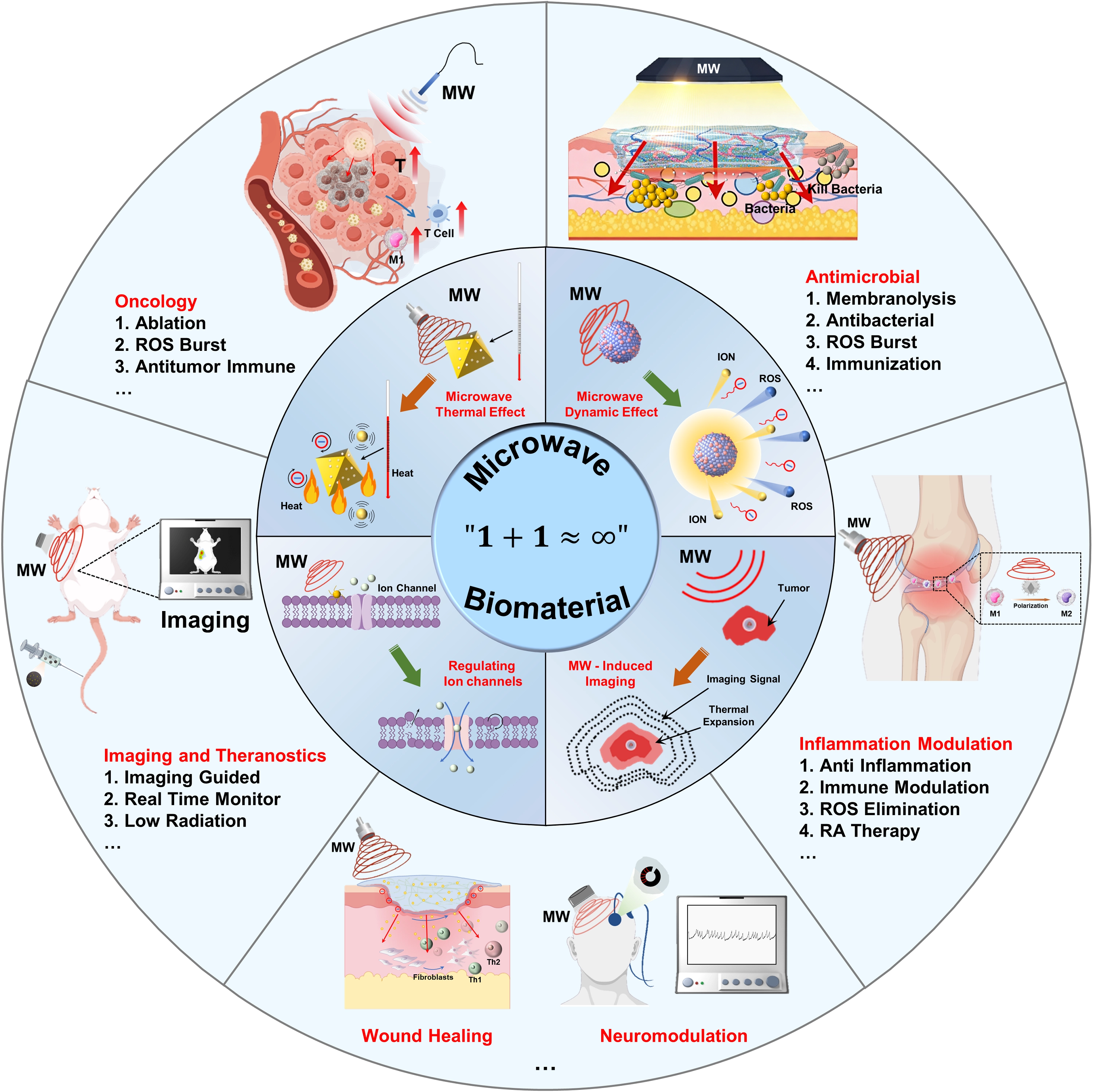
Scheme 1. Engineering biomaterials for microwave medicine: Physicochemical mechanisms and emerging biomedical applications. ROS: reactive oxygen species;
2. Biomaterial-Sensitized Tumor MWA
Among the various biomedical applications of microwave medicine, antitumor therapy represents one of the earliest and currently most intensively investigated directions involving biomaterials. This is mainly because, although microwave-based treatments possess notable physical advantages for the intervention of deep-seated tumors, their ability to precisely control local energy deposition and modulate subsequent biological effects remains insufficient to meet the practical demands of tumor therapy[51]. By introducing material systems with specific dielectric, electromagnetic, or biological functional properties, researchers have sought to reshape microwave energy deposition and the associated biological responses within tumor regions, without markedly increasing the external power burden. This approach aims to improve therapeutic efficiency while reducing the risk of side effects[52]. Toward this goal, biomaterial-assisted microwave antitumor strategies have gradually evolved from the simple enhancement of thermal effects to more comprehensive, multi-level approaches that integrate non-thermal effects and the regulation of post-treatment biological processes.
In early studies, biomaterials were introduced into microwave-based antitumor systems mainly to enhance local microwave hyperthermia or ablation effects. A large body of evidence has demonstrated that materials with high dielectric constants or pronounced dielectric loss properties, such as ionic liquids (ILs), carbon-based materials, and magnetic materials, can significantly increase energy absorption efficiency in tumor regions under microwave irradiation. As a result, effective tumor suppression can be achieved at lower power levels or with shorter irradiation times[53-56]. Such strategies have been widely applied in MWA and localized hyperthermia settings, where they help to alleviate issues related to non-uniform energy distribution and poorly defined ablation margins, and have shown consistent therapeutic outcomes across various tumor models. For example, in early work by Meng’s group, silica nanoparticles with or without a gold core were used as templates to synthesize hollow polydopamine (PDA) nanoparticles loaded with specific ILs. These particles (ILs/PDA) were introduced into the tumor site and served as a
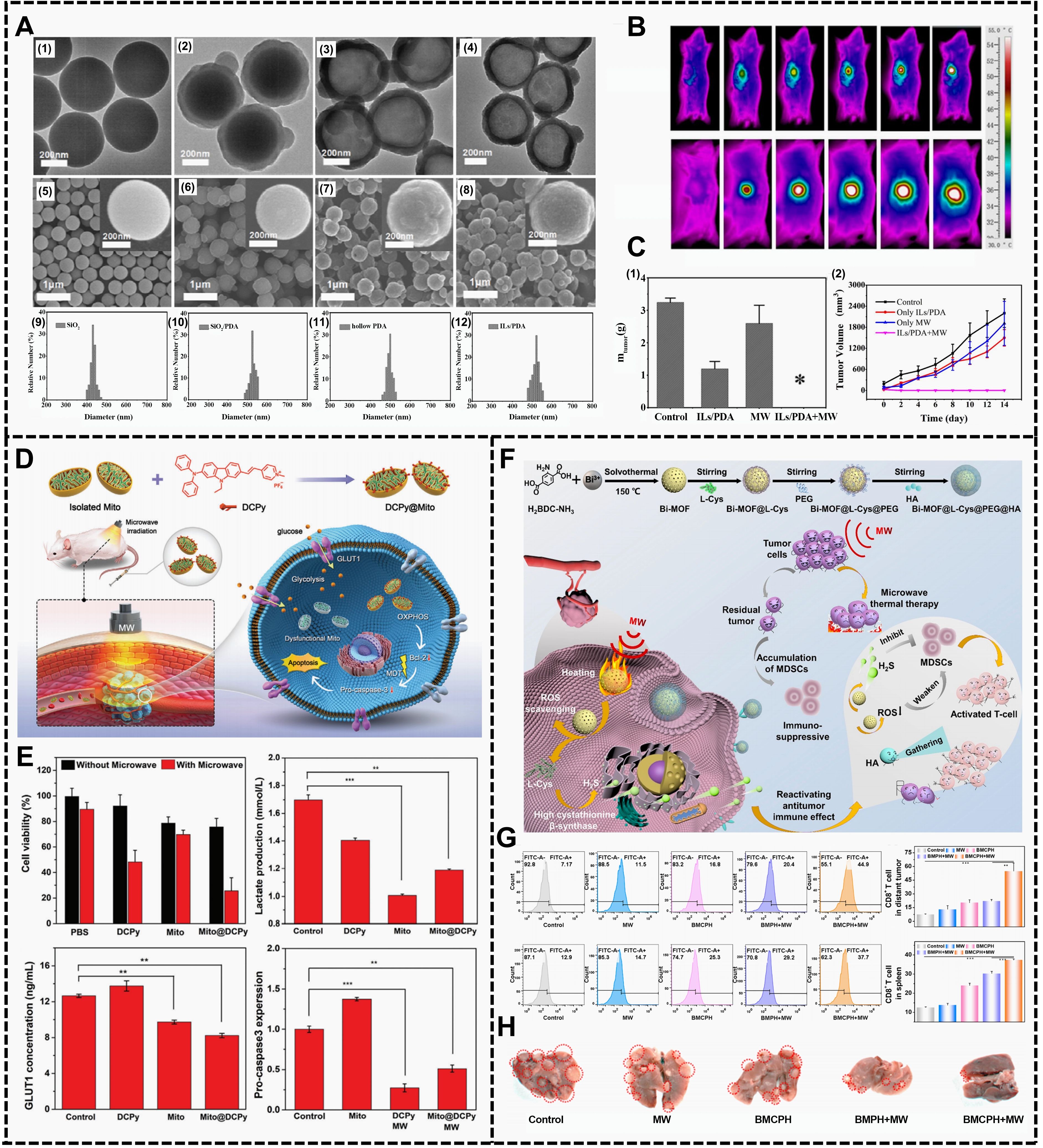
Figure 1. Application and mechanism of biomaterials in microwave tumor therapy. (A) TEM images of (1) SN; (2) SN/PDA core/shell nanoparticles; (3) hollow PDA nanoparticles and (4) ILs/PDA nanocomposites; SEM images of (5) SN; (6) SN/PDA core/shell nanoparticles; (7) hollow PDA nanoparticles and (8) ILs/PDA nanocomposites. The insets are SEM images with higher magnifications. The size distributions of (9) SN; (10) SN/PDA core/shell nanoparticles; (11) hollow PDA nanoparticles and (12) ILs/PDA nanocomposites; (B) Near infrared thermal imaging of ICR mice bearing H22 tumors under microwave treatment for 5 min at 1 min intervals; (C) The curves of relative tumor weight (1) and tumor volumes (2) in the different treatment groups. (* denotes statistical significance for the comparison of other groups, *P < 0.05). Republished with permission from[57]; (D) Preparation of AIEgen-engineered bioactive mitochondria and their application for microwave-dynamic cancer therapy; (E) Healthy Mito@DCPy transplants not only downregulate the Bcl-2 overexpression on their own but also enhance the treatment effect of ROS production by MW irradiation, making the therapeutic efficiency of MDT more synergistic. Republished with permission from[58]; (F) Synthesis of BMCPH nanoparticles and a schematic diagram of BMCPH for mediating tumor MWTT and reactivating the antitumor immune effect; (G) CD8+ T cells in distant tumors and spleen tissues; (H) Metastatic tumor formation in the lung. Republished with permission from[67]. TEM: transmission electron microscopy; SN: silica nanoparticles; PDA: polydopamine; ILs: ionic liquids; SEM: scanning electron microscope; ICR: Institute of Cancer Research; GLUT1: glucose transporter 1; MDT: microwave-dynamic cancer therapy; OXPHOS: oxidative phosphorylation; ROS: reactive oxygen species;
Building on thermal enhancement strategies, some studies have further explored the antitumor potential of microwave therapy under non-ablative or mild-temperature conditions. By introducing biomaterials with catalytic or electromagnetic-responsive properties, microwave irradiation can induce the generation of ROS, thereby triggering oxidative stress and apoptotic responses in tumor cells. Compared with conventional high-temperature ablation, this microwave dynamic therapy modality achieves significant antitumor efficacy while maintaining a relatively low temperature rise, indicating a potential advantage in reducing heat-related side effects. For example, Ben’ group screened a mitochondrial-targeting microwave sensitizer (mitochondrial-targeting
Furthermore, in recent years, there has been an increasing number of studies attempting to apply the combined effects of microwave thermal and non-thermal effects to tumor treatment in order to enhance the local tumor-killing effect[59-61]. Inspired by the combined application strategies of various anti-tumor methods such as photodynamic therapy and chemotherapy, biomaterial-mediated microwave therapy has gradually been introduced and integrated into the design of other mature treatment methods[62-66]. Considering that residual tumor tissues after incomplete MWA may induce immunosuppression and thereby increase the risks of tumor recurrence and metastasis, a recent study constructed a microwave-responsive bismuth-based metal-organic framework (MOF) nano-immunomodulatory system (Bi-MOF@L-Cys@PEG@HA, BMCPH)[67]. During microwave hyperthermia, this system simultaneously enabled tumor ablation, ROS scavenging, and H2S-responsive release, thereby reversing immunosuppression induced by incomplete ablation and activating antitumor immune responses. As a result, tumor recurrence and metastasis were effectively suppressed (Figure 1F,G,H). This strategy demonstrates that the introduction of biomaterials can not only enhance the local tumoricidal effects of microwave therapy, but also regulate the tumor immune microenvironment during the post-treatment stage. Such systemic-level modulation helps reduce the risks of recurrence and metastasis, and provides new insights for improving the overall efficacy of microwave-based antitumor therapies.
In summary, the introduction of biomaterials into microwave-based antitumor therapy has shifted microwave action from reliance on local temperature elevation as a single physical parameter to a treatment process that can be regulated at multiple levels through material design. By modulating the manner in which microwave energy is deposited within tumor regions and coupling this process with biological effects such as oxidative stress or immune regulation, biomaterials have significantly expanded the controllable space of microwave antitumor therapy in terms of therapeutic efficacy, safety, and long-term tumor control. These studies indicate that rational material design is a critical foundation for driving the transition of microwave therapy from single energy-based action to controllable biological effects.
3. Microwave Antimicrobial Strategies
Bacterial infections, particularly those involving deep tissues and drug-resistant pathogens, remain a major challenge in current clinical practice. Conventional antibiotic therapies not only face the problem of rapidly increasing antimicrobial resistance, but also suffer from limited effective accumulation at local infection sites due to tissue barriers and microenvironmental constraints[68]. To address the antimicrobial resistance crisis, alternative strategies that are antibiotic-free or capable of enhancing antibiotic efficacy, such as phototherapy, have been actively explored. However, the limited penetration depth of near-infrared light restricts these approaches to the treatment of superficial wound infections[69], making them unsuitable for deep tissue infections. In recent years, MWs have attracted increasing attention for antibacterial applications because of their strong tissue penetration and high energy transfer efficiency[70].
In the context of food safety, microwave irradiation can inactivate bacteria through thermal effects. However, when microwave hyperthermia is directly applied to antibacterial treatment, prolonged microwave exposure may cause excessive heating and inevitably result in thermal injury to normal tissues, thereby limiting its application in infection control[71]. To address this issue, researchers have introduced materials with specific electromagnetic functions, as well as bacteria-targeting materials capable of controlled antibiotic release. These approaches enhance the local efficiency of microwave action at infection sites and allow for reduced microwave dosage or temperature elevation. When combined with chemotherapy, they enable more effective and controllable antibacterial outcomes. For example, the group of Wu Shuilin proposed a microwave-assisted antibacterial strategy that integrates material-based targeting with magnetic targeting advantages[72]. By constructing a microwave-responsive Fe3O4/carbon nanotubes (CNTs)/gentamicin (Fe3O4/CNT/Gent) nanocomposite system, this system enabled precise capture of methicillin-resistant Staphylococcus aureus (MRSA) in deep tissues, mild localized heating, bacterial membrane disruption, and synergistic drug release. As a result, the system effectively suppressed MRSA dissemination along blood vessels and achieved efficient bacterial clearance, demonstrating broad potential for the treatment of osteomyelitis and other deep-seated bacterial infections (Figure 2A,B,C,D).
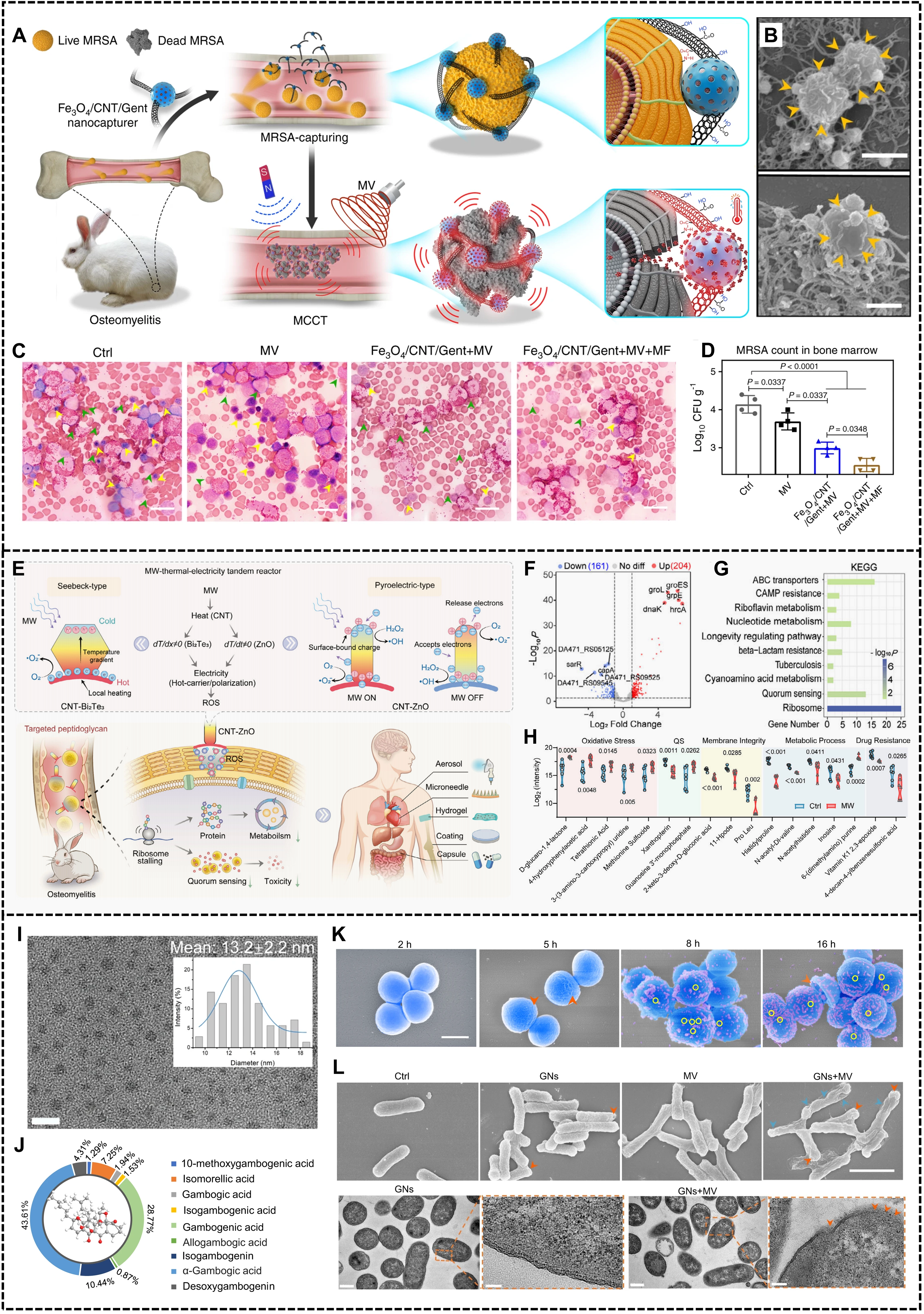
Figure 2. Application and mechanism of biomaterial enhanced microwave antibacterial. (A) The schematic illustration of the MCCT of Fe3O4/CNT/Gent (Fe3O4/carbon nanotubes/gentamicin); (B) Representative SEM observation of Fe3O4/CNT/Gent binding to MRSA; (C) Wright-stained images of infected bone marrow tissue after 14 days of treatment. Scale bars = 20 μm; (D) The MRSA counts in the infected bone marrow after 2 days with different treatments[72]; (E) Schematic illustration of MW-actuated
However, the long-term use of antibiotics still faces the problem of drug resistance. Therefore, combining microwave hyperthermia with microwave dynamic therapy represents a promising approach[14]. A recent study proposed a microwave-driven energy translation and amplification strategy, in which microwave thermal effects are coupled with thermoelectric or pyroelectric processes to significantly enhance non-thermal antibacterial effects[73]. Specifically, researchers combined microwave-sensitive CNTs with thermoelectric materials (Bi2Te3) or pyroelectric materials (ZnO) to construct a microwave–thermal–electric tandem reactor
In addition to material-mediated energy conversion for amplifying microwave antibacterial effects, the direct regulatory effect of microwave electromagnetic fields on bacterial structures has also received increasing attention. Gram-negative bacteria possess an asymmetric outer membrane composed of lipopolysaccharides (LPS) and phospholipids, which acts as a low-permeability barrier and significantly restricts the transmembrane entry of many antibacterial agents and nanomaterials[75]. Based on the high sensitivity of numerous polar molecules in the outer membrane to alternating electric fields, a recent study proposed using MWs as an external physical tool to disrupt the asymmetric outer membrane of Gram-negative bacteria and promote drug entry[76]. The study demonstrated that under microwave irradiation, LPS and phospholipid molecules in the outer membrane undergo continuous orientation rearrangement in response to the alternating electric field, inducing local structural disorder and weakening membrane integrity. This process generates transient pores that facilitate the entry of nanoparticles. Based on this mechanism, researchers developed Garcinia nanoparticles, originally a narrow-spectrum antibacterial system effective only against Gram-positive bacteria. Under microwave assistance, these nanoparticles were successfully delivered into Escherichia coli cells and completely eradicated bacterial pneumonia caused by mixed Gram-negative and Gram-positive infections in an in vivo model (Figure 2I,J,K,L). This work indicates that the synergistic use of biomaterials and MW can not only enhance bactericidal efficacy but also expand the antibacterial spectrum by modulating bacterial structural barriers, providing a new strategy for non-antibiotic treatment of complex deep infections.
Overall, the introduction of biomaterials has significantly expanded the dimensionality and regulatory scope of microwave-based antibacterial therapy. On one hand, by enhancing microwave absorption, enabling targeted accumulation, or constructing
4. Microwave-Mediated Inflammation Modulation
In contrast to infectious inflammation that is primarily driven by pathogen clearance, autoimmune inflammation and tissue
Autoimmune inflammation, such as RA, is currently treated in clinical practice mainly with glucocorticoids, nonsteroidal
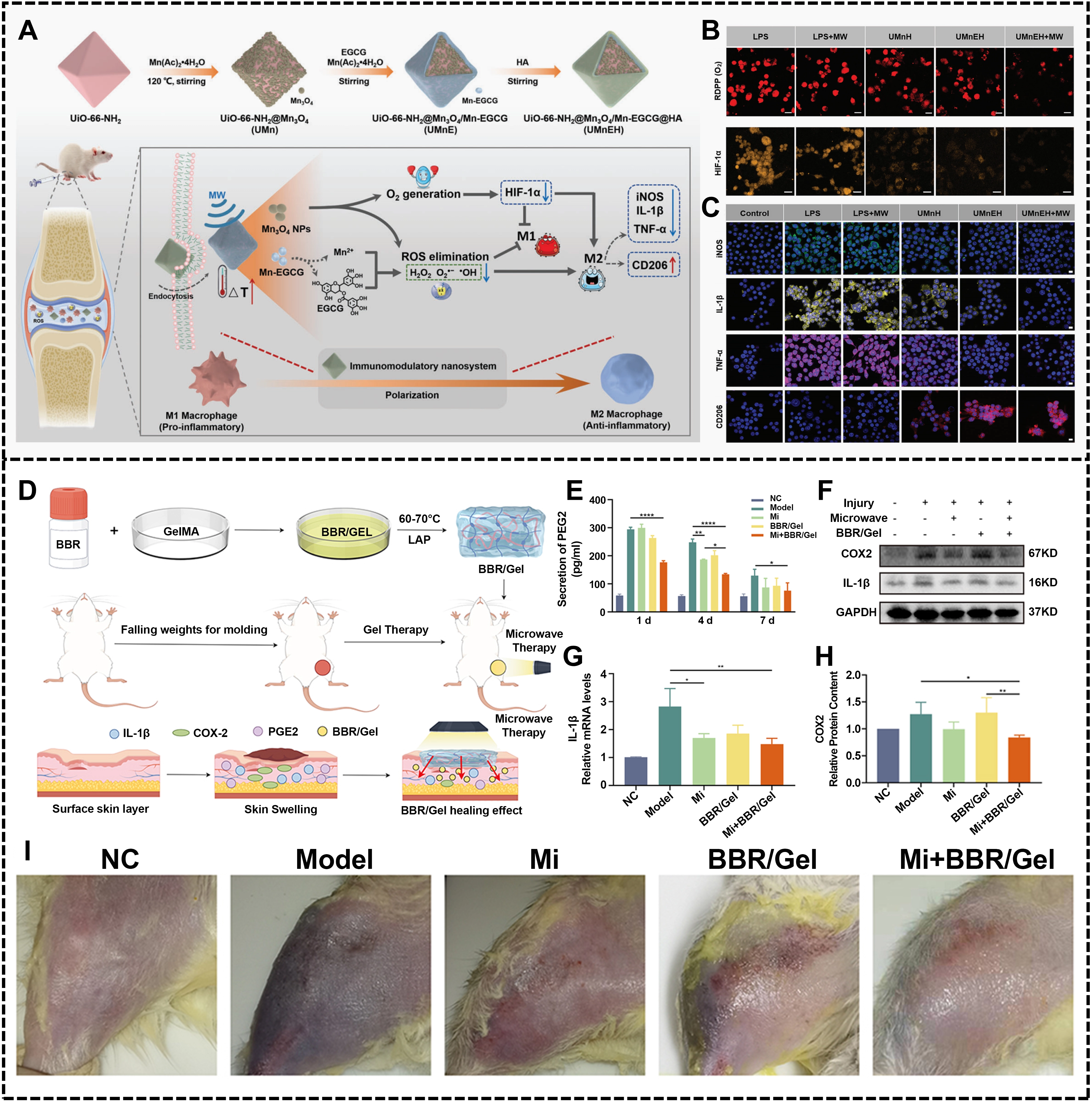
Figure 3. Biomaterial enhanced microwave-mediated inflammation regulation. (A) Schematic illustration of the fabrication procedures and in vivo therapeutic mechanism of UMnEH (UiO-66-NH2@Mn3O4/Mn-EGCG@HA) against RA; (B) Fluorescence images of O2 level in the LPS-induced RAW 264.7 cells with different treatments for 24 h (Scale bar: 20 μm). Fluorescence images of HIF-1α level in the LPS-induced RAW 264.7 cells with different treatments for 24 h (Scale bar: 25 μm); (C) The CLSM images of LPS-induced RAW 264.7 cells incubated with UMnEH@Cy7 for different times (0, 2, 4, and 6 h, Scale bar: 20 μm). Republished with permission from[83]; (D) Effectiveness of microwave therapy combined with Berberine/GelMA via COX-2/IL-1β pathway to treat skeletal muscle injury: An in vivo study in rats; (E) The expression levels of IL-1β, PGE2 in the serum of SD rats in each group at 1/4/7 days after modeling; (F-H) Expression levels of IL-1β, COX-2 in skeletal muscle of SD rats in each group. (*P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001); (I) Demonstration of modeling effects of soft tissue injury in different groups of SD rats[86]. RA: rheumatoid arthritis; LPS: lipopolysaccharides;
In addition to the pathological inflammatory conditions described above, skeletal muscle injury is also an important target for
In summary, microwave-responsive biomaterials have demonstrated substantial therapeutic potential in both autoimmune inflammation and tissue injury–associated inflammation. Through the synergistic interaction between microwave non-thermal effects and material functionalities, this system enables precise regulation of immune cell phenotypes, and improvement of local hypoxia, while also allowing controlled release of drugs or bioactive components. As a result, the inflammatory microenvironment can be systematically remodeled, and excessive inflammatory responses are suppressed. This strategy balances local precision with systemic regulatory capacity, addressing key limitations of conventional pharmacological treatments in terms of targeting specificity, therapeutic durability, and side effects. Moreover, it provides a scalable and versatile platform for the clinical management of chronic autoimmune diseases and trauma-related inflammation, thereby opening new avenues for the application of microwave medicine in anti-inflammatory therapy.
5. Nano-Contrast Agents for Microwave Imaging
As microwave medicine gradually evolves from empirical treatments toward precision and individualized therapy, achieving accurate lesion localization and visualized monitoring of the treatment process has become a critical challenge limiting its further clinical application. The dielectric differences between normal and pathological tissues, such as tumors, provide the basis for microwave imaging. By measuring the scattered microwave field generated by a material, its dielectric constant distribution can be obtained, which forms the goal of microwave imaging[87,88]. However, the limited intrinsic dielectric contrast of biological tissues, the difficulty of precisely locating small lesions, and the relatively immature imaging systems pose significant challenges for single-modality microwave imaging in accurately identifying complex lesions and guiding microwave therapy[89-91]. In recent years, biomaterials with microwave-responsive properties have been introduced into imaging systems. By amplifying local microwave energy deposition signals and coupling with multimodal imaging techniques, they provide a new strategy for constructing integrated imaging, guidance, therapy, and feedback microwave diagnostic and therapeutic platforms.
On one hand, some microwave-absorbing materials inherently possess magnetic properties or high atomic number characteristics, making them naturally compatible with clinical imaging modalities such as magnetic resonance imaging (MRI) and computed tomography, thereby enabling lesion localization and therapy guidance. For example, a recent study constructed manganese-doped titanium metal-organic framework (Mn-Ti MOFs) nanosheets via an in-situ doping strategy for microwave treatment of liver
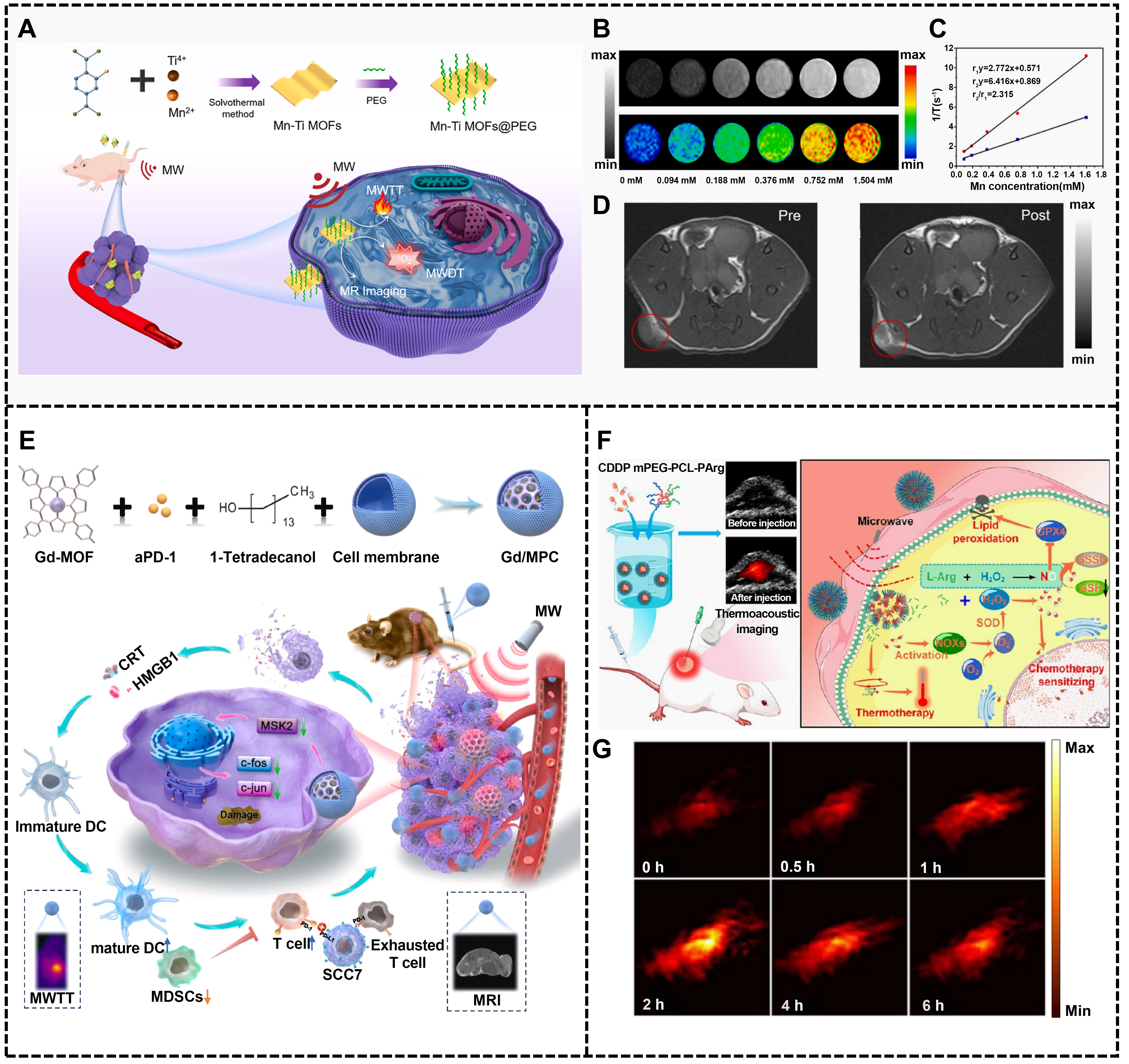
Figure 4. Biomaterial enabled microwave imaging and diagnosis. (A) Schematic images for the synthesis process of Mn-Ti MOFs@PEG nanosheets for MR imaging and MWDT-MWTT; (B) T1-weigthed MR images in vitro; (C) r1 and r2 (relaxation rate and transverse relaxation rate) values; (D) T1-weigthed MR images of Mn/Ti MOF-PEG in vivo[92]; (E) Schematic illustration of the synthesis process and therapeutic effects of Gd/MPC[51]; (F) Schematic illustration of the thermoacoustic imaging in vivo. Republished with permission from[93]. Mn-Ti MOFs: manganese-doped titanium metal-organic framework; PEG: polyethylene glycol; MR: magnetic resonance; MWDT-MWTT: microwave dynamic-thermal therapy; Gd/MPC: Gd-MOF@aPD-1@CM; CM: change material; NPs@CDDP: cisplatin nanocapsules.
On the other hand, materials may exhibit imaging capabilities under microwave excitation. Cui et al. designed a Gd-based
Recent advances in MITAI have significantly improved deep-tissue visualization by combining microwave contrast with acoustic resolution[94,95]. However, conventional MITAI primarily relies on uniform illumination and scalar energy deposition, which limits its ability to resolve sub-organ structures within complex biological tissues. Reconstruction algorithms, system design, and signal acquisition remain factors that limit the quality, signal-to-noise ratio, and resolution of microwave thermoacoustic imaging, and these limitations are particularly prominent in complex biological tissues with multi-layered structures and high heterogeneity[96,97]. To overcome this limitation, Wang et al. proposed an electric vector–adapted thermoacoustic computed tomography (EV-TACT) strategy, in which microwave illumination with different polarizations is employed to selectively enhance energy coupling to tissue structures with specific orientations[98]. By explicitly considering the propagation behavior and polarization characteristics of MW in biological tissues, EV-TACT effectively amplifies thermoacoustic signal excitation from anisotropic sub-organ features, thereby improving the signal-to-noise ratio and revealing structures that are otherwise obscured in conventional MTAT. Notably, EV-TACT enabled label-free, high-contrast imaging of nerve fiber bundles in the brain and dynamic vascular structures in the abdomen of live mice, demonstrating its potential for resolving fine structural and functional information in deep tissues. This work highlights that, beyond material-assisted contrast enhancement, precise regulation of microwave field parameters, such as electric vector orientation, represents a powerful and complementary strategy for improving imaging specificity and spatial resolution in microwave-based biomedical imaging.
Overall, the introduction of biomaterials is driving the evolution of microwave medicine from one-way treatment toward a diagnostic and therapeutic system. Real-time imaging enables acquisition of lesion information and energy deposition distribution, allowing dynamic adjustment of microwave treatment parameters. Post-treatment imaging feedback can further provide objective assessment of therapeutic efficacy and tissue response, supporting more precise and individualized treatment decisions. Looking forward, with the further development of smart responsive materials, multimodal imaging technologies, and microwave devices, integrated platforms that combine imaging guidance, therapy enhancement, and efficacy feedback are expected to become a key direction in microwave medicine, laying a foundation for its clinical translation and standardized application.
6. Emerging and Unconventional Applications
Beyond anticancer, antibacterial, anti-inflammatory therapies, and imaging applications, the integration of MW and biomaterials has also shown exploratory potential in emerging biomedical fields, such as tissue repair and neural modulation. In tissue regeneration and wound healing, microwave therapy has been reported to significantly accelerate wound closure in diabetic mice by enhancing granulation tissue formation, collagen remodeling, and myofibroblast activation[99]. By incorporating biomaterials with controllable thermal conductivity and microwave responsiveness, microwave energy can be precisely regulated, thereby promoting tissue repair while minimizing thermal damage. Zhang from Taiyuan University of Technology developed a Fe3O4@AIPH (2,2-Azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride; AIPH) microneedle patch. The Fe3O4@AIPH core-shell nanoparticles were fabricated by a self-assembly method and then loaded on hyaluronic acid microneedle patches[100]. The microwave absorption characteristics of
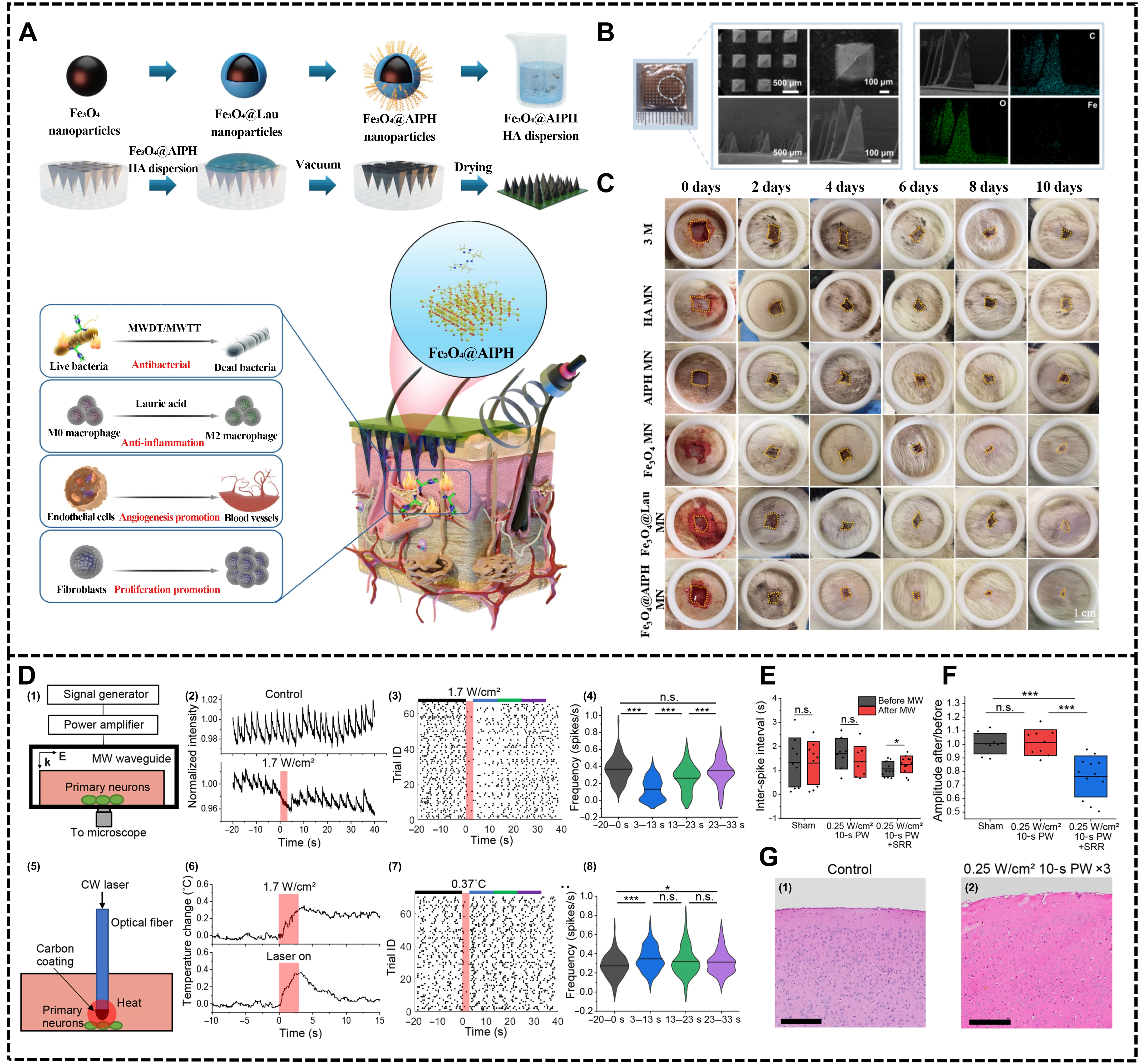
Figure 5. Biomaterials expand the novel and unconventional applications of microwave medicine. (A) The schematic diagram of the preparation of Fe3O4@AIPH microneedle patches and the mechanism of promoting the healing of diabetic infected wounds; (B) SEM and EDS mapping images of Fe3O4@AIPH microneedle patch (Scale bar = 500 μm, 100 μm); (C) Representative images of the effect of each treatment group on promoting wound healing within 10 days (Scale bar = 1 cm)[100]; (D) (1-8) Microwave inhibits neuronal activity via a nonthermal mechanism; (E) Inter-spike interval for 10 s before and after treatment in sham mice, direct microwave treatment, and microwave SRR treatment; the solid line represents data mean and the box represents SD; statistical significance was calculated using a paired sample t-test where *P < 0.05; (F) Percent change in spike amplitude for 10 s after microwave treatment in sham mice, direct microwave mice, and microwave SRR mice; the solid line represents data mean and whiskers represent SD; statistical significance was calculated using a two-sample t-test where ***P < 0.001; (G) Control brain tissue and brain tissue after three microwave treatments stained with H&E (Scale bar = 100 μm)[101]. SEM: scanning electron microscope; EDS: energy dispersive X-ray spectroscopy; SRR: split-ring resonator; SD: standard deviation; AIPH: 2,2-Azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride.
In addition to tissue repair, the potential application of MW in the field of neural modulation has also attracted increasing attention in recent years. As a non-ionizing form of energy capable of penetrating the skull, MWs provide a possible approach for deep neural modulation. One study employed a microwave-enhanced electromagnetic structure and designed a split-ring resonator (SRR), which enabled the suppression of abnormal neuronal firing under microwave excitation[101]. In both in vitro and in vivo epilepsy models, this strategy significantly reduced neuronal calcium spike rate while maintaining local temperature elevation within a safe threshold, thereby demonstrating effective neural inhibition and high spatial precision (Figure 5E,F,G,H). Furthermore, it has been reported that low-intensity microwave electromagnetic fields can directly activate voltage-gated calcium channels, leading to a transient increase in intracellular Ca2+ levels and subsequent physiological responses[102].
Overall, microwave-responsive biomaterials for tissue repair and neural modulation are still at the proof-of-concept and early exploratory stages. Their underlying mechanisms, long-term safety, and capacity for precise regulation require further investigation. Nevertheless, these studies expand microwave medicine from conventional therapeutic ablation toward functional modulation, and provide important insights for its application in broader biomedical contexts.
As mentioned above, microwave therapy is a promising treatment strategy that can be applied in various biomedical applications
| Specific application | Nanoplatform | MW-responsive action mechanism | Features | Ref |
| Tumor Therapy | ILs/PDA | ILs has been investigated as a promising sensitizer to MW irradiation due to their ionic character and high polarizability. ILs can efficiently convert electromagnetic energy into thermal energy | ILs enhance the microwave thermal effect, and the PDA achieves tumor targeting | [57] |
| Mito@DCPy | Microwave irradiation can penetrate very deep tissues and sensitize the DCPy to generate reactive oxygen species | Mito@DCPy targets mitochondria. Under microwave irradiation, it generates ROS and restores oxidative phosphorylation, thereby enhancing the MW dynamic effect | [58] | |
| BMCPH | Bi-MOF enables efficient microwave hyperthermia, serves as an excellent carrier for L-Cys, and can also removes ROS | BMCPH improves MWTT by responsively releasing H2S in tumor cells, thereby reversing immunosuppression and reactivating antitumor immunity | [67] | |
| Anti-Bacteria | Fe3O4/CNT/Gent | Interfacial polarization at CNT/Fe3O4 interfaces increases dielectric and magnetic loss, leading to improved microwave absorption and thermal effect | Fe3O4/CNT constructs a magneto–dielectric microwave-responsive system, enabling magnetic targeting, microwave hyperthermia, and controlled antibiotic release for synergistic antibacterial therapy | [72] |
| CNTs-Bi2Te3 & | The CNT converts MW into a local heat source and rapidly heats Bi2Te3 or ZnO to activate hot-carrier or polarization for | The ROS generated in situ by the MW-thermal-electricity tandem reactors synergize with the ribosome stalling triggered by MW to achieve efficient sterilization | [73] | |
| Fe3O4/CuS/Emodin | The synergy of magnetic-dielectric loss in | Fe3O4/CuS/Emodin enhances microwave response while modulating the immune microenvironment for effective bacterial clearance | [74] | |
| GN + MW | MW creates transient nanopores in bacterial outer membrane | Microwave-induced outer membrane poration significantly boosts the efficacy of narrow-spectrum GN particles against E. coli | [76] | |
| Inflammation intervention | UiO-66-NH2@Mn3O4/Mn-EGCG@HA | UiO-66-NH2 can enhance microwave sensitization and plays the role of microwave hyperthermia in the treatment of inflammation | UiO-66-NH2–mediated microwave hyperthermia, together with Mn3O4-enabled ROS scavenging and O2 generation, downregulates HIF-1α and promotes M1-to-M2 macrophage polarization | [83] |
| UCCH | UiO-66-NH2 with a large specific surface area and abundant micropores can contribute to recruiting the surrounding ions to improve the confined inelastic collision of ions to increase the efficacy of microwave energy conversion to hyperthermia | UCCH exerts anti-inflammatory effects via microwave hyperthermia, CeO2-mediated ROS scavenging/O2 generation–induced M1-to-M2 repolarization, and microwave-triggered drug release | [79] | |
| BBR/GelMA | Microwave accelerates the absorption of BBR in the composite nanogel of BBR/GelMA, to achieve a rapid treatment effect of skeletal muscle injury | BBR modulates the TLR/NF-kB signaling pathway to block the inflammatory response | [86] | |
| Imaging | Mn-Ti MOFs@PEG | The porous structure of Mn-Ti MOFs increases microwave-induced ion collision frequency; Mn doping narrows the bandgap and endows MRI capability | While increasing the thermal effect and the production capacity of ROS, the Mn in the MOF enhanced the magnetic resonance imaging capability | [92] |
| Gd/MPC | Gd, a MRI contrast agent, enables Gd-based MOFs to exhibit effective microwave sensitization | MW promoted rapid warming, combined with aPD-1 to enhance the intensity of immune response | [51] | |
| NPs@CDDP | L-Arg with excellent microwave-absorbing property allowed it to serve as a thermoacoustic imaging contrast agent for accurately delineating the tumor and remarkably increasing tumor temperature under ultralow power microwave irradiation | Non-invasive thermal imaging and low-power MW radiation enhance intracellular lipid peroxidation and improve the efficacy of chemotherapy | [93] | |
| Tissue repair | Fe3O4@AIPH | Fe3O4 is an ideal wave-absorbing material because of the extremely high dielectric loss angular tangent and magnetic loss tangent values | Fe3O4@AIPH nanoparticles can realize efficient and controllable chemodynamic antibacterials under microwave irradiation through a microwave-thermal-chemodynamic chain reaction, and accelerated the healing process of diabetic wounds | [100] |
| Neural modulation | SRR | SSR couples the microwave wirelessly and concentrates it at the gap, producing a localized electrical field | Significantly improve the spatial resolution of neural regulation and inhibit abnormal discharges of neurons | [101] |
MW: microwave; ILs: ionic liquids; PDA: polydopamine; ROS: reactive oxygen species; BMCPH: Bi-MOF@L-Cys@PEG@HA; MOF: metal-organic framework; L-Cys: L-cysteine; MWTT: microwave thermal therapy; CNT: carbon nanotube; GN: garcinia nanoparticle; HIF: hypoxia-inducible factor; UCCH: UiO-66-NH2/CeO2/Cel@HA; BBR: berberine;
7. Conclusions and Future Perspectives
In summary, microwave medicine is evolving from conventional macroscopic thermal therapy toward a mechanism-driven paradigm enabled by engineered biomaterials. By tailoring dielectric and magnetic losses, band structures, and polarization properties, these materials allow precise modulation of microwave–tissue interactions at the micro- and nanoscale. Such designs expand microwave functionality beyond bulk heating, enabling localized energy concentration, microwave-induced ROS generation, and regulation of ionic homeostasis. Through these mechanisms, biomaterial-assisted microwave systems have demonstrated versatile therapeutic capabilities across antitumor, antibacterial, and anti-inflammatory applications, while also advancing high-resolution thermoacoustic imaging. Collectively, these developments establish a unified framework in which materials act as active mediators of electromagnetic energy transduction, facilitating controlled and multifaceted biological regulation.
Future development is expected to move beyond enhancing a single effect toward multifunctional integration and coordinated regulation. Disease-associated microenvironments often involve simultaneous changes in redox balance, energy metabolism, immune responses, and tissue structure. Relying solely on thermal effects or a single type of active species is unlikely to achieve optimal intervention. By rationally designing material composition and structure, it is possible to integrate microwave absorption, energy translation, biological modulation, and imaging feedback into a single platform. This integration can induce multiple synergistic biological responses from a single microwave stimulus, thereby improving overall therapeutic efficiency and stability. Such multifunctional platforms not only broaden the scope of microwave medicine but also provide a material basis for systematic regulation of complex biological processes under pathological conditions.
In this context, a deep understanding and precise control of synergistic sensitization and energy translation mechanisms are particularly critical. Recent studies coupling microwave thermal effects with thermoelectric, pyroelectric, piezoelectric, or magnetic responses have demonstrated unique advantages in amplifying non-thermal biological effects. However, most current research remains at the functional validation stage, with limited quantitative understanding of the contribution ratios, spatiotemporal distribution, and interactions of different translation pathways in biological environments. Future studies should combine in situ characterization techniques with multiscale theoretical models to systematically reveal the intrinsic relationships among material properties, microwave frequency, and irradiation conditions. This approach would enable predictable control of both the intensity and type of biological responses, laying the foundation for the engineering and standardization of microwave medicine.
While the direct effects of microwaves are undoubtedly important, the dynamic evolution of the post-treatment microenvironment is increasingly recognized as a key determinant of long-term therapeutic outcomes. Microwave-induced oxidative stress, structural reorganization, and cellular damage are often accompanied by the release of various endogenous signaling molecules and metabolic byproducts, providing a natural entry point for in situ immune modulation and inflammatory remodeling. By introducing biomaterials that respond to microwave stimulation or changes in the lesion microenvironment, and equipping them with the capacity for molecular capture, enrichment, and controlled release, sustained regulation of immune and inflammatory responses can be achieved during the critical post-treatment phase. Further optimization of the material’s response threshold and release kinetics enables on-demand regulation triggered by microwave or microenvironmental cues, prolonging local action while minimizing systemic side effects, thereby promoting tissue homeostasis and reducing the risk of disease recurrence. This in situ regulation strategy extends microwave therapy from a single intervention to full-course management, demonstrating greater application potential in areas such as oncology. Simultaneously, the development of imaging-guided and integrated therapeutic platforms will further improve the precision and reproducibility of microwave medicine. By endowing microwave-responsive materials with imaging functionality or utilizing signal changes induced under microwave excitation, lesion localization, treatment monitoring, and efficacy assessment can be seamlessly integrated. Imaging feedback not only helps optimize microwave irradiation parameters but also provides critical information for personalized treatment planning. In the future, closed-loop systems combining real-time imaging with microwave modulation are expected to dynamically regulate biological responses, maximizing efficacy while minimizing adverse effects.
Although microwave-responsive biomaterials have shown considerable potential in disease treatment, they still face many practical challenges before they can be clinically translated. Firstly, these material systems are usually complex in structure and often require the cooperation of multiple components such as metals, semiconductors, and organic molecules. It is difficult to ensure the reproducibility and quality controllability of batches during large-scale production. Secondly, the stability, biocompatibility, and long-term safety of the materials in the body have not been fully understood. Especially, their degradation behavior and metabolic pathways still have many unknowns. Moreover, the interaction mechanism between microwave, material, and biology is not yet clear, and there is a lack of quantitative correlation from physical parameters to biological effects, which directly restricts the accuracy of dose control and efficacy prediction. At the same time, the highly heterogeneous tumor or inflammatory microenvironment also makes it difficult to maintain consistent treatment effects. Finally, the relevant regulatory system is not yet complete, standardized evaluation methods are lacking, and the high production cost also slows down the pace of clinical
Acknowledgements
The authors used ChatGPT to improve the clarity and readability of the manuscript. All scientific content, interpretations, and conclusions were developed by the authors.
Authors contribution
Song R: Writing-original draft.
Wang S: Conceptualization, resources, visualization, writing-review & editing.
Zhang L, Lu C: Writing-review & editing.
Wang Y, Yuan M: Supervision, writing-review & editing.
Wen L: Funding acquisition, writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This research was funded by the National Natural Science Foundation of China (Grant No. 82302368), the Guangxi Natural Science Foundation (Grant No. 2025GXNSFAA069529), Guangdong Provincial Applied Science and Technology Research and Development Program (Grant No. 2021B1212040004), Guangdong Province Colleges and Universities Characteristic Innovation Project (Grant No. 2025KTSCX173), and Zhuhai Basic and Applied Basic Research Foundation (Grant No. 2320004002697).
Copyright
© The Author(s) 2026.
References
-
1. Zhao Z, Qing Y, Kong L, Xu H, Fan X, Yun J, et al. Advancements in microwave absorption motivated by interdisciplinary research. Adv Mater. 2024;36(4):2304182.[DOI]
-
2. Du Y, Zhou J, He F, Zang P, Gong H, Liu C, et al. A bright future: Advanced nanotechnology-assisted microwave therapy. Nano Today. 2023;52:101963.[DOI]
-
3. Li J, Li Z, Liu X, Li C, Zheng Y, Yeung KWK, et al. Interfacial engineering of Bi2S3/Ti3C2Tx MXene based on work function for rapid photo-excited bacteria-killing. Nat Commun. 2021;12:1224.[DOI]
-
4. Chen J, Liu B, Peng G, Zhou L, Tan C, Qin J, et al. Achieving high-performance transcranial ultrasound transmission through Mie and Fano resonance in flexible metamaterials. Adv Sci. 2025;12(19):2500170.[DOI]
-
5. Fry FJ, Barger JE. Acoustical properties of the human skull. J Acoust Soc Am. 1978;63(5):1576-1590.[DOI]
-
6. Wu Q, Yu J, Li M, Tan L, Ren X, Fu C, et al. Nanoengineering of nanorattles for tumor treatment by CT imaging-guided simultaneous enhanced microwave thermal therapy and managing inflammation. Biomaterials. 2018;179:122-133.[DOI]
-
7. Wen L, Ding W, Yang S, Xing D. Microwave pumped high-efficient thermoacoustic tumor therapy with single wall carbon nanotubes. Biomaterials. 2016;75:163-173.[DOI]
-
9. Violi NV, Duran R, Guiu B, Cercueil JP, Aubé C, Digklia A, et al. Efficacy of microwave ablation versus radiofrequency ablation for the treatment of hepatocellular carcinoma in patients with chronic liver disease: A randomised controlled phase 2 trial. Lancet Gastroenterol Hepatol. 2018;3(5):317-325.
-
10. Groeschl RT, Pilgrim CH, Hanna EM, Simo KA, Swan RZ, Sindram D, et al. Microwave ablation for hepatic malignancies: A multiinstitutional analysis. Ann Surg. 2014;259(6):1195-1200.[DOI]
-
11. Golden ED, Mutlu A, Knavel-Koepsel EM, Louis J, Shapiro DD, Abel EJ, et al. Microwave ablation for renal cell carcinoma: A literature review and clinical insights. Radiology. 2025;6(1):71-85.[DOI]
-
13. Bichot A, Lerosty M, Radoiu M, Méchin V, Bernet N, Delgenès JP, et al. Decoupling thermal and non-thermal effects of the microwaves for lignocellulosic biomass pretreatment. Energy Convers Manag. 2020;203:112220.[DOI]
-
15. Sun Q, Li J, Zhang H, He X, Wu B, Wang J, et al. The MOFs/COFs-derivant decorating FeSiAl coupling magnetic and electrical losses for enhanced microwave absorption. Appl Surf Sci. 2024;651:159242.[DOI]
-
16. Wang J, Gao D, Xie J, Hu W. Polaron hopping induced giant room-temperature magnetodielectric effect in disordered rutile NiNb2O6. Adv Funct Mater. 2021;31(52):2108950.[DOI]
-
17. Wang X, Pan F, Xiang Z, Zeng Q, Pei K, Che R, et al. Magnetic vortex core-shell Fe3O4@C nanorings with enhanced microwave absorption performance. Carbon. 2020;157:130-139.[DOI]
-
18. Xiong Y, Yao H, Qiu D, Omran M, Wei S, Ren J, et al. Influence of localized thermal effect of microwave heating on the carbothermic reduction process of ZnFe2O4. J Clean Prod. 2024;477:143887.[DOI]
-
20. Shu M, Wang J, Xu Z, Lu T, He Y, Li R, et al. Targeting nanoplatform synergistic glutathione depletion-enhanced chemodynamic, microwave dynamic, and selective-microwave thermal to treat lung cancer bone metastasis. Bioact Mater. 2024;39:544-561.[DOI]
-
21. Yu Y, Wu Q, Niu M, Gou L, Tan L, Fu C, et al. A core-shell liquid metal-Cu nanoparticle with glutathione consumption via an in situ replacement strategy for tumor combination treatment of chemodynamic, microwave dynamic and microwave thermal therapy. Biomater Sci. 2022;10(13):3503-3513.
-
22. Guo Z, Zhu S, Yong Y, Zhang X, Dong X, Du J, et al. Synthesis of BSA-coated BiOI@Bi2S3 semiconductor heterojunction nanoparticles and their applications for radio/photodynamic/photothermal synergistic therapy of tumor. Adv Mater. 2017;29(44):1704136.[DOI]
-
24. Li J, Yang Z, Wang C, Wu S, Zheng Y, Cui Z, et al. Rapid electron transfer via hetero-interface engineering of 2D MOF anchored Ti3C2 MXene nanosheet for enhanced photocatalytic disinfection. Appl Catal B Environ. 2023;339:123163.[DOI]
-
25. Chen H, Kong X, Yan J, Wang T, Lv Y, Zhang F, et al. “Butterfly-effect” nano-platforms for cascade immune amplification and tumor clearance through multi-modal therapy and immune sensitization. Chem Eng J. 2024;495:153103.[DOI]
-
26. Tang XL, Wang Z, Zhu YY, Xiao H, Xiao Y, Cui S, et al. Hypoxia-activated ROS burst liposomes boosted by local mild hyperthermia for photo/chemodynamic therapy. J Control Release. 2020;328:100-111.[DOI]
-
27. Paulides MM, Trefna HD, Curto S, Rodrigues DB. Recent technological advancements in radiofrequency- andmicrowave-mediated hyperthermia for enhancing drug delivery. Adv Drug Deliv Rev. 2020;163:3-18.[DOI]
-
28. Chen Z, Guo W, Tan L, Fu C, Wu Q, Ren X, et al. Biomimetic MOF-based nano-immunoactivator via disruption of ion homeostasis for strengthened tumor microwave-immunotherapy. Adv Funct Mater. 2024;34(36):2401359.[DOI]
-
30. Xu L, Peng M, Gao T, Wang D, Lian X, Sun H, et al. Nanoenabled intracellular metal ion homeostasis regulation for tumor therapy. Adv Sci. 2024;11(7):2306203.[DOI]
-
31. Huang Q, Zhu W, Gao X, Liu X, Zhang Z, Xing B. Nanoparticles-mediated ion channels manipulation: From their membrane interactions to bioapplications. Adv Drug Deliv Rev. 2023;195:114763.[DOI]
-
32. Chen X, Zhang S, Liu J, Ren M, Xing D, Qin H. Controlling dielectric loss of biodegradable black phosphorus nanosheets by iron-ion-modification for imaging-guided microwave thermoacoustic therapy. Biomaterials. 2022;287:121662.[DOI]
-
35. Zhang S, Li W, Chen X, Ren M, Zhang H, Xing D, et al. Manganous-manganic oxide nanoparticle as an activatable microwave-induced thermoacoustic probe for deep-located tumor specific imaging in vivo. Photoacoustics. 2022;26:100347.[DOI]
-
37. Tucci C, Trujillo M, Berjano E, Iasiello M, Andreozzi A, Vanoli GP. Mathematical modeling of microwave liver ablation with a variable-porosity medium approach. Comput Meth Programs Biomed. 2022;214:106569.[DOI]
-
38. Zeng X, Cheng X, Yu R, Stucky GD. Electromagnetic microwave absorption theory and recent achievements in microwave absorbers. Carbon. 2020;168:606-623.[DOI]
-
40. Li J, Wang Z, Luo R, Quan X, Fong HU, Cheng Q, et al. Tumor microenvironment triggered in situ coagulation of supramolecularly engineered platelets for precise tumor embolization. Adv Sci. 2025;12(26):2414879.[DOI]
-
43. Origlia C, Rodriguez-Duarte DO, Tobon Vasquez JA, Bolomey JC, Vipiana F. Review of microwave near-field sensing and imaging devices in medical applications. Sensors. 2024;24(14):4515.[DOI]
-
44. Li B, Chen X, Qiu W, Zhao R, Duan J, Zhang S, et al. Synchronous disintegration of ferroptosis defense axis via engineered exosome-conjugated magnetic nanoparticles for glioblastoma therapy. Adv Sci. 2022;9(17):2105451.[DOI]
-
46. Zheng H, Ran P, Cao W, Zhang G, Su Y, Zhang K, et al. Shear stress-triggered thrombolysis by yolk–shell nanoparticles with piezoelectric-tribovoltaic dynamic Schottky junctions. Adv Funct Mater. 2025;35(42):2503751.[DOI]
-
47. Stoykov NS, Jerome JW, Pierce LC, Taflove A. Computational modeling evidence of a nonthermal electromagnetic interaction mechanism with living cells: Microwave nonlinearity in the cellular sodium ion channel. IEEE Trans Microw Theory Tech. 2004;52(8):2040-2045.[DOI]
-
48. Panagopoulos D, Karabarbounis A, Yakymenko I, Chrousos G. Human-made electromagnetic fields: Ion forced-oscillation and voltage-gated ion channel dysfunction, oxidative stress and DNA damage (Review). Int J Oncol. 2021;59(5):92.[DOI]
-
49. Bai S, Lan Y, Fu S, Cheng H, Lu Z, Liu G. Connecting calcium-based nanomaterials and cancer: From diagnosis to therapy. Nano Micro Lett. 2022;14(1):145.[DOI]
-
50. Wu Q, Yu J, Li M, Tan L, Ren X, Fu C, et al. Nanoengineering of nanorattles for tumor treatment by CT imaging-guided simultaneous enhanced microwave thermal therapy and managing inflammation. Biomaterials. 2018;179:122-133.[DOI]
-
51. Cui H, Zhao YY, Wu Q, You Y, Lan Z, Zou KL, et al. Microwave-responsive gadolinium metal-organic frameworks nanosystem for MRI-guided cancer thermotherapy and synergistic immunotherapy. Bioact Mater. 2024;33:532-544.[DOI]
-
56. Guo W, Kim ES, Chen Z, Wang Q, Wu Q, Tan L, et al. Microwave thermal supercharging therapy enables selective tumor ablation via low-power radio frequency-responsive nanotopographies. Bioact Mater. 2026;61:92-106.[DOI]
-
58. Yu X, Lyu M, Ou X, Liu W, Yang X, Ma X, et al. AIEgens/mitochondria nanohybrids as bioactive microwave sensitizers for non-thermal microwave cancer therapy. Adv Healthc Mater. 2023;12(12):2202907.[DOI]
-
59. Feng Y, Chen Q, Jin C, Ruan Y, Chen Q, Lin W, et al. Microwave-activated Cu-doped zirconium metal-organic framework for a highly effective combination of microwave dynamic and thermal therapy. J Control Release. 2023;361:102-114.[DOI]
-
60. Jin L, Wu S, Mao C, Wang C, Zhu S, Zheng Y, et al. Rapid and effective treatment of chronic osteomyelitis by conductive network-like MoS2/CNTs through multiple reflection and scattering enhanced synergistic therapy. Bioact Mater. 2024;31:284-297.[DOI]
-
62. Liu Z, Tan X, Huang Y, Li W, Yang N, Yuan R, et al. Microwave absorption-based magnetic liquid metal nano-missiles for thermodynamic/immunological cascade hepatoma therapy. Chem Eng J. 2023;471:144688.[DOI]
-
63. Chen R, Wen L, Guo F, He J, Wong KH, Chen M. Glutathione-scavenging natural-derived ferroptotic nano-amplifiers strengthen tumor therapy through aggravating iron overload and lipid peroxidation. J Control Release. 2025;379:866-878.[DOI]
-
68. Stokes JM, Yang K, Swanson K, Jin W, Cubillos-Ruiz A, Donghia NM, et al. A deep learning approach to antibiotic discovery. Cell. 2020;180(4):688-702.[DOI]
-
70. Zhang Z, Wang J, Hu Y, Wang L. Microwaves, a potential treatment for bacteria: A review. Front Microbiol. 2022;13:888266.[DOI]
-
71. Zhang WJ, Jin LG, Wu SL, Wang CF, Zheng YF, Li ZY, et al. Microwave excited hyperthermy and catalysis of heterostructured Au/Cu–BTA for effective bacteria killing by accelerating charge separation. Rare Met. 2024;43(10):5186-5201.[DOI]
-
73. Qiao Y, Sang Z, Zhang T, Liu X, He Q, Qin J, et al. Microwave-actuated hot-carrier/polarization triggers catalysis to Coordinate Staphylococcus aureus Ribosome stalling and treat deep-seated infections. Adv Mater. 2026;38(7):e02693.[DOI]
-
77. Ben H, Agarwal H, Gurnani B, Pradhan AA, Khan AA, Jain N. Breaking the barrier: Disruption of bacterial biofilms using microwave radiation. Front Cell Infect Microbiol. 2025;15:1670237.[DOI]
-
78. Cutolo M, Soldano S, Smith V, Gotelli E, Hysa E. Dynamic macrophage phenotypes in autoimmune and inflammatory rheumatic diseases. Nat Rev Rheumatol. 2025;21(9):546-565.[DOI]
-
80. Ma X, Ren X, Guo X, Fu C, Wu Q, Tan L, et al. Multifunctional iron-based Metal−Organic framework as biodegradable nanozyme for microwave enhancing dynamic therapy. Biomaterials. 2019;214:119223.[DOI]
-
86. Du T, Zhou L, Liu J, Wang X, Xie H, Yang X, et al. Effectiveness of microwave therapy combined with berberine/GelMA via COX-2/IL-1β pathway to treat skeletal muscle injury: An in vivo study in rats. Int J Nanomed. 2025;20:5509-5527.[DOI]
-
87. Nikolova N. Microwave imaging for breast cancer. IEEE Micro Mag. 2011;12(7):78-94.[DOI]
-
88. AlSawaftah N, El-Abed S, Dhou S, Zakaria A. Microwave imaging for early breast cancer detection: Current state, challenges, and future directions. J Imaging. 2022;8(5):123.[DOI]
-
91. Abbosh YM, Sultan K, Guo L, Abbosh A. Synthetic microwave focusing techniques for medical imaging: Fundamentals, limitations, and challenges. Biosensors. 2024;14(10):498.[DOI]
-
92. Qin Q, Yang M, Shi Y, Cui H, Pan C, Ren W, et al. Mn-doped Ti-based MOFs for magnetic resonance imaging-guided synergistic microwave thermal and microwave dynamic therapy of liver cancer. Bioact Mater. 2023;27:72-81.[DOI]
-
94. Wang B, Sun Y, Wang Z, Wang X. Three-dimensional microwave-induced thermoacoustic imaging based on compressive sensing using an analytically constructed dictionary. IEEE Trans Microw Theory Tech. 2020;68(1):377-386.[DOI]
-
96. Wang Y, Zhang HM, Qin H. Biomedical microwave-induced thermoacoustic imaging. Acta Phys Sin. 2023;72(20):204301.[DOI]
-
97. Lodi MB. A preliminary propagation study on magnetic scaffolds for microwave theranostics. In: 2023 IEEE 23rd International Conference on Nanotechnology (NANO); 2023 Jul 02-05; Jeju City, South Korea. Piscataway: IEEE; 2023. p. 807-812.[DOI]
-
98. Wang Y, Huang F, Fu Y, Tang S, Wen L, Qin H. Electric vector-adapted thermoacoustic computed tomography for dynamic imaging
sub-organ features in deep tissue. Appl Phys Lett. 2025;126(26):263703.[DOI] -
100. Wang J, Cheng H, An M, Yang Y, Han P, Qin J, et al. An antibacterial microneedle with chemodynamic therapy triggered by microwave thermal effect for diabetic infected wound healing. J Adv Res. 2026.[DOI]
-
102. Pall ML. Scientific evidence contradicts findings and assumptions of Canadian Safety Panel 6: Microwaves act through voltage-gated calcium channel activation to induce biological impacts at non-thermal levels, supporting a paradigm shift for microwave/lower frequency electromagnetic field action. Rev Environ Health. 2015;30(2):99-116.
-
103. Tong F, Wang Y, Gao H. Progress and challenges in the translation of cancer nanomedicines. Curr Opin Biotechnol. 2024;85:103045.[DOI]
-
104. Montoya-Villada S, Reyes-Vera E, Orozco J. Microwave technologies for biomedical diagnosis and therapy: Advances, challenges, and perspectives. Microchim Acta. 2026;193(4):218.[DOI]
-
105. Liu Y, Zhang Y, Li H, Hu TY. Recent advances in the bench-to-bedside translation of cancer nanomedicines. Acta Pharm Sin B. 2025;15(1):97-122.[DOI]
-
106. Lodi MB, Curreli N, Dachena C, Fedeli A, Scapaticci R, Randazzo A, et al. Feasibility analysis of theranostic magnetic scaffolds for microwave monitoring of hyperthermia treatment of bone tumors. IEEE J Electromagn RF Microw Med Biol. 2023;7(4):344-353.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite



