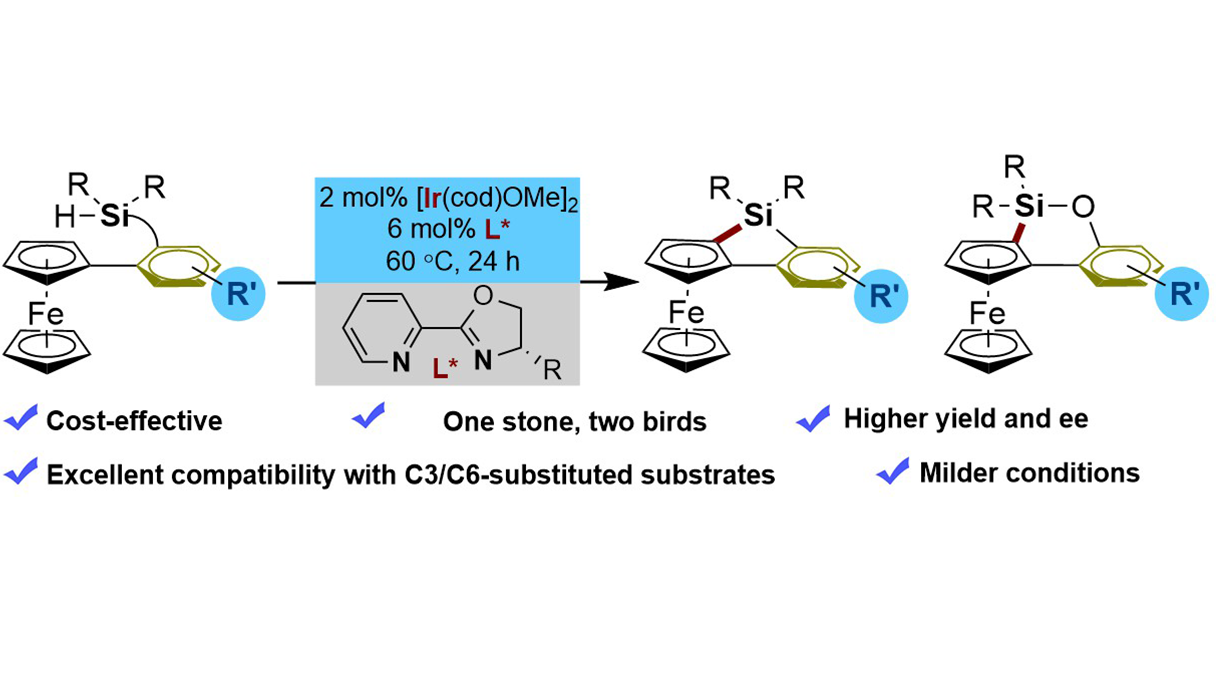
Abstract
We report the first Ir(I)-catalyzed enantioselective intramolecular C–H silylation of ferrocenes for the efficient construction of planar-chiral 5- and 6-membered ferrocenyl silacycles. This method overcomes key limitations of existing Rh(I)-based systems, offering superior reactivity, enhanced cost-effectiveness, and broader substrate scope, including challenging C3- and C6-substituted derivatives, as well as excellent enantiocontrol under mild conditions without requiring exogenous H2 acceptors. The utility of the obtained planar-chiral scaffolds is preliminarily demonstrated through the synthesis of novel monophosphine ligands, which exhibit promising activity in asymmetric catalysis.
Graphical Abstract
Keywords
1. Introduction
Planar-chiral ferrocene-based ligands and catalysts such as Mopf, Josiphos, Walphos, and Fc-Phox, have been widely utilized in asymmetric catalysis (Figure 1a)[1-9]. These compounds are classically formed by relying on enantioselective directed ortho-metalation (DoM), diastereoselective DoM, or chiral resolution[10,11]. In view of the step/atom-economies and functional group tolerance, enantioselective C–H bond functionalization offers significant advantages over traditional approaches and should be a highly desirable alternative for delivering planar chiral ferrocenes.
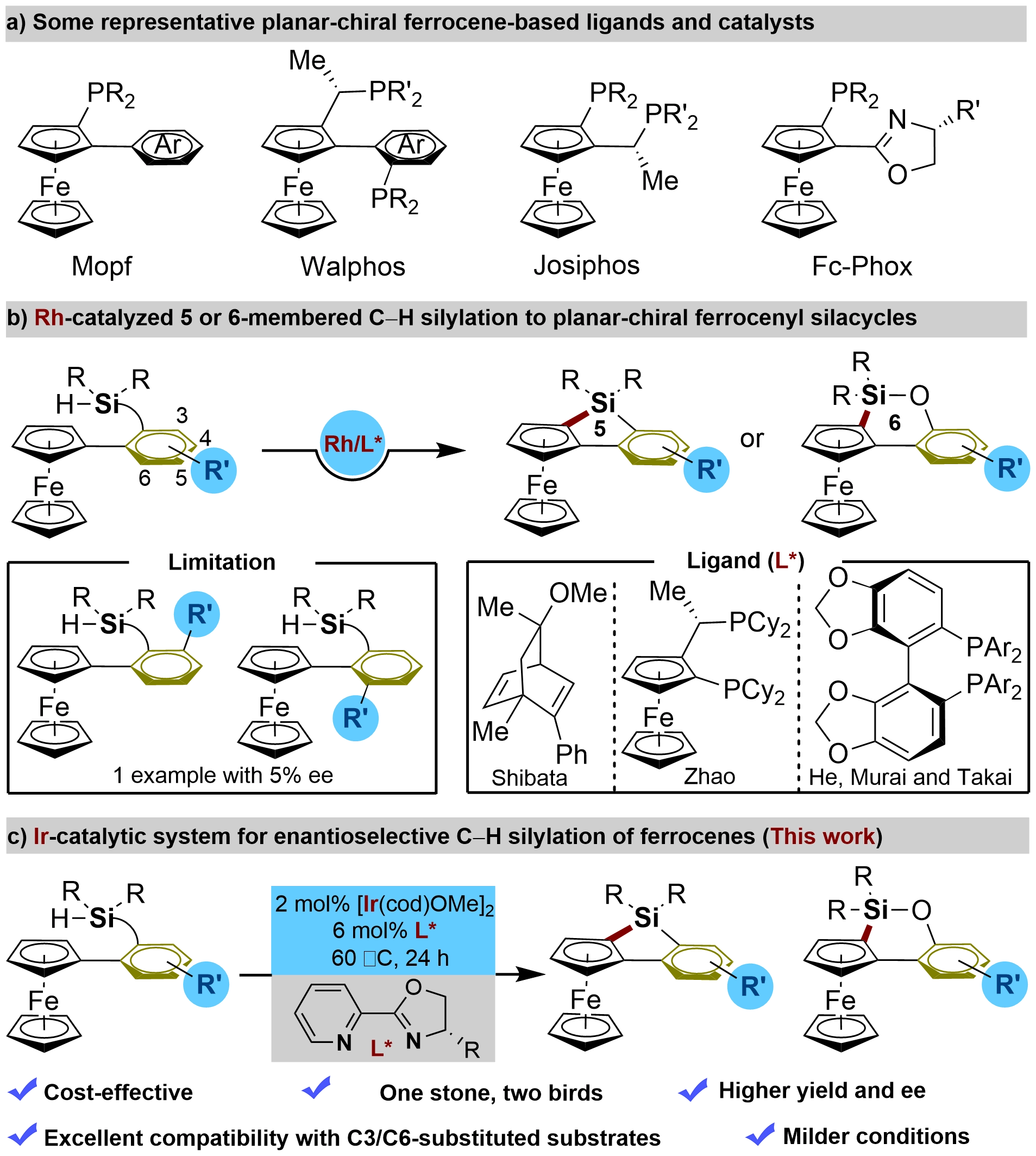
Figure 1. The limitations on the existing Rh(I)-catalytic systems for enantioselective 5-/6-membered C–H silylation of ferrocenes and our study on the first Ir-catalyzed asymmetric 5- and 6-membered C–H silylation of ferrocenes.
To date, some significant advancements have been achieved in enantioselective C–H bond functionalization of ferrocenes[12-30]. Among these, the enantioselective C–H silylation for the synthesis of planar chiral ferrocenyl-silacycles[31-35] attracted our attention due to the fundamental importance of silacycles[36-40] and our persistent interest in organosilicon chemistry[41-45]. Shibata[31], He[32], Murai[33] et al. independently developed three Rh(I)-catalytic systems for enantioselective 5-membered C–H silylation to construct planar-chiral ferrocene siloles (Figure 1a). Moreover, we recently accessed planar chiral ferrocene oxasilolanes via Rh(I)-catalyzed 6-membered C–H silylation (Figure 1b)[34]. However, despite impressive advances, these existing Rh(I)-catalytic systems for enantioselective C–H silylation of ferrocenes are far from satisfactory. There are still several shortcomings which remain to be addressed, including: 1) all of the systems relied on the use of Rh(I) complexes of chiral bisphosphine or diene ligands as catalysts, which not only resulted in high cost but also suffered from the difficulty of preparing and modifying the specific ligands; 2) enantioselective C–H silylation of substrates bearing a substituent group on the C3 or C6-position of the phenyl ring still remains an elusive challenge. In all cases, only Shibata’s study showed a single example, albeit with 5% ee; 3) these systems are usually conducted at high reaction temperatures and long reaction times; and 4) excess H2 acceptor and/or molecular sieve is essential to improve the yield and enantiomeric excess (ee) in Shibata’s and our systems. These problems have motivated efforts to discover a universal and inexpensive catalytic platform applicable to both enantioselective 5- and 6-membered C–H silylations of ferrocenes with improved efficiency, milder reaction conditions, and broader substrate scopes.
Besides the Rh catalytic system, Ir(I)-catalysis has also been demonstrated to enable C–H silylation. However, Ir-catalyzed enantioselective C–H silylation remains largely underdeveloped[46-48]. Recently, breakthroughs in the construction of central chirality via Ir-catalyzed C–H silylation have been made[49-53]. Inspired by those studies, we wondered whether Ir-catalyzed C–H silylation is applicable for introducing planar chirality, even though Ir-catalyzed C–H silylation of ferrocenes is unprecedented. Here we have developed an Ir(I)-catalytic system for both 5- and 6-membered enantioselective silylation of ferrocene C–H bonds to create planar-chiral ferrocenyl-silacycles (Figure 1c). In contrast to those existing Rh(I) catalytic systems, salient advantages of our study include: 1) enhanced cost-effectiveness (the use of an inexpensive and more accessible ligand, and no need for an H₂ acceptor), 2) one stone, two birds (it provides expedient access to both planar-chiral 5- and 6-membered ferrocenyl-silacycles under identical conditions), 3) excellent compatibility with C3/C6-substituted substrates, and 4) milder conditions, and higher yields and ee in most cases.
2. Experimental
2.1 Materials
All reactions were carried out under a nitrogen atmosphere in oven-dried glassware. Unless otherwise mentioned, all reagents were purchased from commercial sources and used without further purification. Solvents were purified according to the procedures described in Purification of Laboratory Chemicals. Flash column chromatography was performed on silica gel (particle size 200-300 mesh). Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker 400 MHz spectrometer and were calibrated using the residual solvent as an internal reference. Enantiomeric excess (ee) values of the products were determined by chiral high-performance liquid chromatography (HPLC) analysis using a Thermo U3000 instrument (n-hexane/iPrOH as eluent) with Daicel Chiralcel® OD-H, OD-3, IA-3, or IC-3 columns (column temperature 25 °C). Optical rotations were measured using an IP-digi300/2 apparatus with a sodium lamp (589 nm) at 25 °C. Melting points were determined on an X-4B melting point apparatus. High-resolution mass spectra were recorded on an IonSpec Fourier transform ion cyclotron resonance (FT-ICR) mass spectrometer or an Agilent 6520 Q-TOF LC-MS with an electrospray ionization (ESI) source. Absolute configurations were determined by comparison of specific rotations with literature data. Racemic compounds were prepared using (rac)-4-phenyl-2-(pyridin-2-yl)-4,5-dihydrooxazole as the ligand under the same reaction conditions.
2.2 Typical procedure for Iridium-catalyzed 5-membered enantioselective C–H silylation
In a N2-flushed glovebox, an oven-dried 8 mL tube equipped with a Teflon-coated magnetic stir bar was charged with [Ir(cod)OMe]2 (2.6 mg, 0.004 mmol, 2 mol%), chiral N,N-ligand (0.012 mmol, 6 mol%), and anhydrous 1,4-dioxane (0.5 mL). The resulting mixture was stirred in the glovebox at room temperature for 1 hour, followed by the addition of starting material 1 (0.2 mmol, 1 equiv) in 1,4-dioxane (0.5 mL). The reaction tube was capped, removed from the glovebox, and the reaction mixture was stirred at 60 °C for 24 h. Then, the reaction mixture was cooled to room temperature, concentrated under vacuum, and purified by silica gel column chromatography to afford planar chiral ferrocene silacycles 2. The ee value of the product was determined by chiral HPLC analysis. Chiral N,N-ligand L2 was used for the synthesis of planar chiral ferrocene silacycles 2a–l and 2u–w under the reaction conditions. On the other hand, chiral N,N-ligand L6 was used for the Ir-catalyzed enantioselective C–H silylation of C3- or C6-substituted substrates 1m–t. Product 2u was synthesized at 90 °C for 48 h. Product 2v was synthesized at 100 °C for 24 h.
2.3 Typical procedure for Iridium-catalyzed 6-membered enantioselective C–H silylation
In a N₂-flushed glovebox, an oven-dried 8 mL tube equipped with a Teflon-coated magnetic stir bar was charged with [Ir(cod)OMe]2 (2.6 mg, 0.004 mmol, 2 mol%), chiral N,N-ligand L2 or L6 (0.012 mmol, 6 mol%), and anhydrous 1,4-dioxane (0.5 mL). The resulting mixture was stirred in the glovebox at room temperature for 1 hour, followed by the addition of the starting material 3 (0.2 mmol, 1 equiv) in 1,4-dioxane (0.5 mL). The reaction tube was capped, removed from the glovebox, and the reaction mixture was stirred at 60 °C for 24 h. Then, the reaction mixture was cooled to room temperature, concentrated under vacuum, and subjected to silica gel column chromatography to afford planar chiral ferrocene silacycles 4. The ee value of the product was determined by chiral HPLC analysis. Ligand L2 was employed as the chiral ligand for the enantioselective 6-membered C–H silylation of substrates 3a–n as well as 3w–y. On the other hand, ligand L6 was utilized as the chiral ligand for the enantioselective 6-membered C–H silylation of C3- or C6-substituted substrates 3o–v.
3. Results and Discussion
3.1 Optimize the reaction conditions
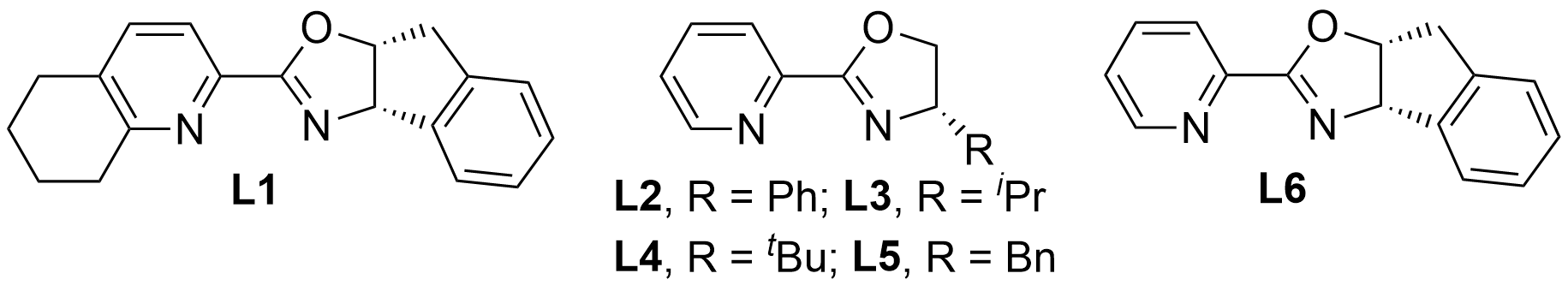
As mentioned above, Ir-catalyzed C–H silylation of Cp rings in ferrocenes remains unknown. Thus, in our preliminary experiment, we tested an Ir catalyst containing an achiral N,N-donor ligand that had been widely utilized in the C–H bond silylation of aromatic and alkyl groups[10] for the intramolecular silylation of ferrocene 1a. To our delight, we found that 5-membered C–H silylation of ferrocenes occurred smoothly to afford the corresponding product 2a in 81% yield by employing [Ir(cod)OMe]2 (2 mol%), Me4Phen (6 mol%), and norbornene (NBE) (hydrogen acceptor) in tetrahydrofuran (THF) at 60 °C for 24 h (Table 1, entry 1). Encouraged by this result, we rationalized that a combination of the proper Ir(I) precatalyst and chiral N,N-donor ligands could control the enantioselectivity of the 5-membered C–H silylation. We began the condition optimization by using hydrosilane 1a as a model substrate, which had also been used in the previous Rh catalytic systems, giving 89% yield and 87% ee of the planar chiral ferrocene 2a[5b]. In the preliminary experiment, hydrosilane 1a was treated with [Ir(cod)OMe]2 (2 mol%) and chiral N,N-donor ligand L1 (6 mol%) in 1,4-dioxane (1 mL) at 60 °C for 24 h. Fortunately, we exclusively obtained the corresponding dehydrogenative silylated product 2a in 96% yield and 95% ee (Table 1, entry 2). Then, we evaluated the influence of different chiral N,N-donor ligands. Chiral N,N-ligand L2 proved to be an excellent ligand for further improving the reactivity and the enantioselectivity (Table 1, entry 3). A change of ligand L2 to the other N,N-ligands L3–L6 resulted in a decrease in the level of enantioselectivity (entries 4–7). A brief optimization of the solvents demonstrated that dioxane was the best for this reaction (entries 8–10). The addition of NBE (1.2 equiv) as an H₂ acceptor was incapable of further improving the yield and ee (entry 11). Switching [Ir(cod)OMe]2 to [Ir(cod)Cl]2 led to lower yield and ee (entry 12). Attempts to shorten the reaction time to 12 h, decrease the reaction temperature to 40 °C, or reduce the amount of the ligand L2 from 6 mol% to 4.8 mol% led to incomplete conversion of 1a and low yields with maintenance of good enantioselectivity (Table 1, entries 13–15). The absolute configuration of 2a was assigned as Rp by comparison of the HPLC retention time with that reported in the literature. The absolute configurations of the other products were assigned by analogy to 2a.

| Entry | Ligand | Solvent | Yield [%]b | Ee [%]c |
| 1d | Me4Phen | THF | 81 | --- |
| 2 | L1 | dioxane | 96 | 95 |
| 3 | L2 | dioxane | 99 | 98 |
| 4 | L3 | dioxane | 93 | 73 |
| 5 | L4 | dioxane | 93 | 80 |
| 6 | L5 | dioxane | 96 | 61 |
| 7 | L6 | dioxane | 90 | 96 |
| 8 | L2 | THF | 95 | 93 |
| 9 | L2 | toluene | 92 | 91 |
| 10 | L2 | DCE | 83 | 82 |
| 11d | L2 | dioxane | 96 | 90 |
| 12e | L2 | dioxane | 82 | 89 |
| 13f | L2 | dioxane | 81 | 99 |
| 14g | L2 | dioxane | 68 | 99 |
| 15h | L2 | dioxane | 90 | 99 |
 | ||||
a: [Ir(cod)OMe]2 (2 mol%), chiral N,N-ligand (6 mol%), and 1a (0.1 mmol) were stirred at room temperature in solvent (1 mL) for 1 h, then the dehydrogentive silylation was performed at 60 °C for 24 h under N2 atmosphere; b: Yield of 2a was determined by NMR using TCE as internal standard; c: ee was determined by chiral HPLC; d: Norbornene (1.2 eq.) was used as the additive under the reaction; e: [Ir(cod)Cl]2 was used as the precatalyst; f: 12 h; g: 40 °C; h: Chiral ligand L2 (4.8 mol%) was used; THF: tetrahydrofuran; DCE: 1,2-dichloroethane.
3.2 Substrate scope
We then assessed the substrate scope of this 5-membered enantioselective silylation to deliver planar chiral ferrocene sioles. As depicted in Figure 2, various substituents, both electron-withdrawing (COOMe, CF3, F, Cl) and electron-donating (Me, OMe, OCF3), were well tolerated in the C4- or C5-position of the phenyl ring so that the planar chiral ferrocenyl-silacycles 2b–k were delivered in excellent yields and enantioselectivities (83-99% yields, 96-99% ee). Substrate 1l bearing 4, 5-dimethoxy groups on the phenyl ring was also successfully applied, affording the cyclized product 2l in 80% yield and 98% ee. Following the success, we aimed at the enantioselective silylation of the ferrocenes possessing a substituent on the C3 or C6-position of the phenyl ring, which could not be tolerated in previous Rh catalytic systems. We found that the use of the C3-substituted substrate 1m under the optimized condition led to incomplete consumption (85% yield) with 92% ee. To improve the reactivity of 2m, we carried out further screening of the chiral ligands. It revealed that the reaction of the substrate 1m occurred smoothly in 98% yield with 95% ee by employment of L6 as the chiral ligand in dioxane at 60 °C for 24 h. Under the modified conditions, a variety of substituents on the C3-position of the phenyl ring, regardless of their electronic properties, reacted smoothly to furnish the desired products 2m–o in high yields (93-98%) with excellent enantioselectivities (95-97% ee). Pleasingly, the enantioselective C–H silylation of the sterically hindered C6-substituted substrates also went smoothly under the modified conditions, affording the silylated products 2p–t with excellent yields and ee values. Actually, these sterically hindered C6-substituted substrates have never been investigated in the previous Rh-catalytic systems. Additionally, the effect of substituents on the silicon atom was also investigated. The switch of the methyl to an ethyl group led to the significant decrease of both yield and enantioselectivity (2u). The diphenyl substrate 1v achieved full conversion with 86% ee (2v) but the substrate with a diisopropyl group is unreactive. These results are consistent with the previously reported rhodium-catalyzed system. It suggests that steric hindrance on the silicon atom also plays a crucial role in determining both reactivity and enantioselectivity for this Ir-catalyzed conditions. However, how the steric effects of the substrate and ligands influence the reaction remains unclear. Elucidating the rate-limiting step and the origin of asymmetric induction will require detailed mechanistic studies. Ruthenocene was also reactive with 92% yield and 98% ee (2w). In general, compared with the best results obtained (yield and ee) in the previously reported Rh-catalyzed conditions, which have been presented in the parentheses of Figure 2, the current Ir-catalytic platform successfully overcomes the substrate limitations of Rh-catalyzed 5-membered enantioselective silylation, and delivers the planar chiral ferrocene sioles in higher yield and ee.
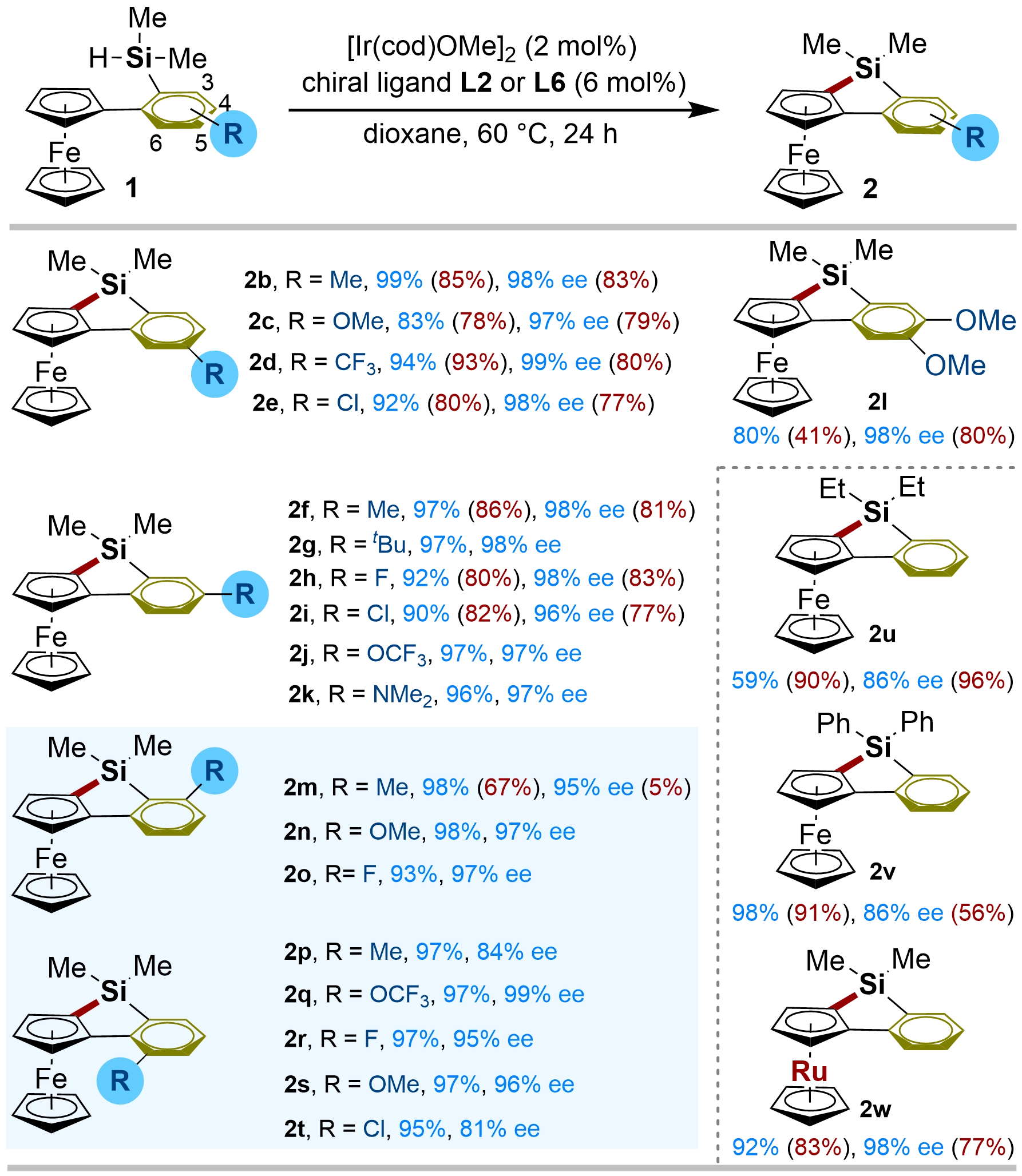
Figure 2. Substrate scope for Ir-catalyzed 5-membered enantioselective C–H silylation of ferrocenesa. a: [Ir(cod)OMe]2 (2 mol%), chiral ligand L2 or L6 (6 mol%), and 1 (0.2 mmol) were stirred at room temperature in dioxane (1 mL) for 1 h, then the silylation was performed at 60 °C for 24 h under N2 atmosphere. ee was determined by HPLC on a chiral stationary phase. Yields of 2 were determined by isolated products. The best results (yield and ee) obtained in the previously reported Rh-catalytic systems are given in parenthesis with red colour. HPLC: high-performance liquid chromatography.
Following the success of Ir-catalyzed enantioselective 5-membered C–H silylation of ferrocenes, we proceeded to examine whether such efficient Ir-catalytic systems are also suitable for enantioselective 6-membered silylation of ferrocene C–H bonds, thereby leading to planar-chiral ferrocene oxasilolanes with higher yields and enantiocontrol than those of the Rh-catalytic system. In fact, Ir-catalyzed enantioselective 6-membered silylation has never been achieved in the past. To our delight, with substrate 3a, the reaction performance was highly efficient under standard conditions, affording the corresponding product 4a in 93% yield with 98% ee. The substrate scope of the 6-membered C–H silylation of ferrocenes was then examined under the optimized conditions (Figure 3). In all cases, the iridium catalyst system exhibited much higher reactivity, efficiency, and enantiocontrol, as well as broader substrate tolerance than the previously reported Rh-catalytic system in most cases. Additionally, this iridium catalyst system no longer required an H₂ acceptor or molecular sieve. For instance, under standard conditions, the reaction of all C4- or C5-substituted ferrocene derivatives gave the desired cyclized products 4b–n in excellent yields with high ee values. Various functional groups, such as alkyl, phenyl, oxygen-based groups, and halides, were well tolerated under the reaction conditions. Varying the electronic properties of the substituent group at the C4- or C5-position did not significantly affect the yield or ee. Furthermore, as with the 5-membered enantioselective C–H silylation of ferrocenes, substrates containing a range of groups at the C3- or C6-position of the phenyl ring (3o–v), which were rarely studied in the previous Rh-catalytic system, also proved to be highly reactive with excellent enantioselectivities in this protocol when the optimized ligand L6 was used. Additionally, the cyclized products 4w–x were obtained in good yields and ee when 3,4-disubstituted substrates bearing an electron-withdrawing (3w) or electron-rich (3x) group were subjected to this reaction. We also tested the reactivity of the 2-ruthenocenyl phenol-derived silyl ether 3y to expand the versatility of our catalytic system. The reaction proceeded uneventfully to yield product 4y in 90% yield with 97% ee. For comparison, the reaction occurred in 71% yield with 93% ee under the Rh-catalytic conditions. Notably, the absolute configuration of 4a was assumed to be Rp by comparison of the optical rotation with the literature. Other products were assigned by analogy.
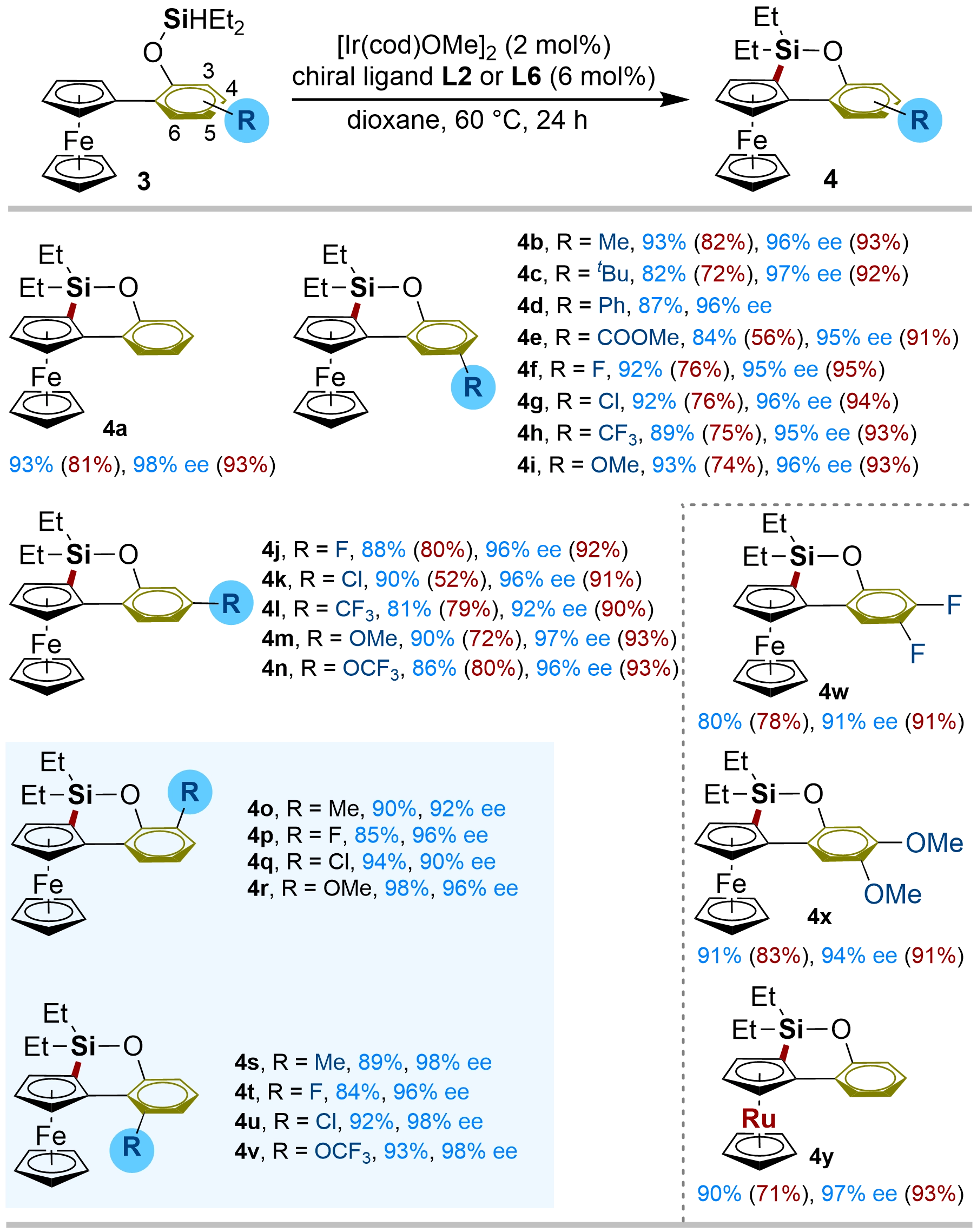
Figure 3. Substrate scope for Ir-catalyzed 6-membered enantioselective C–H silylation of ferrocenesa. a: [Ir(cod)OMe]2 (2 mol%), chiral ligand L2 or L6 (6 mol%), and 3 (0.2 mmol) were stirred at room temperature in dioxane (1 mL) for 1 h, then the C–H silylation was performed at 60 °C for 24 h under N2 atmosphere. ee was determined by HPLC on a chiral stationary phase. Yields of 4 were determined by isolated products. The best results (yield and ee) obtained in the previously reported Rh-catalytic systems are given in parenthesis with red colour. HPLC: high-performance liquid chromatography.
3.3 Synthetic utility
To test the practicality of our method, we first carried out both the Ir-catalyzed enantioselective 5- and 6-membered C–H silylation of 1a and 3a on a large scale (5 mmol). The reactions gave the desired planar-chiral ferrocenes 2a or 4a, respectively, with maintained yields and enantioselectivities (Figure 4a). As a further demonstration of the utility of our methodology, the planar-chiral monophosphine ligands FcL1 and FcL2 were prepared from product 4a obtained in the current study. As illustrated in Figure 4b, product 4a was first converted into the corresponding planar-chiral ferrocenyl-phenols 5a and 5b through a nucleophilic ring opening reaction of PhLi or the Hiyama–Denmark cross-coupling of iodobenzene, which further underwent –OH to –PPh2 group transformation to accomplish the planar-chiral monophosphine ligands FcL1 and FcL2. Notably, the drop of ee in the Hiyama–Denmark cross-coupling of (Rp)-4a results from the competitive reaction of the protonation and sequential directed C–H bond arylation, which was confirmed in our previous study[34]. A preliminary examination of FcL1 and FcL2 in a palladium-catalyzed asymmetric hydrosilylation of styrene[54,55] disclosed that this type of P-ligand is highly efficient, although the enantioselectivity needs further improvement (Figure 4c). Additionally, we proved that the ligands FcL1 and FcL2 also showed catalytic activity and produced chiral induction in Ir-catalyzed asymmetric ring-opening of oxabenzonorbornadiene with 1-phenylpiperazine (Figure 4c)[56]. These preliminary results demonstrate that the planar-chiral scaffolds of ferrocenyl-silacycles are promising for developing a new class of chiral monophosphine ligands. Further investigations on the development and application of the new planar chiral ferrocene-based monophosphine ligands in asymmetric catalysis are ongoing.
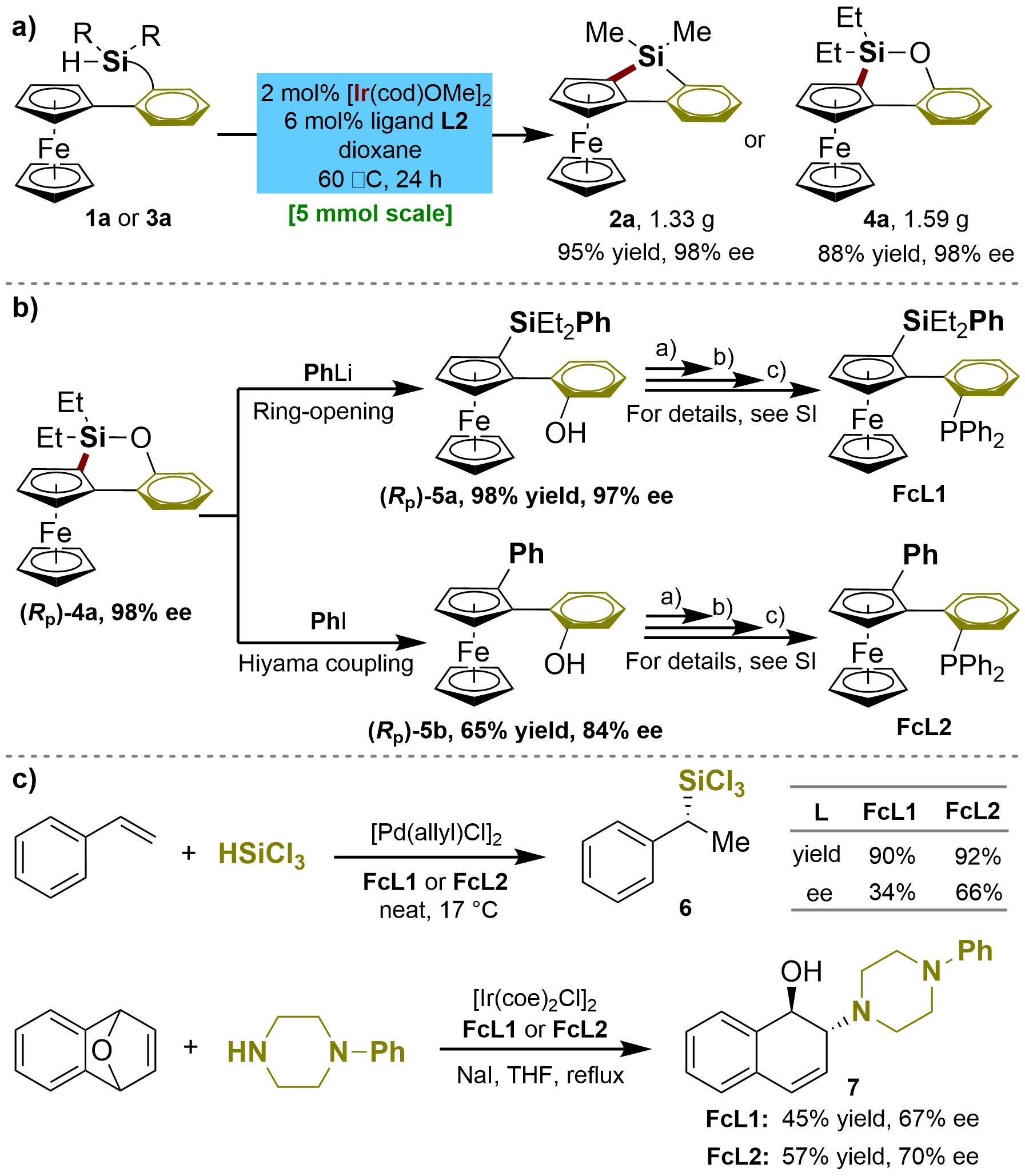
Figure 4. Large-scale synthesis and applications of the planar-chiral ferrocene 4a in development of novel chiral phosphine ligands for asymmetric catalysis.
4. Conclusion
In summary, we have addressed some of the limitations of existing Rh(I)-catalyzed enantioselective silylation of ferrocene C–H bonds by developing an Ir-catalytic platform that is also applicable to C3/C6-substituted ferrocenes. The use of cost-effective chiral pyridinyloxazoline ligands, the absence of a requirement for an H₂ acceptor or molecular sieve, broader substrate tolerance, and good compatibility with both 5- and 6-membered C–H silylation suggest that this Ir-catalytic system offers certain advantages over previously reported Rh-catalytic conditions in the enantioselective C–H silylation of ferrocenes.
Supplementary materials
The supplementary material for this article is available at: Supplementary materials.
Authors contribution
Gao F, Sun, H, Zhang, H: Investigation, writing-review & editing.
Zhao D: Conceptualization, writing-original draft, writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
We are grateful for the financial support from the National Natural Science Foundation of China (Grant Nos. 22221002 and 22371132), the Natural Science Foundation of Tianjin (23JCJQJC00130), the “Frontiers Science Center for New Organic Matter”, Nankai University (Grant No. 63181206), Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (Grant No. JYB2025XDXM410).
Copyright
© The Author(s) 2026.
References
-
1. Fu GC. Enantioselective nucleophilic catalysis with “planar-chiral” heterocycles. Acc Chem Res. 2000;33(6):412-420.[DOI]
-
2. Dai LX, Tu T, You SL, Deng WP, Hou XL. Asymmetric catalysis with chiral ferrocene ligands. Acc Chem Res. 2003;36(9):659-667.[DOI]
-
3. Blaser HU, Brieden W, Pugin B, Spindler F, Studer M, Togni A. Solvias josiphos ligands: From discovery to technical applications. Top Catal. 2002;19(1):3-16.[DOI]
-
5. Štěpnička P. Ferrocenes: ligands, materials and biomolecules. New York: John Wiley & Sons; 2008.[DOI]
-
6. Dai LX, Hou XL. Chiral ferrocenes in asymmetric catalysis: Synthesis and applications. New York: John Wiley & Sons; 2010.[DOI]
-
7. Phillips ES. Ferrocenes: Compounds, Properties and Applications. Hauppauge: Nova Science Publishers; 2011.
-
9. Drusan M, Šebesta R. Enantioselective C–C and C–heteroatom bond forming reactions using chiral ferrocene catalysts. Tetrahedron. 2014;70(4):759-786.[DOI]
-
10. Ferber B, Kagan H. Metallocene sulfoxides as precursors of metallocenes with planar chirality. Adv Synth Catal. 2007;349(4-5):493-507.[DOI]
-
11. Schaarschmidt D, Lang H. Selective syntheses of planar-chiral ferrocenes. Organometallics. 2013;32(20):5668-5704.[DOI]
-
12. Zhu DY, Chen P, Xia JB. Synthesis of planar chiral ferrocenes by transition-metal-catalyzed enantioselective C–H activation. ChemCatChem. 2016;8(1):68-73.[DOI]
-
14. Liu CX, Gu Q, You SL. Asymmetric C–H bond functionalization of ferrocenes: New opportunities and challenges. Trends Chem. 2020;2(8):737-749.[DOI]
-
15. Zhang ZZ, Huang DY, Shi BF. Recent advances in the synthesis of ferrocene derivatives via 3d transition metal-catalyzed C–H functionalization. Org Biomol Chem. 2022;20(20):4061-4073.
-
16. Qi H, Hang NN, Ming J. Synthesis of planar chiral ferrocenes by catalytic desymmetrization reactions. ChemCatChem. 2024;16(19):e202400736.[DOI]
-
17. Gao DW, Shi YC, Gu Q, Zhao ZL, You SL. Enantioselective synthesis of planar chiral ferrocenes via palladium-catalyzed direct coupling with arylboronic acids. J Am Chem Soc. 2013;135(1):86-89.
-
20. Zhou L, Cheng HG, Li L, Wu K, Hou J, Jiao C, et al. Synthesis of planar chiral ferrocenes via enantioselective remote C–H activation. Nat Chem. 2023;15(6):815-823.[DOI]
-
22. Liu H, Deng Y, Ling D, Chen L, Jin Z. Asymmetric catalysis for the synthesis of planar chiral ferrocene derivatives. Chin Chem Lett. 2026;37(3):111793.[DOI]
-
23. Huang FR, Zhang P, Yao QJ, Shi BF. Construction of planar chiral ferrocenes by cobalt-catalyzed enantioselective C–H acyloxylation enabled by dual ligands. CCS Chem. 2024;6(11):2783-2793.[DOI]
-
27. Zhang ZZ, Zhou G, Yue Q, Yao QJ, Shi BF. Copper/BINOL-catalyzed enantioselective C–H functionalization toward planar chiral ferrocenes under mild conditions. ACS Catal. 2024;14(6):4030-4039.[DOI]
-
29. Zhao F, Li Y, Dong Z, Liu CX, Wang Q, Gu Q, et al. Accessing planar chiral ferrocenes via transient directing group-enabled C–H alkenylation under Pd(II) catalysis. Chem Catal. 2025;5(11):101485.[DOI]
-
30. Liu CX, Zhao F, Feng Z, Wang Q, Gu Q, You SL. Kinetic resolution of planar chiral metallocenes using Rh-catalysed enantioselective C–H arylation. Nat Synth. 2023;2(1):49-57.[DOI]
-
34. Zhao WT, Lu ZQ, Zheng H, Xue XS, Zhao D. Rhodium-catalyzed 2-arylphenol-derived six-membered silacyclization: Straightforward access toward dibenzooxasilines and silicon-containing planar chiral metallocenes. ACS Catal. 2018;8(9):7997-8005.[DOI]
-
39. Li L, Zhang Y, Gao L, Song Z. Recent advances in C–Si bond activation via a direct transition metal insertion. Tetrahedron Lett. 2015;56(12):1466-1473.[DOI]
-
40. Xu LW, Huang WS, Wang Q, Yang H. State-of-the-art advances in enantioselective transition-metal-mediated reactions of silacyclobutanes. Synthesis. 2022;54(24):5400-5408.[DOI]
-
44. Zhang J, Pan D, Zhang HX, Yan N, Xue XS, Zhao D. Reversing site-selectivity in formal cross-dimerization of benzocyclobutenones and silacyclobutanes. CCS Chem. 2023;5(8):1753-1762.[DOI]
-
45. Zhang H, Zhao D. Synthesis of silicon-stereogenic silanols involving iridium-catalyzed enantioselective C–H silylation leading to a new ligand scaffold. ACS Catal. 2021;11(17):10748-10753.[DOI]
-
46. Ge Y, Huang X, Ke J, He C. Transition-metal-catalyzed enantioselective C–H silylation. Chem Catal. 2022;2(11):2898-2928.[DOI]
-
47. Zheng L, Nie XX, Wu Y, Wang P. Construction of Si-stereogenic silanes through C–H activation approach. Eur J Org Chem. 2021;2021(44):6006-6014.[DOI]
-
48. Li L, Huang WS, Xu Z, Xu LW. Catalytic asymmetric silicon-carbon bond-forming transformations based on Si–H functionalization. Sci China Chem. 2023;66(6):1654-1687.[DOI]
-
53. Suto T, La C, Hartwig JF. Iridium-catalyzed, regio- and stereoselective silylation of primary and secondary C(sp3)–H bonds in primary amines. J Am Chem Soc. 2025;147(42):37940-37948.[DOI]
-
55. Ji W, Wu HH, Zhang J. Axially chiral biaryl monophosphine oxides enabled by palladium/WJ-phos-catalyzed asymmetric suzuki-miyaura cross-coupling. ACS Catal. 2020;10(2):1548-1554.[DOI]
-
56. Yang D, Long Y, Zhang J, Zeng H, Wang S, Li C. Iridium-catalyzed asymmetric ring-opening reactions of oxabenzonorbornadienes with amine nucleophiles. Organometallics. 2010;29(16):3477-3480.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite