All Articles
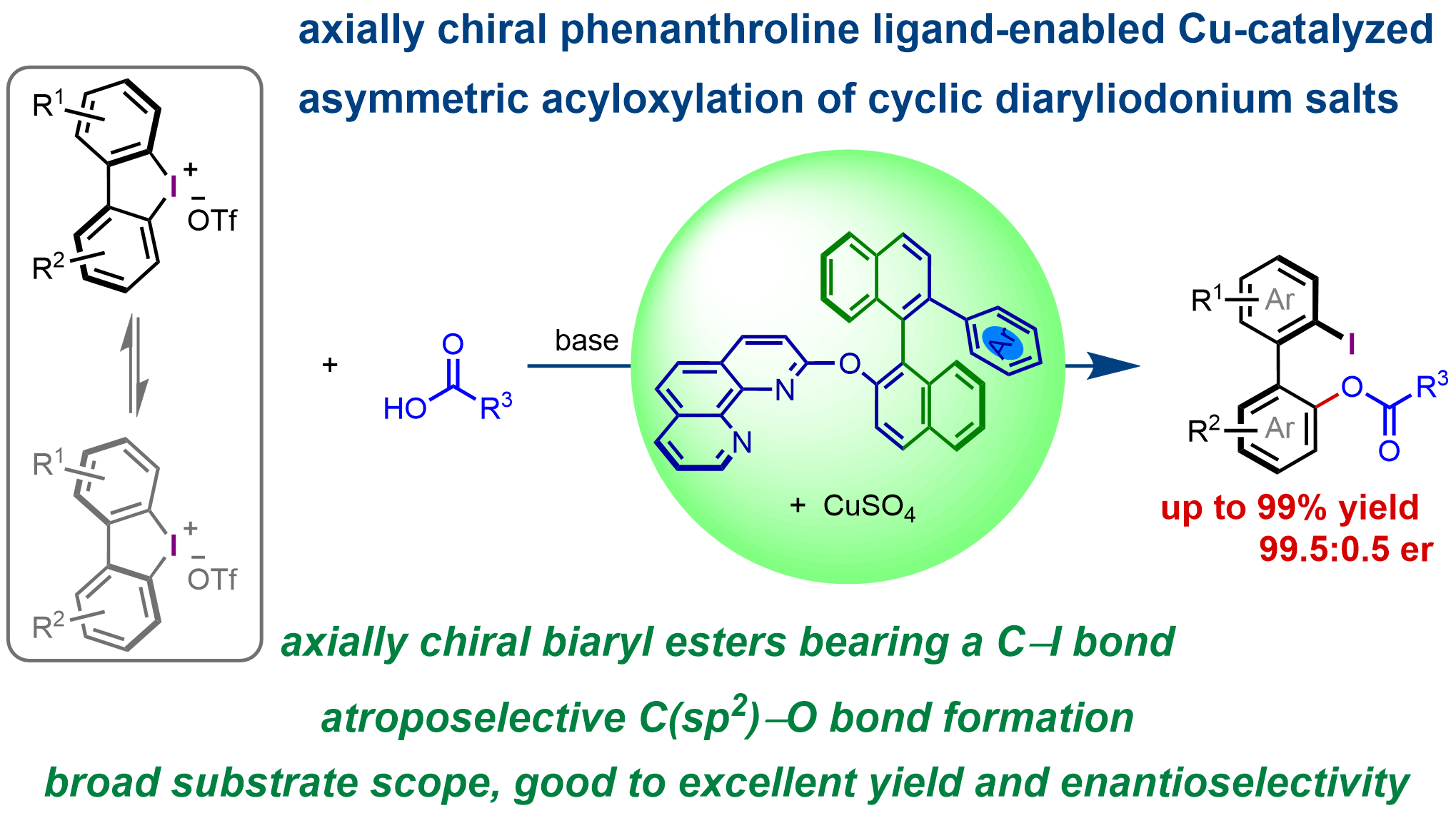
Axially chiral phenanthroline ligand-enabled Cu-catalyzed asymmetric acyloxylation of cyclic biaryliodonium salts
-
Chiral ligands play a crucial role in enantioselective transition-metal catalysis, not only by coordinating to metal centers and modulating their electronic properties, but also by providing the essential chiral environment for efficient enantiodiscrimination. ...
MoreChiral ligands play a crucial role in enantioselective transition-metal catalysis, not only by coordinating to metal centers and modulating their electronic properties, but also by providing the essential chiral environment for efficient enantiodiscrimination. For a given reaction, high enantioselectivities can often be achieved with different chiral ligands, offering valuable enantioinduction models, even when these ligands possess distinct structures and chiral environments. Herein, we report that a class of conformationally flexible C1-symmetric axially chiral phenanthroline ligands enables the formation of chiral copper catalysts capable of promoting the highly enantioselective ring-opening acyloxylation of cyclic biaryliodonium salts. Using this approach, a range of valuable axially chiral biaryl compounds bearing an ester group and a carbon–iodine bond were synthesized in good to excellent yield (up to 99%), with good to excellent enantioselectivity (up to 99.5:0.5 er).
Less -
Shouyi Cen, ... Zhipeng Zhang
-
DOI: https://doi.org/10.70401/cc.2026.0023 - April 17, 2026
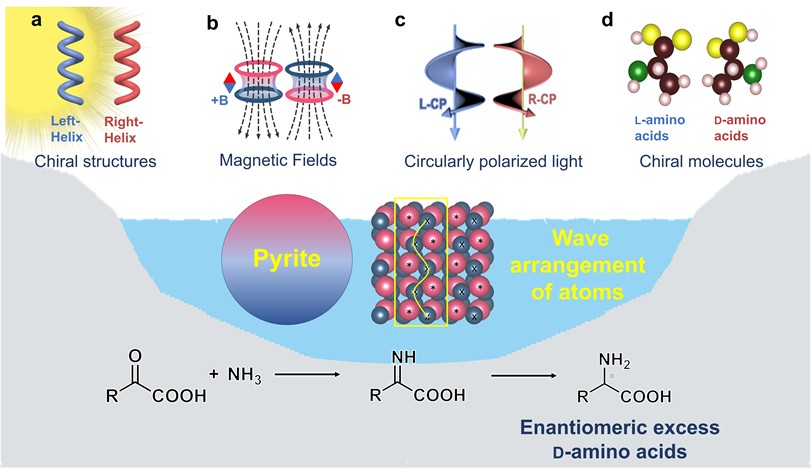
Pyrite interference in prebiotic chiral selection of amino acids
-
L-amino acids are ubiquitous in biology, yet the origin of their homochirality is an enduring mystery. Prebiotic chiral selection process of amino acids could significantly influence subsequent molecular evolution and the emergence of life. However, the ...
MoreL-amino acids are ubiquitous in biology, yet the origin of their homochirality is an enduring mystery. Prebiotic chiral selection process of amino acids could significantly influence subsequent molecular evolution and the emergence of life. However, the distribution of chirality of amino acids remains convoluted, which poses obstacles to the in-depth study of prebiotic chiral selection of amino acids. Here, we propose that minerals in the prebiotic environment might have hindered the absolute selection of L-amino acids to induce a random distribution of L- and D-amino acids during the early stages of life’s evolution. Pyrite, abundant in the prebiotic world, predominantly selected D-amino acids in the photocatalytic reductive amination of α-keto acids, resisting the enantioselectivity of chiral driving forces like chiral structures, magnetic fields, circularly polarized light, and chiral molecules. This resistance is due to the wavy atomic arrangement of pyrite surfaces, which causes larger reaction energy differences between enantiomers. Our findings provide evidence to assess the validity of prebiotic chiral selection scenarios for biomolecules, an important step in understanding the origin of life.
Less -
Ruiqi Li, ... Yuxi Fang
-
DOI: https://doi.org/10.70401/cc.2026.0021 - April 10, 2026
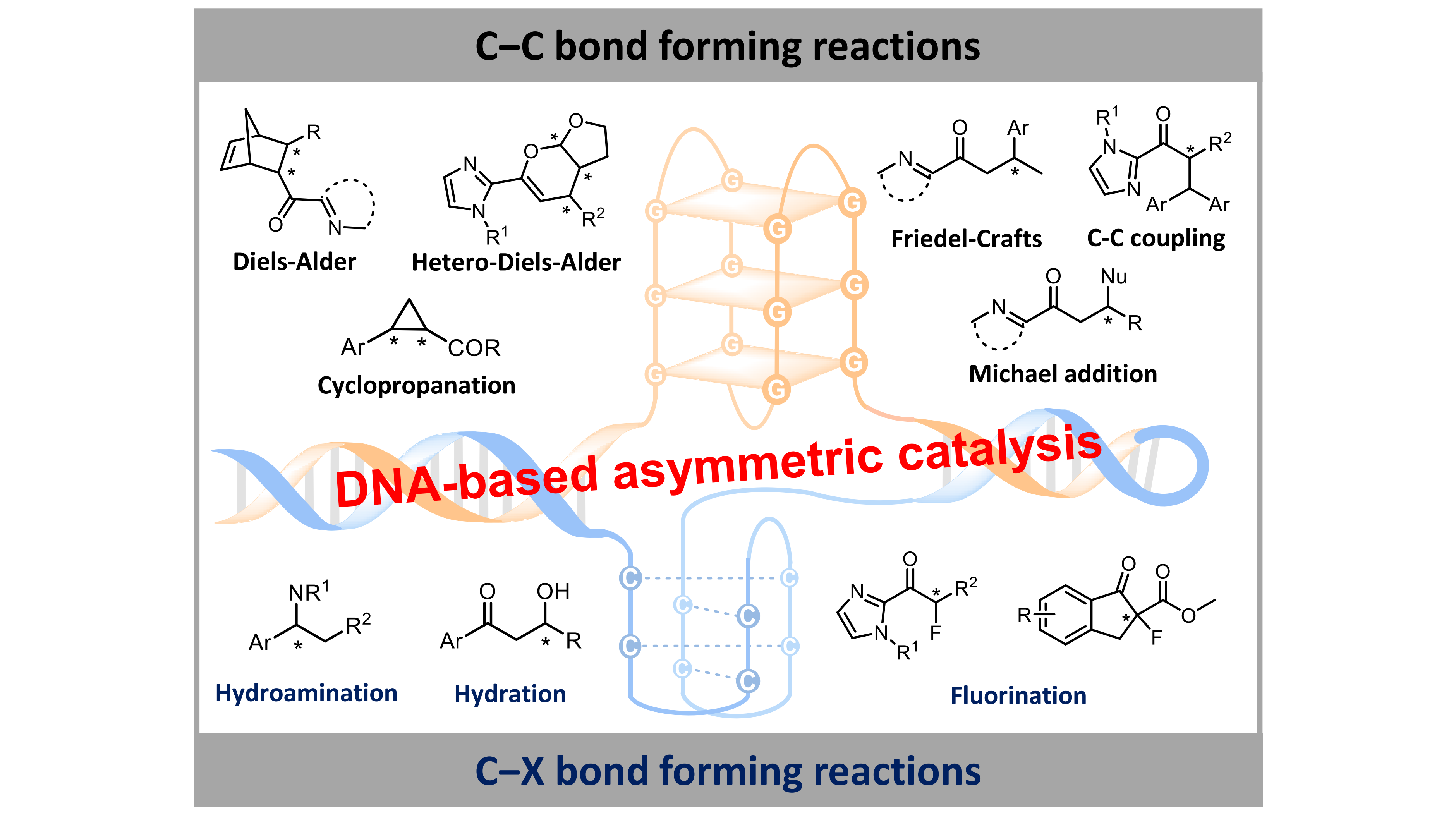
Programmable DNA as stereocontrol ligand for asymmetric catalysis in aqueous media
-
Owing to its intrinsic chirality, programmability and structural diversity, DNA functions as an effective chiral ligand for asymmetric catalysis in water. DNA hybrid catalysts are constructed by anchoring metal species into DNA scaffolds through covalent ...
MoreOwing to its intrinsic chirality, programmability and structural diversity, DNA functions as an effective chiral ligand for asymmetric catalysis in water. DNA hybrid catalysts are constructed by anchoring metal species into DNA scaffolds through covalent or noncovalent strategies. These DNA-based catalytic systems exhibit potent reactivity and high enantioselectivity in various C–C, C–N, C–O, and C–F bond formation reactions in aqueous media. Importantly, their catalytic activity and product stereochemistry can be precisely directed by modulating DNA sequences, tertiary structures, metal species, and host-guest interaction. This review highlights recent advances in DNA-mediated asymmetric catalysis and discusses future prospects toward tailored stereocontrol under aqueous conditions.
Less -
Shanmei Xu, ... Changhao Wang
-
DOI: https://doi.org/10.70401/cc.2026.0022 - April 10, 2026
Enantiopure chiral multilayer polymers – design, asymmetric synthesis and property study
-
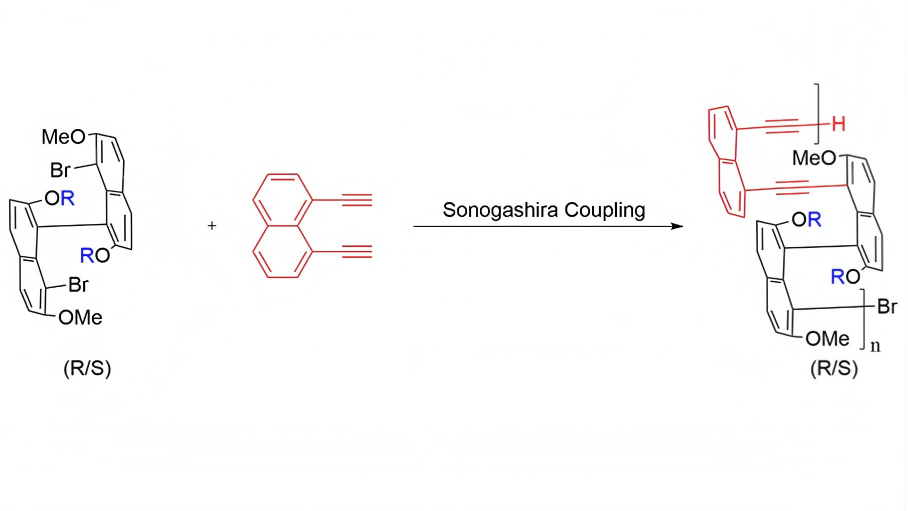
Axially chiral binaphthyl scaffolds provide a robust platform for encoding permanent stereochemistry into conjugated polymer backbones while promoting nonplanar organization and higher-order aggregation. Building on our group’s earlier development ...
MoreAxially chiral binaphthyl scaffolds provide a robust platform for encoding permanent stereochemistry into conjugated polymer backbones while promoting nonplanar organization and higher-order aggregation. Building on our group’s earlier development of multilayer 3D folding polymers and related achiral/chiral multilayer polymer series, we designed and synthesized the first enantiopure chiral multilayer polymers from enantiopure binaphthyl-derived monomers via CuI-assisted, Pd-catalyzed Sonogashira polycondensation. To probe structure–property relationships, the monomer library was designed to contrast flexible alkoxy-substituted systems with a conformationally constrained tethered analogue. The resulting polymers were obtained in moderate yields, with number-average molecular weights on the order of 103-104 g mol-1 and relatively narrow dispersities. UV–visible spectra showed the expected aromatic π-π* absorptions. Photoluminescence measurements in tetrahydrofuran (THF)/H2O mixtures displayed a nonmonotonic solvent-composition dependence, with emission enhancement at intermediate water fractions followed by quenching in more water-rich mixtures. Dynamic light scattering indicated the presence of light-scattering dispersed aggregates in CHCl3 under the tested conditions, while scanning electron microscopy revealed hierarchical agglomerates in the dried state, in which submicrometer particulates coalesced into micrometer-scale clusters. Together, these results show that axial stereochemistry and aggregation/packing behavior cooperatively influence radiative and nonradiative decay pathways in chiral multilayer backbones, providing useful design guidance for aggregation-responsive emissive polymer materials.
Less -
Hao Liu, ... Guigen Li
-
DOI: https://doi.org/10.70401/cc.2026.0020 - April 08, 2026
Iridium(I)-catalyzed enantioselective five- and six-membered C–H silylation of ferrocenes
-
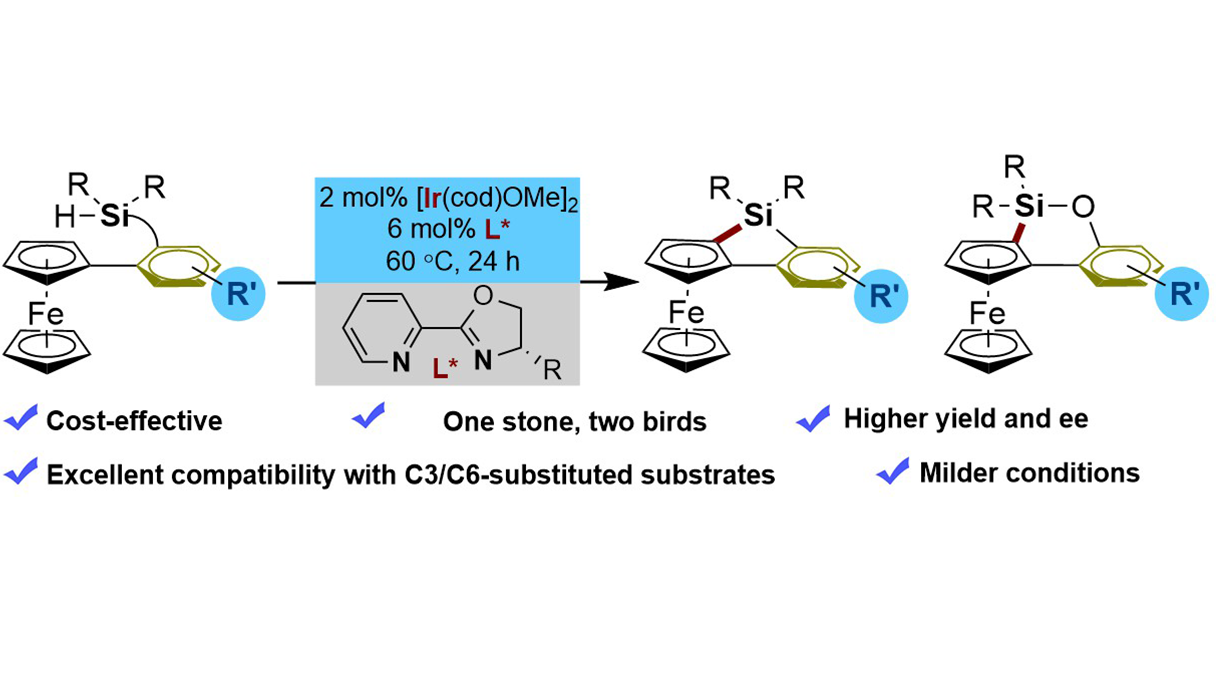
We report the first Ir(I)-catalyzed enantioselective intramolecular C–H silylation of ferrocenes for the efficient construction of planar-chiral 5- and 6-membered ferrocenyl silacycles. This method overcomes key limitations of existing Rh(I)-based ...
MoreWe report the first Ir(I)-catalyzed enantioselective intramolecular C–H silylation of ferrocenes for the efficient construction of planar-chiral 5- and 6-membered ferrocenyl silacycles. This method overcomes key limitations of existing Rh(I)-based systems, offering superior reactivity, enhanced cost-effectiveness, and broader substrate scope, including challenging C3- and C6-substituted derivatives, as well as excellent enantiocontrol under mild conditions without requiring exogenous H2 acceptors. The utility of the obtained planar-chiral scaffolds is preliminarily demonstrated through the synthesis of novel monophosphine ligands, which exhibit promising activity in asymmetric catalysis.
Less -
Fang Gao, ... Dongbing Zhao
-
DOI: https://doi.org/10.70401/cc.2026.0019 - April 07, 2026
Catalytic atroposelective synthesis of C–N axially chiral 5-pyrazolyl indoles via multicomponent tandem cycloaddition
-
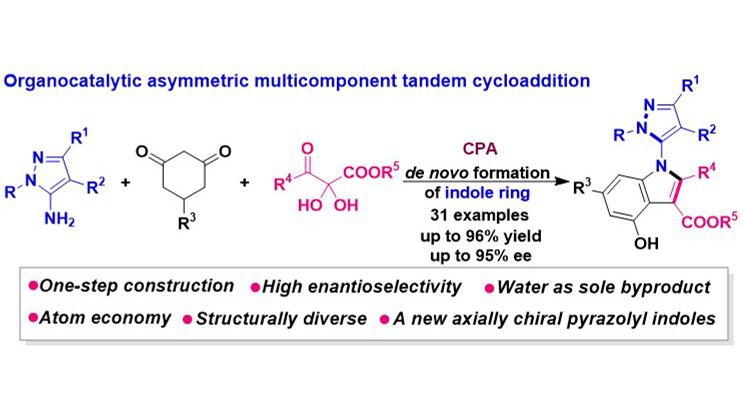
An organocatalytic asymmetric multicomponent tandem cycloaddition has been established for the construction of C–N axially chiral 5-pyrazolyl indole frameworks, which represent a novel class of five-five-membered indole scaffolds. Utilizing 5-aminopyrazoles, ...
MoreAn organocatalytic asymmetric multicomponent tandem cycloaddition has been established for the construction of C–N axially chiral 5-pyrazolyl indole frameworks, which represent a novel class of five-five-membered indole scaffolds. Utilizing 5-aminopyrazoles, cyclohexanediones, and 2,3-diketoester precursors, this methodology constructs the indole ring de novo in a single step, demonstrating high atom- and step-economy with water as the exclusive byproduct. The protocol enabled the efficient and high-enantioselective synthesis of structurally diverse C–N axially chiral 5-pyrazolyl indoles. This achievement not only introduces a new member to the family of axially chiral pyrazolyl indoles but also provides an efficient and novel strategy for their construction.
Less -
Yu-Yu Chen, ... Feng Shi
-
DOI: https://doi.org/10.70401/cc.2026.0018 - April 02, 2026
Enantio- and diastereoselective NHC/Cu-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters
-
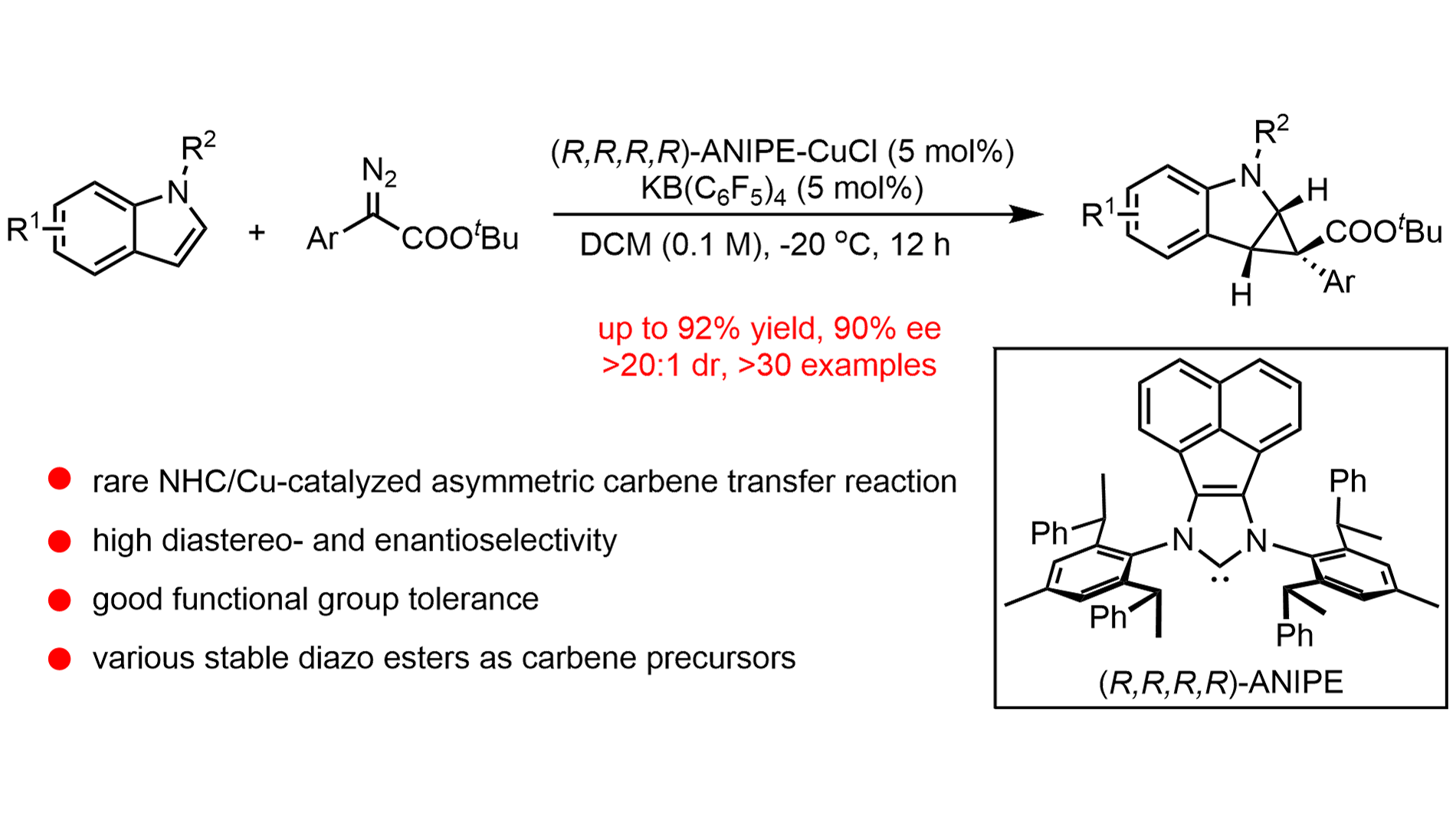
We report a highly enantioselective N-heterocyclic carbene (NHC)/copper-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters. This protocol enables the efficient construction of cyclopropane-fused indolines featuring ...
MoreWe report a highly enantioselective N-heterocyclic carbene (NHC)/copper-catalyzed intermolecular dearomative cyclopropanation of indoles with diazo esters. This protocol enables the efficient construction of cyclopropane-fused indolines featuring quaternary stereogenic centers under mild conditions. A broad range of substituted indoles and diazo esters is tolerated, delivering the corresponding products in high yields with enantioselectivities of up to 90% ee. Notably, the employment of a bulky chiral NHC ligand is crucial for achieving effective stereocontrol over the challenging quasi-linear copper–carbenoid intermediate. Furthermore, gram-scale reactions and downstream transformations demonstrate the synthetic utility of this methodology. Mechanistic investigations, including kinetic isotope effect and Hammett studies, support a reaction pathway involving nucleophilic attack of the indole on a copper–carbenoid species. This earth-abundant copper catalytic system serves as a sustainable alternative to traditional noble-metal-based asymmetric carbene-transfer reactions.
Less -
Jiawei Lu, ... Shi-Liang Shi
-
DOI: https://doi.org/10.70401/cc.2026.0017 - March 24, 2026
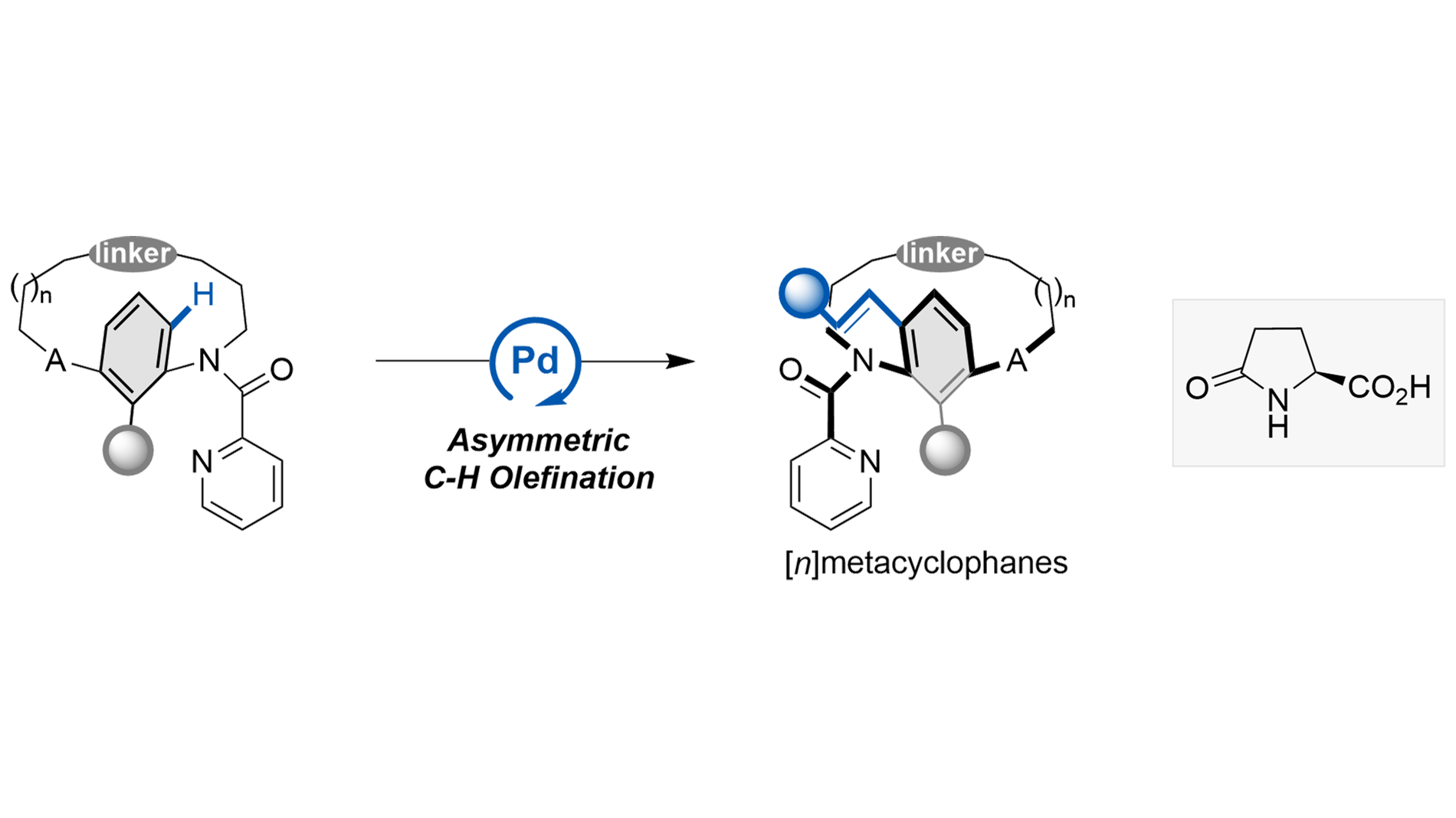
Pd(II)-catalyzed atroposelective and enantioselective C−H olefination of [n]metacyclophanes
-
A Pd(II)-catalyzed C–H olefination strategy has been established for the enantioselective synthesis of [n]metacyclophanes. The method demonstrates excellent functional group tolerance across a series of olefins and metacyclophane precursors ...
MoreA Pd(II)-catalyzed C–H olefination strategy has been established for the enantioselective synthesis of [n]metacyclophanes. The method demonstrates excellent functional group tolerance across a series of olefins and metacyclophane precursors with varying ansa-chain lengths, achieving high stereocontrol through kinetic or dynamic kinetic resolution. Racemization experiments were conducted to investigate the relationship between molecular structure and conformational stability. Furthermore, the synthetic utility of this approach was demonstrated through gram-scale synthesis and the application of a resulting bifunctional thiourea catalyst in asymmetric Michael additions, highlighting its potential in asymmetric synthesis.
Less -
Xin-En Yan, ... Changgui Zhao
-
DOI: https://doi.org/10.70401/cc.2026.0016 - March 24, 2026
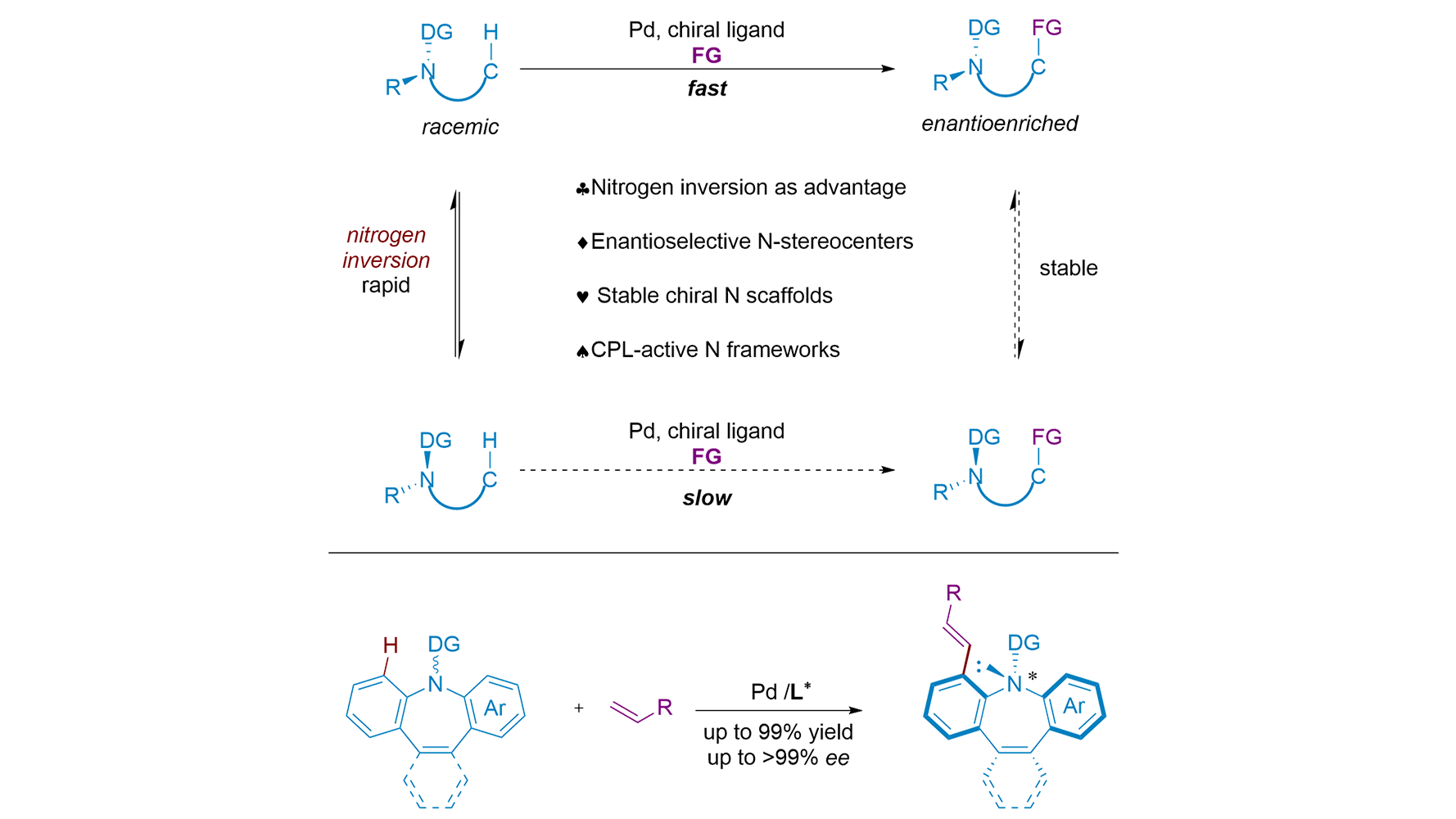
Harnessing molecular motion for asymmetry – nitrogen inversion as a springboard for stereoselective C–H functionalization
-
The asymmetric construction of nitrogen stereocenters is notoriously difficult due to rapid nitrogen inversion. Zhang and co-workers now showcase a Pd-catalyzed enantioselective C-H activation strategy that turns this inversion into an advantage, delivering ...
MoreThe asymmetric construction of nitrogen stereocenters is notoriously difficult due to rapid nitrogen inversion. Zhang and co-workers now showcase a Pd-catalyzed enantioselective C-H activation strategy that turns this inversion into an advantage, delivering stable chiral azepines with high enantioselectivity. Coincidentally, almost at the same time, Shi and co-workers reported a similar C-H alkylation reaction. Beyond methodology, the products serve as promising scaffolds for asymmetric catalysis and chiroptical materials, bridging synthesis and function. This Perspective discusses how their works not only provide a powerful synthetic method but also open a new avenue for constructing heteroatom stereocenters by harnessing, rather than suppressing, molecular dynamics. Here, nitrogen inversion is deliberately exploited as a dynamic feature that enables enantioselective C-H functionalization, rather than being suppressed as a stereochemical liability.
Less -
Chun-Yan Guan, Guang-Jian Mei
-
DOI: https://doi.org/10.70401/cc.2026.0015 - March 11, 2026
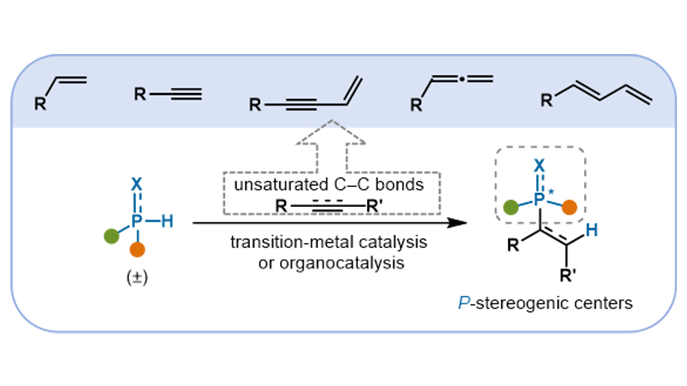
Catalytic construction of P-stereogenic centers through asymmetric hydrophosphination of unsaturated C–C bonds
-
P-Stereogenic centers are important structural motifs prevalent in natural products, bioactive molecules, and high-performance ligands. Their presence confers significant value across medicinal chemistry, materials science, and asymmetric ...
MoreP-Stereogenic centers are important structural motifs prevalent in natural products, bioactive molecules, and high-performance ligands. Their presence confers significant value across medicinal chemistry, materials science, and asymmetric catalysis. In recent years, catalytic asymmetric hydrophosphination has emerged as a powerful and efficient strategy for constructing such P-stereogenic compounds. Distinguished by high atom economy, broad substrate compatibility, and excellent stereocontrol under mild conditions, these transformations align closely with the principles of sustainable and green synthesis. This review summarizes recent advances in the catalytic asymmetric hydrophosphination of unsaturated C–C bonds for the synthesis of P-stereogenic centers. It covers substrates including alkenes bearing electron-withdrawing groups, alkynes, as well as specialized systems such as enynes, allenes, and conjugated dienes. Emphasis is placed on the design of catalytic systems, encompassing transition-metal catalysts (e.g., Pd, Ni, Cu, Co, Mn) and organocatalysts, along with their mechanisms. Current challenges, such as the low reactivity of unactivated or sterically hindered substrates and difficulties in achieving stereochemical differentiation between phosphorus substituents, are also discussed. This review aims to provide a reference for further innovation and methodological development in the synthesis of P-stereogenic molecules.
Less -
Bing-Lin Wang, ... Xiao-Hui Yang
-
DOI: https://doi.org/10.70401/cc.2026.0014 - March 10, 2026
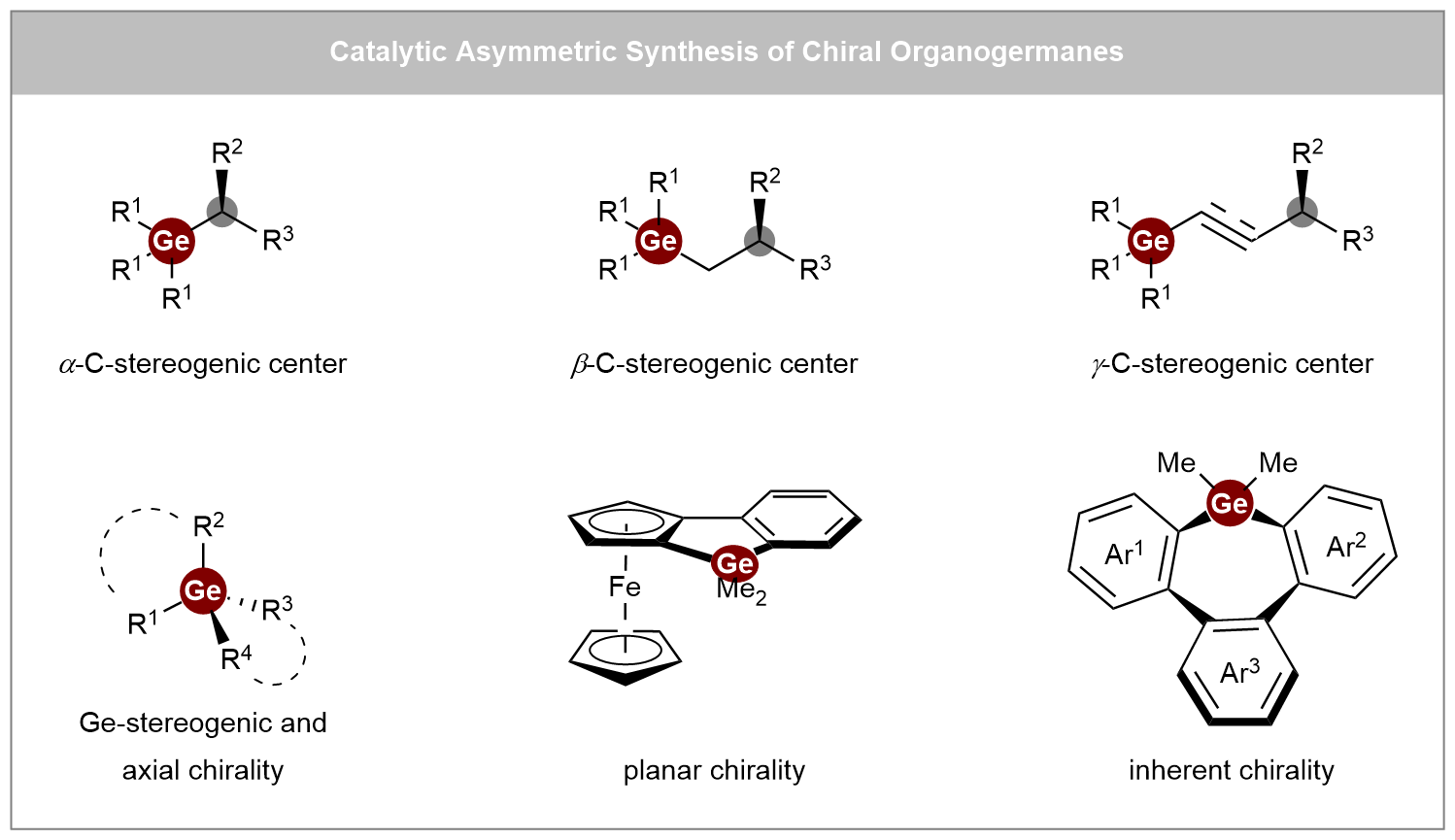
Recent advances in catalytic asymmetric synthesis of chiral organogermanes
-
Chiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent ...
MoreChiral organogermanes hold great potential as bioisosteres in medicinal chemistry and functional materials, yet their development has long been hindered by a scarcity of efficient synthetic strategies. This review offers a comprehensive overview of recent advances in the catalytic asymmetric synthesis of chiral organogermanes, highlighting a shift from traditional resolution methods toward asymmetric catalytic approaches. The content is organized into three main categories: (i) synthesis of C-stereogenic germanes, (ii) synthesis of Ge-stereogenic germanes, and (iii) synthesis of other chiral germanes, including planar, inherent, and axially chiral types. Key synthetic methodologies are systematically examined, such as enantioselective alkene hydrofunctionalization, carbene insertion, coupling reactions, and [2+2+2] cycloaddition, utilizing a variety of catalytic systems ranging from transition metals (Rh, Cu, Ni, Co) and Lewis acids to engineered metalloenzymes. Particular emphasis is placed on the mechanistic insights and ligand design principles that enable stereochemical control in these transformations. We hope this review will inspire chemists working in related areas and contribute to the future advancement of this field.
Less -
Shao-Wu Liu, ... Chuan He
-
DOI: https://doi.org/10.70401/cc.2026.0013 - February 13, 2026
Enantioselectivity synthesis of isoquinolin-1-one derivatives with C–N axial chirality via cobalt-catalyzed oxidative formal (4+2) cycloaddition: Light or not
-
Developing mild and sustainable strategies for the synthesis of complex molecules is a pivotal yet challenging goal in modern synthesis. While cobalt catalysis offers a sustainable alternative to noble metals, achieving such transformations at room temperature ...
MoreDeveloping mild and sustainable strategies for the synthesis of complex molecules is a pivotal yet challenging goal in modern synthesis. While cobalt catalysis offers a sustainable alternative to noble metals, achieving such transformations at room temperature remains elusive. Herein, we report a cobalt-catalyzed aerobic oxidative asymmetric formal cycloaddition involving C(sp2)–H bond activation of benzamides with unactivated alkynes at room temperature using air as the oxidant. For challenging low-activity substrates, reaction efficiency is enhanced via a photoinduced catalytic cycle. Mechanistic and substrate scope studies indicate that substrate reactivity is influenced by electronic and steric effects.
Less -
Liang-Neng Wang, ... Liang-Qiu Lu
-
DOI: https://doi.org/10.70401/cc.2026.0012 - January 28, 2026
Asymmetric synthesis of mechanically planar chiral rotaxanes via organocatalyzed enantioselective desymmetrization
-
Xiao-Hua Zhou, ... Wei Wang
-
DOI: https://doi.org/10.70401/cc.2026.0011 - January 22, 2026
Development of a scalable iridium-catalyzed asymmetric hydrogenation process for synthesis of chiral 2-methyl-1,2,3,4-tetrahydroquinoline
-
Asymmetric hydrogenation is an efficient tool for rapid synthesis of a diverse range of chiral compounds with high yields and excellent enantioselectivities. Chiral 2-methyl-1,2,3,4-tetrahydro-quinoline is a valuable building block for organic synthesis ...
MoreAsymmetric hydrogenation is an efficient tool for rapid synthesis of a diverse range of chiral compounds with high yields and excellent enantioselectivities. Chiral 2-methyl-1,2,3,4-tetrahydro-quinoline is a valuable building block for organic synthesis and has been widely used in the synthesis of bioactive molecules and chiral ligands. Herein, we report an improved iridium-catalyzed asymmetric hydrogenation of 2-methylquinoline on a hundred-gram scale for efficient synthesis of chiral 2-methyl-1,2,3,4-tetrahydroquinoline with up to 91.4% ee value and an 80,000 turnover number. Optically pure 2-methyl-1,2,3,4-tetrahydroquinoline could be obtained in high yields through recrystallization or chemical resolution with the tartaric acid derivative (L)-DMTA.
Less -
Huan Jing, ... Yong-Gui Zhou
-
DOI: https://doi.org/10.70401/cc.2026.0010 - January 08, 2026
Patterned cholesteric liquid crystal polymer network film with precisely controlled structural colors prepared using a handedness invertible photochromic cholesteric liquid crystal mixture
-
Cholesteric liquid crystal polymer network (CLCN) patterns composed of oppositely handed helical structures are attractive for anti-counterfeiting. Different areas can be observed under the circularly polarized light with opposite handedness. However, ...
MoreCholesteric liquid crystal polymer network (CLCN) patterns composed of oppositely handed helical structures are attractive for anti-counterfeiting. Different areas can be observed under the circularly polarized light with opposite handedness. However, achieving precise control over the reflection band wavelengths of CLCN patterns using photochromic cholesteric liquid crystals (CLCs) remains a significant challenge. Herein, we synthesized a chiral cyanostilbene derivative that demonstrated significant modulation of its helical twisting power upon 365 nm ultraviolet (UV) irradiation. When incorporated into CLC mixtures, this additive enabled precise control over both the handedness and reflection wavelength of the resulting CLCN patterns within seconds under UV exposure. The system allowed for the preparation of colorful CLCN patterns through in-situ photopolymerization. These findings demonstrate that the developed CLC mixtures are well-suited for high-throughput fabrication of CLCN patterns on industrial coating lines.
Less -
Xingchen Liu, ... Yonggang Yang
-
DOI: https://doi.org/10.70401/cc.2025.0009 - December 31, 2025
Catalytic asymmetric dearomative formal [4+2] annulation of indoles with O-Silylated hemiaminals as dienes: The dual role of chiral phosphoric acid
-
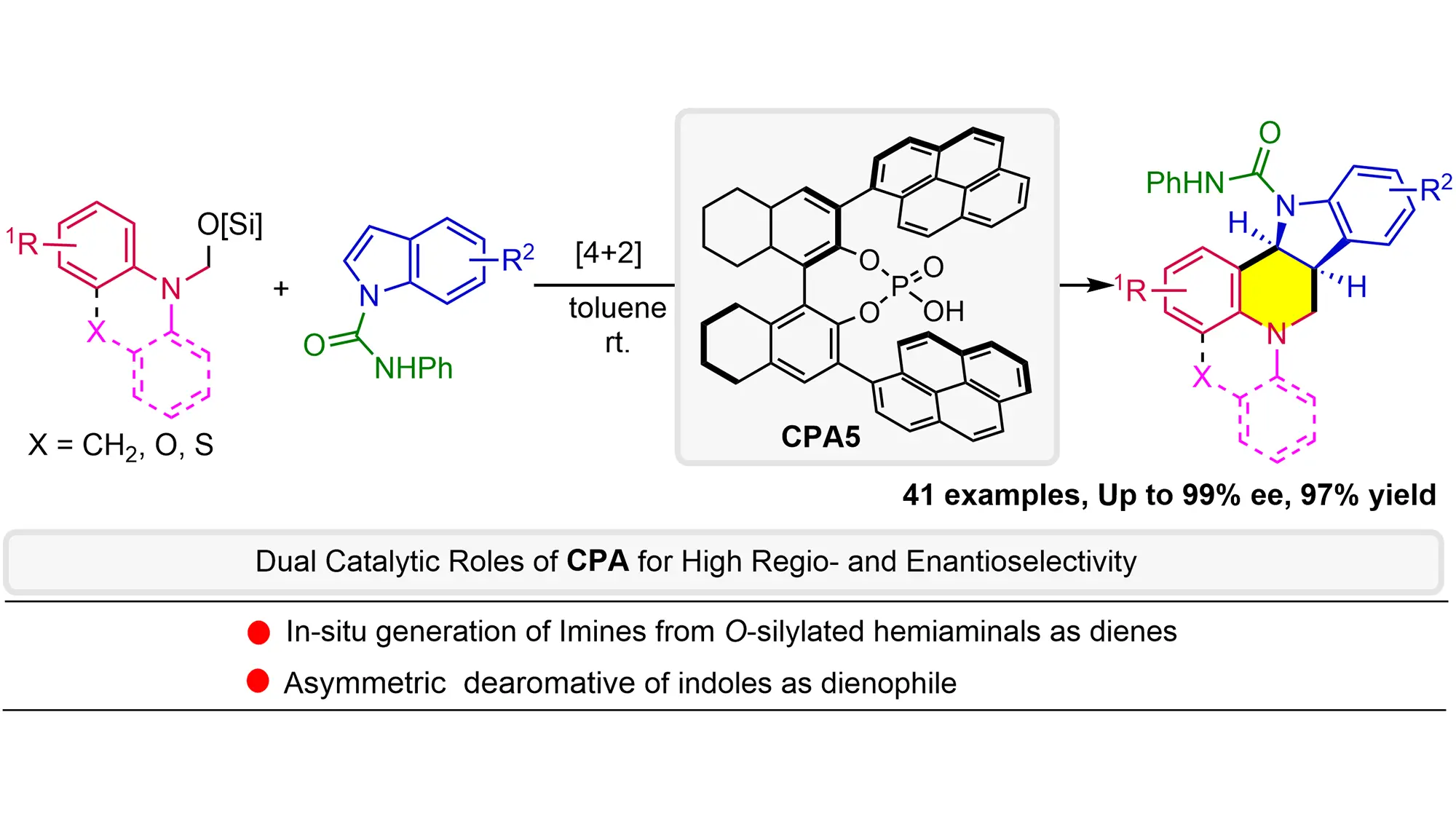
O-Silylated hemiaminals are utilized as elegant imine precursors in the formal asymmetric [4+2] annulation of indoles for the first time, wherein chiral phosphoric acid (CPA) acts (1) as a Brønsted acid catalyst to facilitate methanimine formation ...
MoreO-Silylated hemiaminals are utilized as elegant imine precursors in the formal asymmetric [4+2] annulation of indoles for the first time, wherein chiral phosphoric acid (CPA) acts (1) as a Brønsted acid catalyst to facilitate methanimine formation under mild conditions and then (2) as an anion-binding catalyst for dearomative annulation. This methodology exhibits a broad substrate scope with remarkable functional group tolerance and enantioselectivity (up to 97% yield and 99% ee), providing straightforward access to the challenging indoline-fused tetrahydroquinolines bearing multiple stereogenic centers. Mechanistic studies reveal the critical role of PhNHCO- groups in enhancing both reactivity and enantioselectivity, probably due to non-covalent interactions with CPA. The kinetic isotope effects experiment and negative linear Hammett correlation suggest a concerted process.
Less -
Nan-Fang Mo, ... Zheng-Hui Guan
-
DOI: https://doi.org/10.70401/cc.2025.0008 - December 26, 2025
Transition metal-catalyzed remote asymmetric C–H activation of arenes
-
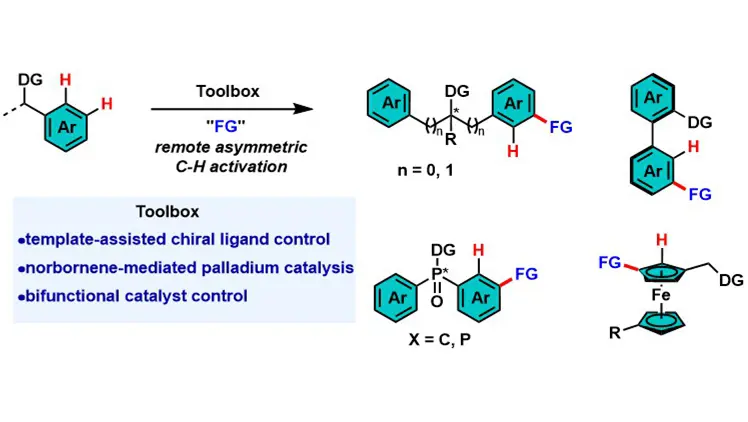
Transition metal-catalyzed asymmetric C–H activation is vital for chiral molecule synthesis but faces challenges in remote C–H functionalization due to traditional metallacycle constraints and difficulties in long-range chiral recognition. This review ...
MoreTransition metal-catalyzed asymmetric C–H activation is vital for chiral molecule synthesis but faces challenges in remote C–H functionalization due to traditional metallacycle constraints and difficulties in long-range chiral recognition. This review summarizes three core strategies to address these issues: template-assisted chiral ligand control, norbornene-mediated palladium catalysis, and bifunctional catalyst control. These strategies achieve high enantioselectivity for diverse chiral architectures. Future directions include expanding to para-C–H bonds of arenes and aliphatic C–H bonds, developing robust chiral mediators/ligands, and applying the methodology to natural products and complex materials.
Less -
Lili Chen, Senmiao Xu
-
DOI: https://doi.org/10.70401/cc.2025.0006 - December 16, 2025
NHCs-catalyzed enantioselective synthesis of biaryl axially chiral imides
-
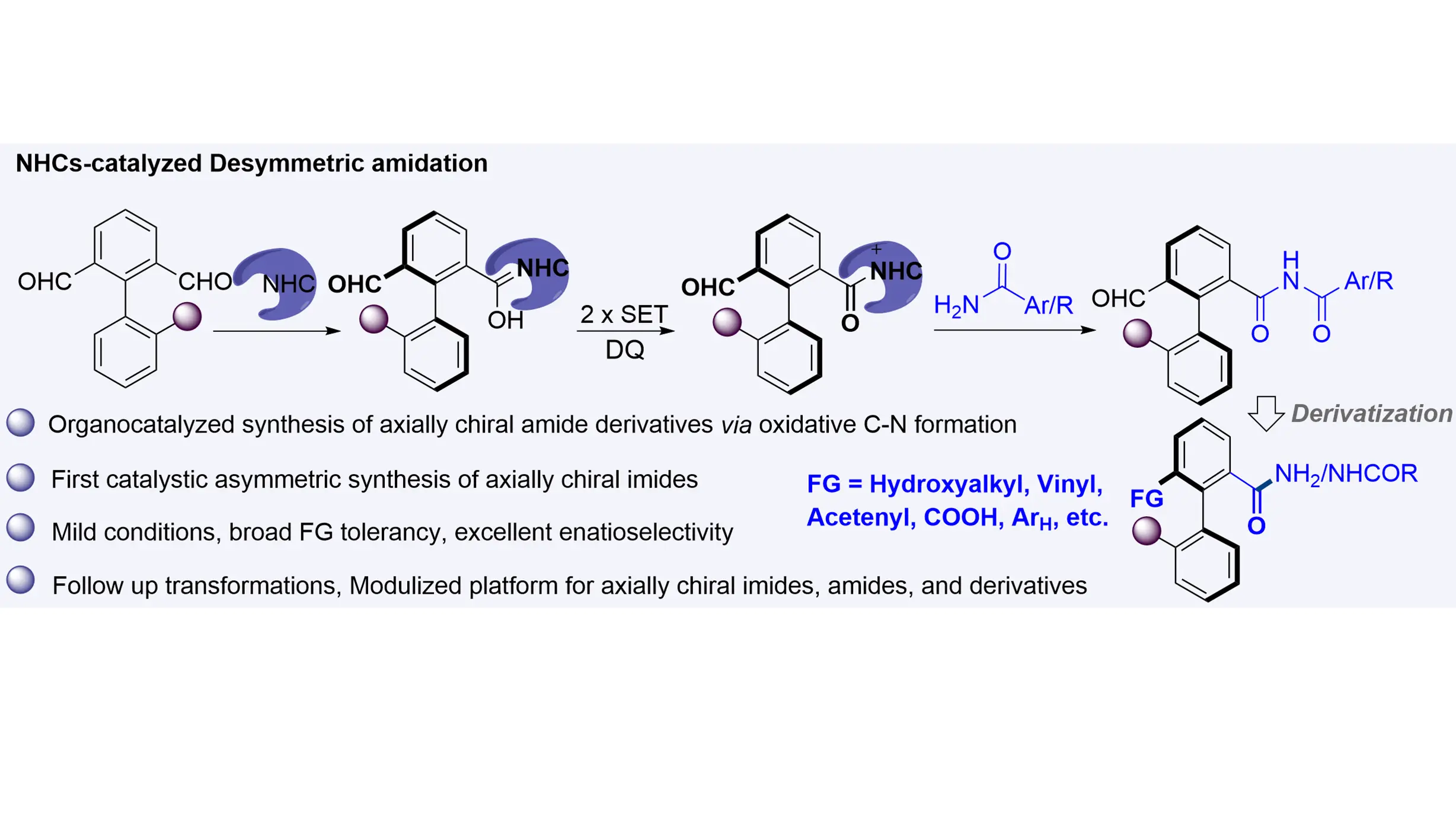
The synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic ...
MoreThe synthesis of biaryl axially chiral amides and their derivatives—compounds that have shown promise as additives or catalysts in asymmetric catalysis—has traditionally relied on transition-metal catalysts. Herein, we report an NHC-catalyzed organocatalytic atropoenantioselective amidation between axially prochiral biaryl dialdehydes and amides that efficiently affords axially chiral imides. This method operates under metal-free and mild conditions, exhibits broad functional group tolerance and substrate scope, and delivers products with excellent enantioselectivities. Furthermore, a wide variety of axially chiral imides, amides, and related derivatives can be accessed through enantio-retentive transformations, offering a versatile and attractive strategy for their synthesis.
Less -
Yingtao Wu, ... Qian Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0005 - December 15, 2025
Enantioselective aza-Mislow-Evans rearrangement through S-allenylation of sulfenamides with alkynyl carbenes
-
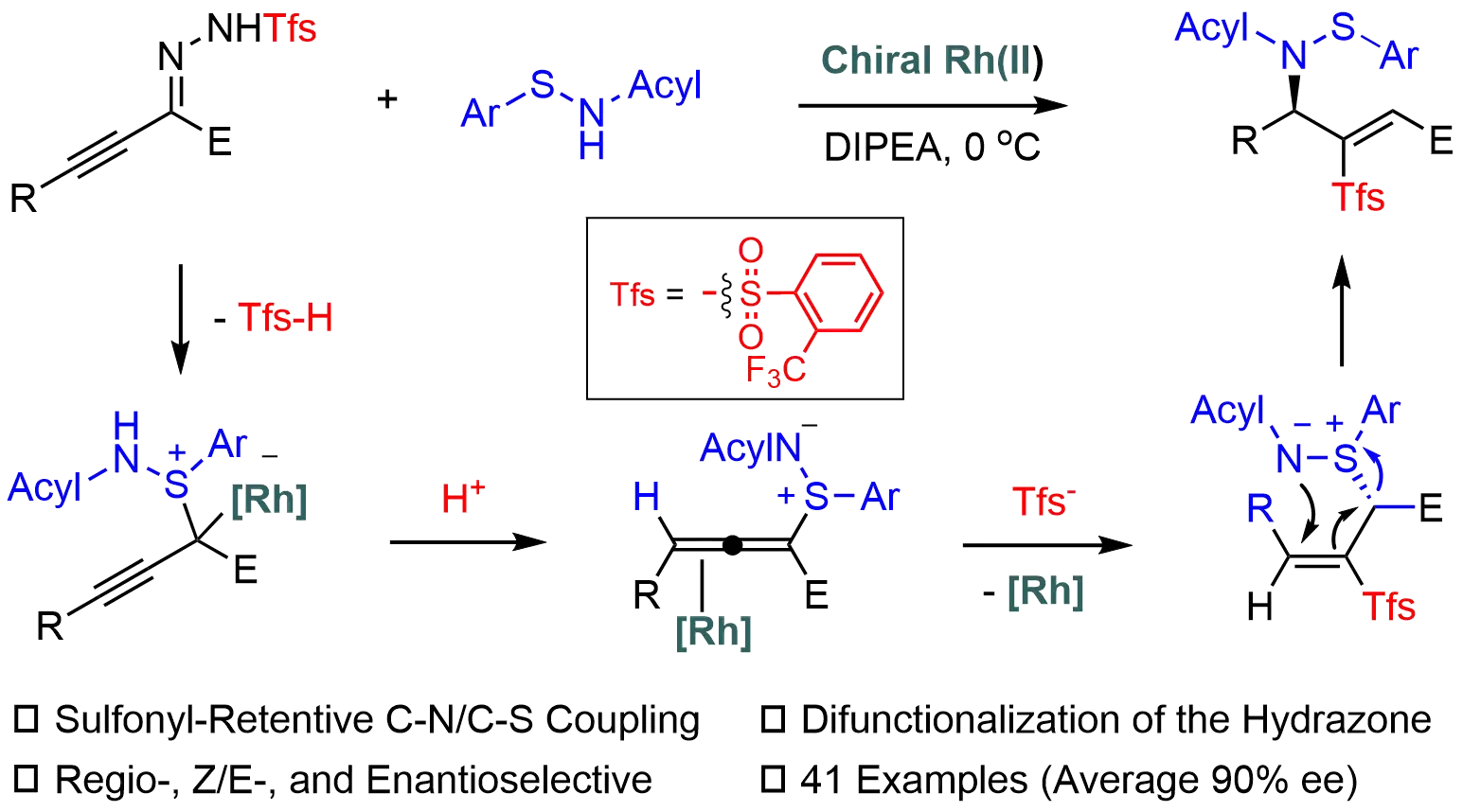
Sulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated ...
MoreSulfonylhydrazones are valuable carbene precursors in asymmetric synthesis; however, their use typically generates sulfinic acid, which is inevitably discarded as stoichiometric waste. In this work, the carbene chemistry and sulfur chemistry are integrated in Rh-catalyzed asymmetric sulfonyl retentive S,N-difunctionalization of alkynyl N-sulfonylhydrazones with sulfenamides. The sulfonyl group or the corresponding sulfinic acid is retained in a formal migration. This mild and efficient protocol allows for straightforward construction of enantioenriched sulfonyl-based allylic sulfenamides with excellent chemo-, E/Z-, and enantioselectivity, thereby offering a creative strategy for achieving more atom-economic transformations of carbene precursors. Mechanistic studies reveal a three-step process involving S-allenylation, hydrosulfonylation, and an aza-Mislow-Evans rearrangement, with the hydrosulfonylation step governing enantioselectivity.
Less -
Bin Wei, ... Xingwei Li
-
DOI: https://doi.org/10.70401/cc.2025.0004 - December 12, 2025
Asymmetric construction of non-central chiral compounds with N-heterocyclic carbenes organocatalysis
-
This review systematically summarizes recent advances in N-heterocyclic carbene (NHC)-catalyzed asymmetric synthesis of non-central chiral compounds. The key synthetic strategies include oxidative and redox-neutral acylation reactions, LUMO-activation-mediated ...
MoreThis review systematically summarizes recent advances in N-heterocyclic carbene (NHC)-catalyzed asymmetric synthesis of non-central chiral compounds. The key synthetic strategies include oxidative and redox-neutral acylation reactions, LUMO-activation-mediated conjugate additions, cyclization reactions, benzoin/Stetter reactions, and imine activation processes. In this work, the versatility of NHCs in asymmetric synthesis is highlighted by their capacity to achieve high stereoselectivity across a wide range of substrates. These advances provide valuable insights for applications in chiral drug development, materials science, and catalysis.
Less -
Peng Chen, ... Zhichao Jin
-
DOI: https://doi.org/10.70401/cc.2025.0003 - December 09, 2025
Advances in catalytic asymmetric hydrogenation of third-row heteroatom-substituted alkenes
-
The asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction ...
MoreThe asymmetric hydrogenation of vinyl silanes, vinyl sulfides, vinyl phosphines, and vinyl chlorides, those substituted with heteroatoms from the third-row of the periodic table, has emerged as a valuable and environmentally friendly method for the construction of the related optically active organosilanes, organosulfides, organophosphine, and organochlorides. These compounds have shown considerable potential for preparing functional molecules and synthesizing natural products. Over the past few decades, considerable research efforts have focused on the design and development of transition-metal catalysts featuring chiral ligands for the asymmetric hydrogenation of such substrates. In parallel, in-depth mechanistic studies have been conducted to elucidate the pathways of these enantioselective hydrogenation reactions, significantly advancing the understanding of their catalytic behavior and stereocontrol. This review focuses on the recent momentum and key advancements in the enantioselective hydrogenation of vinyl silanes, vinyl sulfides, and vinyl chlorides. In addition, given the widespread industrial interest in these compounds, the practical utility of this transformation in the synthesis of chiral silanes, chiral thioethers, chiral alkyl chlorides, as well as related derivatives, is also discussed.
Less -
Jian Zhang, ... Wanbin Zhang
-
DOI: https://doi.org/10.70401/cc.2025.0002 - November 27, 2025
Transition-metal-catalyzed asymmetric denitrogenative transannulation
-
Over the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product ...
MoreOver the past few decades, denitrogenation has proven to be an effective method for synthesizing high-value chiral heterocyclic compounds. These compounds find widespread applications in pharmaceutical chemistry, drug development, and natural product synthesis. Denitrogenation demonstrates high activity and can engage in cyclization reactions with olefins, alkynes, carbon-heterocycles, aldehydes, and other reagents. This one-step operation enables the rapid construction of chiral heterocycles such as pyrroles and indoles, significantly shortening complex synthetic pathways. Innovations in chiral ligands, optimization of catalytic systems, and detailed studies on mechanisms have significantly enhanced the enantioselectivity and substrate applicability of denitrogenation reactions. This review highlights recent advancements in the synthesis of chiral heterocycles via denitrogenation reactions and systematically examines the reaction characteristics of various metal catalytic systems.
Less -
Wen-Ge Guo, Ren-Rong Liu
-
DOI: https://doi.org/10.70401/cc.2025.0001 - November 06, 2025