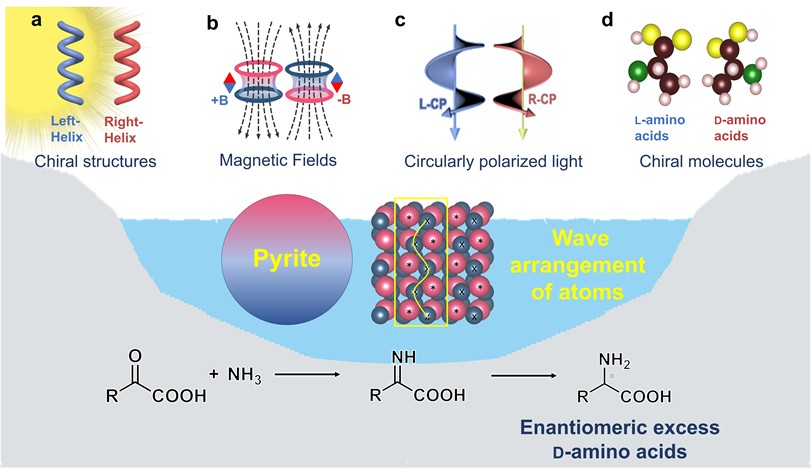
Abstract
L-amino acids are ubiquitous in biology, yet the origin of their homochirality is an enduring mystery. Prebiotic chiral selection process of amino acids could significantly influence subsequent molecular evolution and the emergence of life. However, the distribution of chirality of amino acids remains convoluted, which poses obstacles to the in-depth study of prebiotic chiral selection of amino acids. Here, we propose that minerals in the prebiotic environment might have hindered the absolute selection of L-amino acids to induce a random distribution of L- and D-amino acids during the early stages of life’s evolution. Pyrite, abundant in the prebiotic world, predominantly selected D-amino acids in the photocatalytic reductive amination of α-keto acids, resisting the enantioselectivity of chiral driving forces like chiral structures, magnetic fields, circularly polarized light, and chiral molecules. This resistance is due to the wavy atomic arrangement of pyrite surfaces, which causes larger reaction energy differences between enantiomers. Our findings provide evidence to assess the validity of prebiotic chiral selection scenarios for biomolecules, an important step in understanding the origin of life.
Graphical Abstract
Keywords
1. Introduction
The absolute selection of L-amino acids has been elucidated through a variety of potential chiral factors that may have been present on primitive Earth[1-9]. These factors include the parity violation in weak interactions[10,11], enantioselective adsorption on mineral surfaces[12-15], chiral physical fields (including magneto-chiral effects[7,16,17], vortices within magnetic fields[18,19]), chirality-induced spin selectivity (CISS)[20-22], circularly polarized light (CPL)[23-25], and trace amounts of chiral molecules delivered by extraterrestrial meteorites[26-28].
The random distribution of L- and D-amino acids in primitive Earth[5,29,30] is supported by several key observations. For instance, ribosomes can interact with both L-peptides and D-peptides[31], RNA composed of L-ribose nucleotides exhibits biological activity towards D-peptides[32], and even racemic amino acids can spontaneously form homochiral peptide chains with “folding chirality” during polymerization[30,33]. However, these findings only indicate that some of the initial steps in the production of biological macromolecules may not require homochirality of amino acids[7]. There is insufficient evidence to support the random emergence and selective preference for the chirality of amino acids at the early stage of life’s evolution.
Herein, we propose that pyrite, known to be widely found in marine systems and on early Earth[34], is a possible factor interfering with the absolute chiral selection of L-amino acids (Figure 1). The photocatalytic reductive amination of α-keto acids is considered a potential pathway for prebiotic amino acid formation[38-40]. In our previous work, it has been proved that the wavy arrangement of atoms on the surface of pyrite leads to the preferential formation of D-amino acids due to the stronger affinity compared to their enantiomers[41]. The intrinsic enantioselectivity of the pyrite surface for D-amino acids[41] may resist the influence of various chiral driving forces (CDFs) that have been demonstrated to potentially favor L-amino acids[6,42-44], which may support the second hypothesis for the random distribution of L- and D-amino acids[38] by interfering with the chiral selection of L-amino acids.
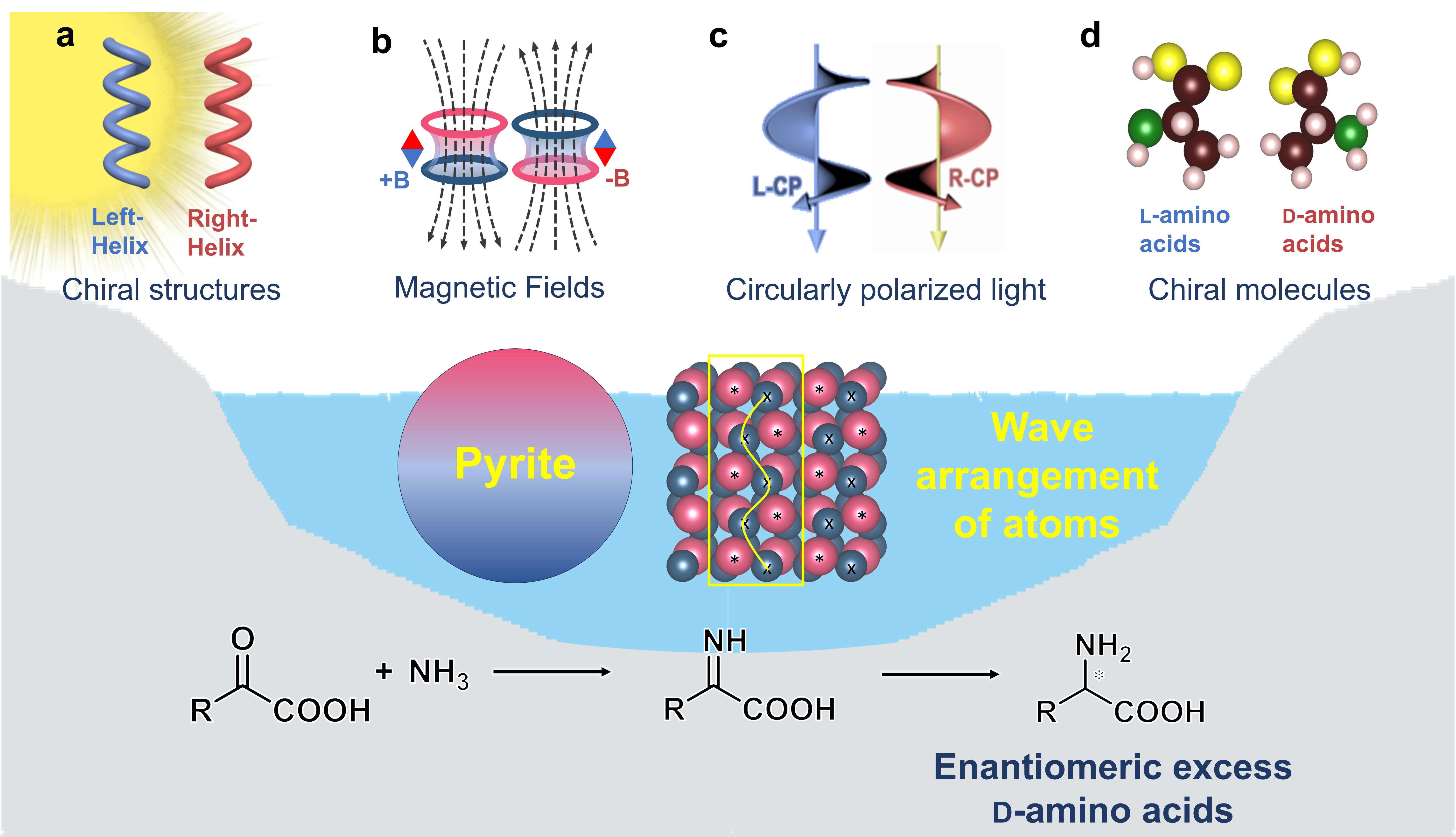
Figure 1. Schematic diagram for investigating the influence of various CDFs on the asymmetric photocatalytic generation of D-amino acids by reductive amination of α-keto acids on pyrite. (a) Chiral inorganic structures may have been induced by various chiral force fields present on primitive Earth during their deposition[35]; (b) Magnetic field has been supported to exist on primitive Earth by several lines of evidence, such as paleomagnetic records, magnetized zircon crystals, and Geodynamic modeling[21]; (c) CPL could be generated by the Faraday effect, which stems from sunlight’s interaction with Earth’s magnetic field[36]; (d) Chiral molecules may have been delivered from outer space[37] or produced by other CDFs on primitive Earth. CDFs: chiral driving forces; CPL: circularly polarized light.
To explore this possibility, we evaluated the enantiomeric excess (ee) of amino acids produced from the asymmetric photocatalysis on pyrite under the influence of various CDFs (Figure 1a,b,c,d). These CDFs include chiral structure, magnetic fields[16], CPL[23,24] and chiral molecules[37], all of which are recognized to have existed in the primitive Earth environment (for details, see the caption of Figure 1). As one of the typical chiral inorganic structures, chiral mesostructures have been proven to exhibit significant enantiomeric selection due to their spin polarization effect, which provides an energy difference between the activation energies for L- and D-amino acids[22], or due to chiral surfaces enabling selective adsorption[14,15,45]. Thus, the chiral mesostructured pyrite (CMP) was synthesized for investigating the role of chiral structures of minerals on the primitive Earth. Here, the natural pyrite (NP), synthesized pyrite (SP), and CMP were employed as photocatalysts for the synthesis of alanine (Ala) from the amination of pyruvic acid as a model reaction, in the presence of magnetic fields or CPL or chiral molecules.
2. Experimental
2.1 Preparation of CMP
The fluorine-doped tin oxide (FTO) substrates (40*10*1.1 mm, coating thickness = 500 nm, sheet resistance ≤ 15 Ω) were washed with acetone, ethanol and DI water for 15 min respectively, in an ultrasonic bath. For the surface activation, the substrates were placed in glass vials filled with 20 mL of 0.04-10 mM fresh KMnO4 solution and 50 μL of n-butanol (as a reducing agent for the KMnO4) at 85 °C for 30 min. Permanganate-treated FTO substrates were then thoroughly rinsed in deionized water and sonicated for 10 min.
CMP was synthesized by a hydrothermal method. Deionized water used in all reactions was passed through N2 for 30 minutes to remove oxygen. In a typical synthesis of D-CMP, FeCl2·4H2O (0.1 g, 0.5 mmol) and D-Pen (0.23 g, 1.5 mmol) were dissolved in deionized water (40 mL). After the mixture was stirred for 20 min, the above mixed solution and permanganate-treated FTO substrate were transferred into a 50 ml Teflon-lined autoclave, and hydrothermally heated at 180 °C for 3 h. After cooling to room temperature, the FTO substrates coated with chiral pyrite were washed with deionized water and ethanol several times to remove the residual organics and then dried in vacuum for 12 h.
2.2 Morphology and structural characterizations of the catalyst
Powder XRD patterns were recorded on a Rigaku MiniFlex 600 powder diffractometer equipped with Cu Kα radiation (40 kV, 15 mA), at a rate of 0.2o min-1 over the range of 20-80o. SEM was conducted on a JEOL JSM-7100F electron microscope operating at 2 kV. TEM images were taken with an JEOL JEM-F200 TEM microscope operated at 200 kV. Images were recorded using a Gatan OneView IS camera. The CD spectra were obtained on a JASCO J-1500 spectropolarimeter fitted with a DRCD apparatus, and data were collected with a scanning rate of 100 nm/min ranging from 200 to 800 nm at 293 K. XPS spectra were recorded on a Kratos AXIS UltraDLD spectrometer with a monochromated Al Kα source (1,486.6 eV); all energies are reported as binding energies in eV and referenced to the C 1s signal (corrected to 284.8 eV) for aliphatic carbon on the analyzed sample surface. The mc-AFM spectra were obtained on Bio-Fast Scan AFM with a magnetic Pt-coated Cr tip (Multi75E-G, Budget Sensors) with nominal spring constant 3 N/m was used to acquire I-V curves. The tips are pre-magnetized using a 1.6 T permanent magnet. The samples were cut into 0.2 × 0.2 cm pieces using scissors and attached to the base of the AFM with conductive tape. I-V curves were measured in the middle area 5 × 5 µm, equally spaced at 25 locations. Absorption spectra under magnetic fields were recorded by mounting an electromagnet in the sample chamber of the JASCO J-1500 spectropolarimeter, which affords a magnetic field up to 1.5 T.
2.3 Photocatalytic reductive amination
In a typical photocatalytic procedure, deionized water used in all reactions was passed through N2 for 30 minutes to remove oxygen. Pyrite catalysts (10 mg of NP and SP and 1 × 1 cm2 of CMP) were added to the photocatalytic reactor. Then the reactor was deoxygenated by purging N2 for 10 minutes; after that, the mixture of pyruvic acid (30 μmol) and NH3•H2O (0.21 mL, 28% wt) in 1.5 mL deionized water was injected into the reactor. The reaction liquid was irradiated by a 254 nm wavelength ultraviolet lamp (Beijing NBET Technology Co., Ltd., NBET-LED100, with a power of 100 W), the average light intensity irradiated on the photoreactor was ca. 27 mW cm-2. The reaction was performed by manually shaking for 8 h or by magnetic stirring for 24 h. The precipitates were removed by cooling and centrifugation. Finally, the solution was lyophilized to remove water. The yields and ee of Ala were analyzed by high-performance liquid chromatography–mass spectrometry (HPLC-MS).
In this work, catalytic performance was described by production yield calculated via equations (1, 2, and 3).
The production yield of Ala (Ys, µmol·h-1·gcat-1).
where Vcat is the volume of catalyst, Scat is the surface area of catalyst, Thkcat is the thickness of catalyst, mcat is the mass of catalyst, ρcat is the density of catalyst (ρcat = 5 g·cm-3), cumulative production (ns)was obtained using the LC-MS analyzer, while t is the reaction time.
2.4 The yields and ee of Ala analysis
The purified product was derivatized by L-FDLA before separation by HPLC-MS. To derivatize amino acid compounds, the product was dissolved in 500 μl of deionized water, and then 50 μL of an aqueous solution was mixed with 20 μL of 200 mM sodium bicarbonate and 20 μL of 1 % L-FDLA in acetone. The mixture was incubated at 37 °C for 30 min. After returning to room temperature, 200 μL of methanol was added to the sample, and the impurity was centrifuged (8,049 g, 10 min). The sample was kept in the dark condition.
HPLC-MS analysis was performed by using an Acquity UPLC & XEVO G2-XS QTOF (Waters, United States) equipped with a UPLC column (Acquity UPLC HSS T3 1.8 μm, 2.1*100 mm). The detection of samples was conducted in positive ESI mode, and the function type was TOF-MS with a mass range of 200-600. Then, a 0.1 μL sample was injected and eluted using mobile phase A (0.1% mass concentration of formic acid aqueous solution) and mobile phase B (acetonitrile) at a flow rate of 0.35 ml/min. The programmed mobile phase gradient was as follows: 0.00-1.00 min, 25% B; 1.00-4.30 min, 35% B; 4.30-6.50 min, 100% B; 6.50-40 min, 25% B. The column was equilibrated prior to sample injection, and the temperature of the column oven was set at 45 °C.
To generate a standard curve of L- and D-Amino acids (AAs), the standard compounds were diluted in water at concentrations adjusted to be relevant to product contents. After derivatization, 10 μl of the standard solution was separated and detected using the HPLC-MS system.
The ee was calculated based on the integrated peak areas of L- and D-AAs as indicated in equation 4.
where L-AAs and D-AAs represent the obtained peak areas of L-FDLA derivatized L-AAs and L-FDLA-derivatized D-AAs, respectively. The standard error of the mean of the %ee was calculated by means of equation 5.
where S is the standard deviation and n is the number of experiments.
2.5 Photocatalytic reductive amination on pyrite under magnetic fields
A permanent magnet was placed beside the light source to apply magnetic fields parallel to the direction of light propagation to the reaction vessel. The reaction vessel could be inserted into the permanent magnet setup (Figure S16).
The reaction solution was exposed to magnetic fields and irradiated by a 254 nm ultraviolet lamp through a 1 cm diameter light window at 25 °C. Since magnetic stirring was not feasible inside the strong magnetic chamber, periodic manual shaking was employed to facilitate the reaction. Every 15 minutes, the reactor was removed from the magnetic field, thoroughly shaken, and then returned to its original position. The reaction was carried out for 8 h.
As a control experiment, the photocatalytic reaction without magnetic fields was conducted under similar conditions by placing the reactor in a perforated dark box to replace the permanent magnet.
2.6 Photocatalytic reductive amination on pyrite under CPL
The reaction solution was irradiated with L-/R-CPL produced by a 254 nm ultraviolet lamp equipped with an adjustable circular polarizer (ELLITOP Scientific Co., Ltd.). The reaction was conducted under magnetic stirring for 24 h. Other steps followed the typical photocatalytic procedure.
2.7 Photocatalytic reductive amination on pyrite with additional chiral molecules
To introduce chiral molecules into the SP catalytic system, SP powder (10 mg) and chiral molecules (400 μmol) were added to 2 mL of Millipore water in the photocatalytic reactor and vigorously stirred for 1 h to ensure complete coordination between the chiral molecules and pyrite. Then, pyruvic acid (30 μmol) and NH3•H2O (0.21 mL, 28% wt.) were injected into the reactor. The reaction was conducted under magnetic stirring for 24 h. Other steps followed the typical photocatalytic procedure.
2.8 Computational details
Density functional theory calculations were performed using the plane-wave based Vienna Ab Initio Simulation Package (VASP)[46]. The wavefunction was expanded in a plane-wave basis set with an energy cutoff of 480 eV. The electron exchange and correlation effects were described by the generalized gradient approximation (GGA) in the form of the Perdew–Burke–Ernzerhof (PBE) functional[47]. The interaction between atomic cores and electrons was described by the projector augmented wave (PAW) method[47]. A cut-off energy of 480 eV for the plane-wave basis set and an atomic force convergence of 0.02 eV/Å were employed. The adsorption energy of each amino acid was calculated as Eads = EAA* - (Eslab + EAA).
NormaL-FeS2 is derived from the (200) plane of the bulk phase pyrite, and maintains the periodicity in the x, y direction while extending in the z direction. Screw- FeS2 is to move the atom on the basis of NormaL- FeS2, thus inserting a Burgers vector ±2.3 Å (length of Fe-S bond, ± represents torsion in right (+) and left (-) directions, respectively) screw dislocation along the Z direction in the plane perpendicular to XY plane.
The screwed (200) was modelled using (2 × 2) supercells with a (1 × 1 × 1) k-point grid. In addition, the models of the (200) have a 4-atom layer in the z-axis direction and a 25 Å vacuum slab between mirror images in the z-axis in the unit cell.
3. Results and Discussion
3.1 Preparation of NP, SP and CMP
The NP was meticulously ground and sieved to achieve uniform dispersion in the reaction solution[41]. SP was selected as a clean catalyst to eliminate potential interference from chiral factors that may arise due to biological activity on NP. CMP films were synthesized on fluorine-doped tin oxide (FTO) substrates using penicillamine (Pen) as both a symmetry-breaking agent and sulfide source, and using ferrous salts as the iron source (Figure S1 and Figure S2). The asymmetric arrangement of Fe-NH2 and Fe-COOH bonds around the chiral carbon center regulates the oriented, misaligned growth of nanocrystals, leading to the formation of a chiral mesostructure. The pure inorganic CMP can be obtained by removing organics through washing the films (Figure S3 and Figure S4). The final products synthesized with D- and L-Pen were denoted as D- and L-CMP, respectively.
3.2 Structural characterization of NP, SP and CMP
Figure 2a shows the wide-angle X-ray diffraction (XRD) patterns of NP, SP, D- and L-CMP. The reflections are well indexed to the pure cubic phase of FeS2 with a space group of Pa-3 (JCPDS Card No. 42-1340). Compared to NP and SP, the intensity of the 200 reflection in CMP is relatively enhanced, while the 311 reflection is decreased, indicating that the pyrite crystal exhibited preferential growth along the [100] direction.
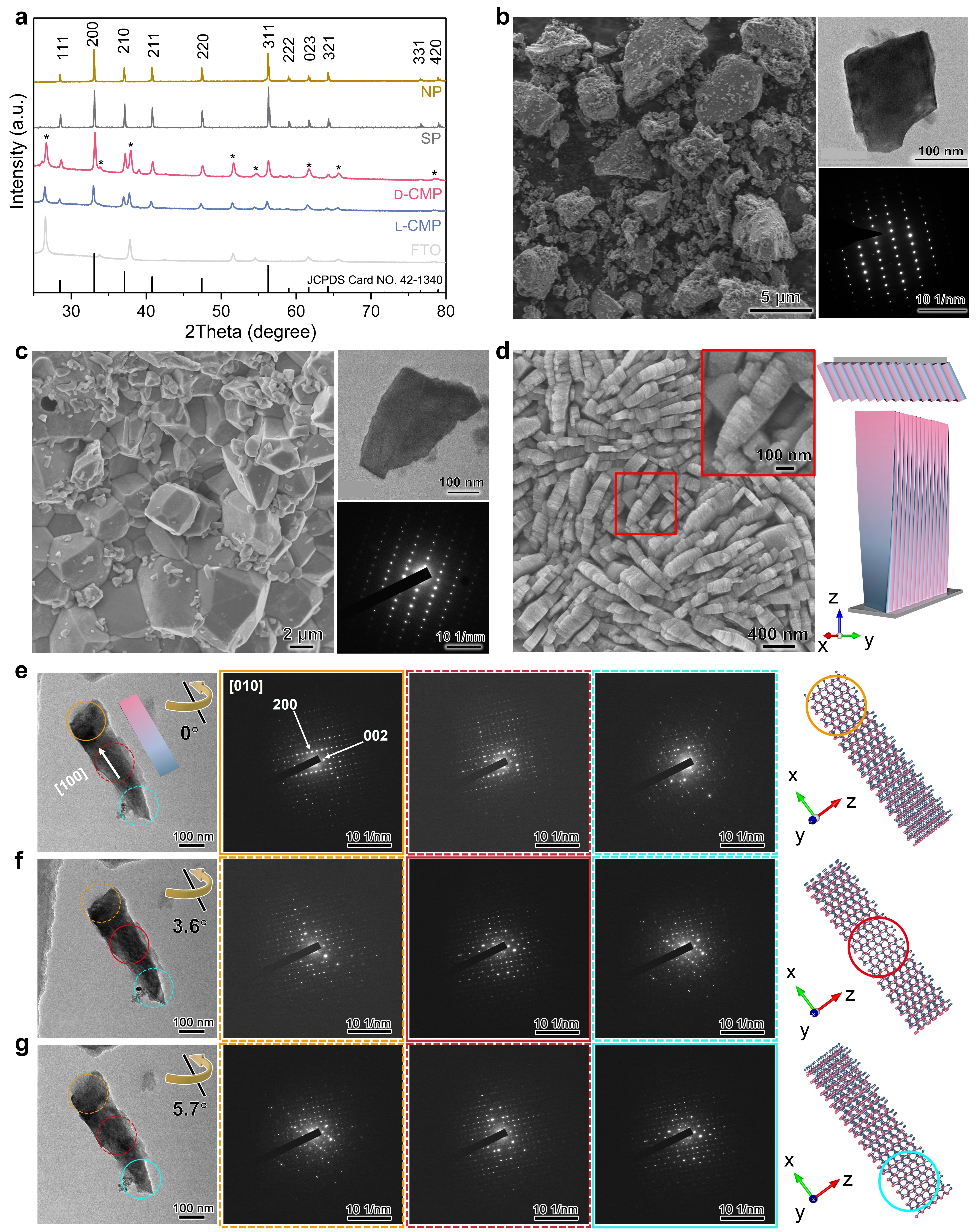
Figure 2. Morphologies and structures of NP, SP, and antipodal CMP. (a) XRD patterns. Reflections of FTO are labeled by *; (b-g) SEM images, TEM images, and corresponding SAED patterns of NP (b), SP (c) and D-CMP (d-g), as well as the structural models of D-CMP. The [100] axis in the nanoflakes (e) can only be aligned to the incident electron beam in the top region (orange circle), by tilting the nanoflakes along the [100] axis by 3.6° and 5.7°, the middle region (red circle) (f) and bottom region (cyan circle) (g) can be well aligned, respectively. The structural models depicting these nanoflakes with a chiral relationship were generated based on the TEM observations. The synthetic molar composition of D-CMP was 1.5 D-Pen: 0.5 FeCl2•H2O: 2222 H2O. NP: natural pyrite; SP: synthesized pyrite; CMP: chiral mesostructured pyrite; XRD: X-ray diffraction; FTO: fluorine-doped tin oxide; SEM: scanning electron microscopy; TEM: transmission electron microscopy; SAED: selected area electronic diffraction.
The scanning electron microscopy (SEM) shows that NP particles exhibit an irregular morphology with sizes ranging from ca. 2 to 10 µm (Figure 2b), whereas SP displays a regular polygonal crystalline structure with sizes of ca. 1.3 µm indicating its purity (Figure 2c). The three crystal surfaces {111}, {200}, and {220} are mostly exposed in the NP and SP particles[41], judging from the transmission electron microscopy (TEM) images of NP and the morphology of SP crystals (Figure S5). The TEM images of NP and SP display a single-crystalline cubic structure (Figure 2b,c).
The SEM image of D-CMP (Figure 2d) shows that the films are composed of pyrite nanoflakes with lengths in the range of ca. 500-700 nm, thicknesses of ca. 100 nm, and heights of ca. 3 µm (Figure S6). Each nanoflake is assembled with nanosheets with a thickness of ca. 10 nm (in set of Figure 2d). The TEM images and the corresponding selected area electronic diffraction (SAED) patterns of D-CMP (Figure 2e,f,g) reveal the crystal growth direction along the [100] axis, which is consistent with the XRD patterns. Crystal planes such as {200}, {102}, {302} and {402} were predominantly observed (Figure S7). The alignment of the electron beam to [100] axis, observed by tilting the sample along the [100] direction in the right-direction, indicates that the crystal structure of the D-CMP nanoflakes is helically distorted along the [100] direction in a right-handed configuration, and vice versa in L-CMP (Figure S8).
3.3 Spin polarization and optical activity (OA) of NP, SP and CMP
Given that spin polarization is the key factor for the enantioselectivity of asymmetric reactions[21,44], the spin polarization properties of NP, SP, and antipodal CMP were characterized using magnetic conductive atomic force microscopy (mc-AFM) measurements (Figure S9). As shown in Figure 3a,b, the averaged I-V curves of four samples showed that the current increased with increasing bias voltage measured by applying a voltage from -0.45 to 0.45 V (Figure S10). In NP and SP, a slightly stronger tunneling current was detected when the tip was magnetized in the DOWN orientation compared to the UP orientation, exhibiting slight spin DOWN polarization, which may be attributed to the intrinsic asymmetric factor[48] of the wavy arrangement of surface atoms. D- and L-CMP exhibit substantial spin-DOWN and spin-UP polarization, respectively, evidenced by the significant differences in tunneling current observed using a magnetization tip with opposite orientations. The average spin polarization degrees of NP and SP were calculated to be consistently negative, with values of ca. -5.62% and -5.55%, respectively. Those of D- and L-CMP exhibited significant values, ca. -78.89% and ca. +79.91%, respectively (Figure S11).
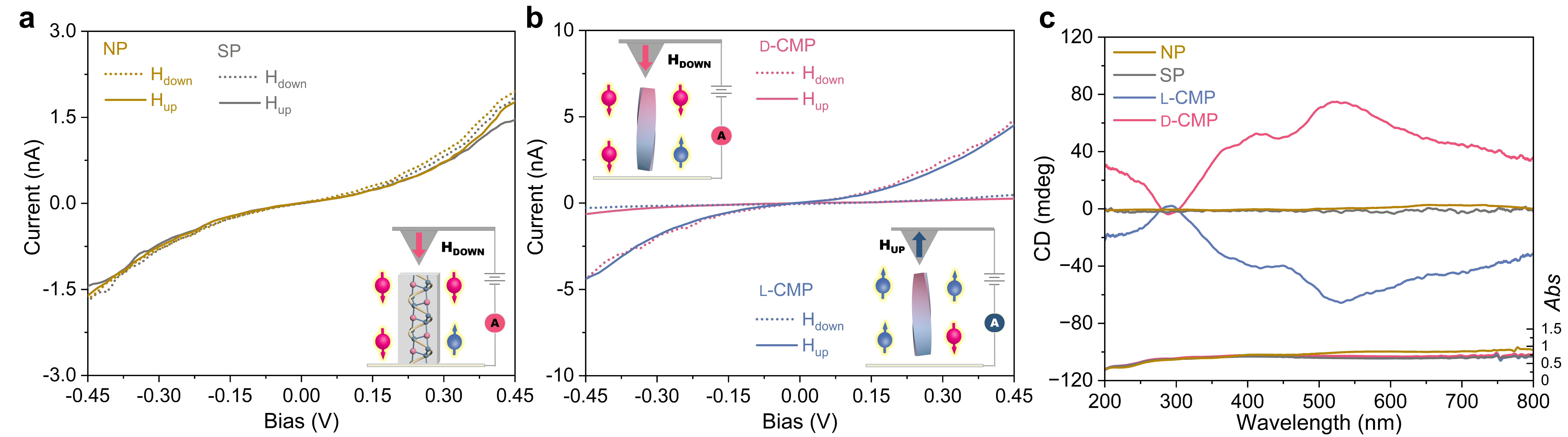
Figure 3. Spin polarization and OA of NP, SP, and antipodal CMP. (a, b) Averaged I−V curves of mc-AFM measurements; (c) DRUV−vis and DRCD spectra. OA: optical activity; NP: natural pyrite; SP: synthesized pyrite; CMP: chiral mesostructured pyrite; AFM: atomic force microscopy; DRUV: diffuse reflection UV; DRCD: diffuse reflection circular dichroism.
The spin polarization of four samples can be explained by the CISS effect[49,50]. The applied voltage causes electrons to transport along a helical path, generating directional magnetic fields (Bchiral) depending on the asymmetric factor in NP and SP[48], and the helical mesostrcutue of CMP. The chiral potential induces spin-orbit coupling (SOC), represented by µ·B, where the magnetic moment of an electron (µ) interacts with the magnetic field (B), stabilizing one spin direction and destabilizing the other, depending on their handedness[50-52]. This asymmetric SOC in opposite chirality leads to differing energy shifts for opposite spin states of moving electrons, causing more spin-DOWN electrons to transition through NP, SP, and D-CMP and more spin-UP electrons through L-CMP.
The chiral structure can be characterized by OA due to their selective interaction with CPL[53]. Figure 3c presents the diffuse reflection UV/Vis absorption spectrum and diffuse reflection circular dichroism (DRCD) spectra of NP, SP and antipodal CMP. All samples exhibit total absorption in the range of 200-800 nm attributed to the electron transition from t2g (valence band [VB]) to eg (conduction band [CB])[54]. NP and SP exhibit the silent CD signals. Significantly mirror-imaged CD signals were observed in the L-/D-CMP (Figure S12).
The DRCD signals of chiral materials are a combination of three circular dichroic sources (Figure S13)[52]: (i) natural CD signals (µ•m) from electron transition from VB to CB, (ii) CD signals from spin polarization induced by Bchiral, and (iii) scattering-based CD signals from circular Bragg resonance. Based on the OA behavior observed in the CD spectra before and after water infiltration (Figure S13), right- and left-handed chiral structures in D- and L-CMP were confirmed, respectively, which is consistent with TEM observations. In NP and SP, almost no CD signals were detected, despite the presence of spin polarization, likely due to the weak interaction between the CPL and atomic scale arrangement. However, the slight spin polarization that can be detected is probably a result of signal amplification by mc-AFM measurements (Figure S11).
3.4 Photocatalytic formation of Ala on NP, SP and antipodal CMP under various CDFs
The results of photocatalytic reactions on NP, SP, and antipodal CMP, performed under various CDFs with optimized conditions[41] (detailed in METHODS, Figure S14 and Figure S15), are illustrated in Figure 4. A permanent magnet was used to apply magnetic fields parallel (+B) and antiparallel (-B) to the light propagation direction (Figure S16). CPL was generated using a 254 nm ultraviolet lamp equipped with an adjustable circular polarizer. To investigate the influence of chiral molecules, the enantiopure amino acids, including L-/D-serine (Ser), L-/D-valine (Val), and L-/D-phenylalanine (Phe), were added to the photocatalytic reaction system of SP.
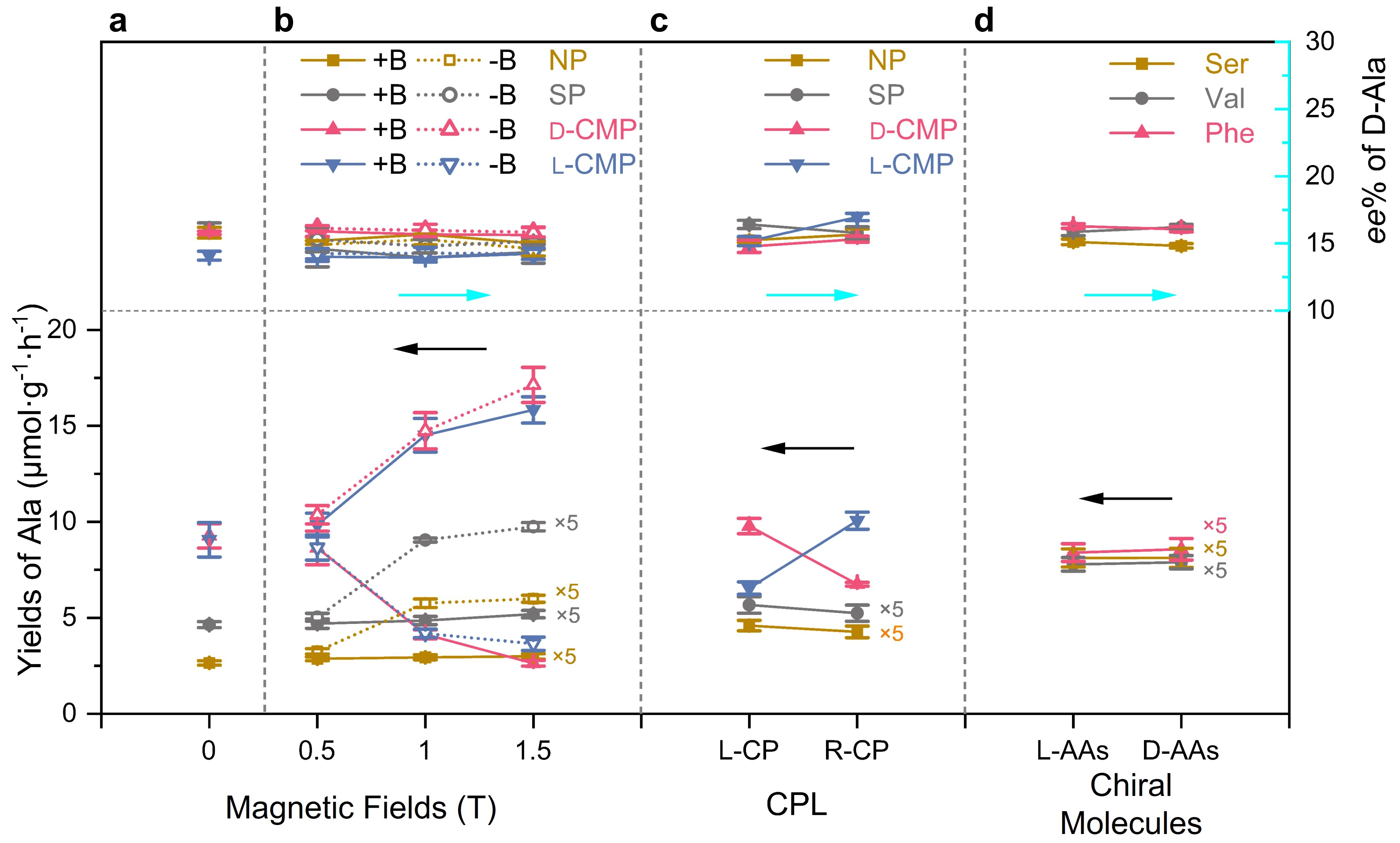
Figure 4. Yields and ee values of Ala formed through the photocatalytic reductive amination of pyruvic acid on NP, SP, D- and L-CMPs, under various CDFs. (a) Without CDFs; (b) Different magnetic fields, oriented parallel (+B, solid line) and antiparallel (-B, dashed line) to the direction of light propagation; (c) Irradiation with L- and R-CPL; (d) Different additional chiral molecules of L-/D-Ser, Val, and Phe in the SP photocatalytic system. Error bars represent the standard deviations calculated from three independent photocatalytic experiments. Reactions were carried out with a composition of 30 µmol α-keto acids, 3 mmol ammonia, 1.5 ml H2O, and 10 mg of pyrite powders (or 1 × 1 cm2 CMP film) with irradiation of a 254 nm UV lamp (or L-/R-CP with 254 nm) at room temperature for 24 h. The different yields under various CDFs were caused by differences in the mixing conditions of the reactants. The yields obtained with shaking in the magnetic fields were lower than those obtained with stirring in the other systems. NP: natural pyrite; SP: synthesized pyrite; CMPs: chiral mesostructured pyrites; CDFs: chiral driving forces; CPL: circularly polarized light.
Figure 4a shows that, without any external CDFs, the yields of Ala from NP, SP, and antipodal CMP are in the order of CMP >> SP > NP, relating to their particle size, light absorption, and charge carrier behavior. When an external magnetic field is applied (Figure 4b), the yield of Ala for both NP and SP increases slightly under +B, while it increases significantly under -B. For D-CMP, the yield of Ala significantly decreases under +B and increases under -B, and vice versa for L-CMP. These results demonstrate that the chirality induced magnetic field and external magnetic field can induce asymmetric interactions with chiral systems, significantly affecting the activity of the photocatalyst probably due to light absorption properties (Figure S17 and Figure S18) and charge carrier behavior of pyrite (Figure S19) in the presence of a magnetic field (Figure S20). As shown in Figure 4c, with L-CPL light, the yields of Ala on NP and SP were slightly higher than with R-CPL light. This difference was more pronounced with D-CMP, where the L-CPL light yield of Ala is higher than that of R-CPL, and vice versa for L-CMP. This could be attributed to the selective absorption of L- and R-CPL by chiral materials, and selective spin polarization by L- and R-CPL. In the SP reaction system containing chiral molecules, the yields of Ala were identical with enantiomers (Figure 4d), indicating that this pyrite reaction system was not influenced by chiral molecules.
As shown in Figure 4, the ee value of Ala shows exceptional consistency throughout all CDFs employed, despite the significant differences in yield among them. In the absence of any external CDFs (Figure 4a), Ala with similar average ee values of the D-form (15.8, 16.1, 15.8, and 14.1 %) were synthesized on NP, SP, D-, and L-CMP, respectively, regardless of their differences in morphology, mesoscopic structure, size, and particularly catalytic activity. Such results excluded the influence of these factors on enantioselectivity. Given that the chiral inorganic mesostructure can achieve an ee value of over 90% due to its chirality induced spin polarization[14,22], these results suggest that the intrinsic enantioselectivity of pyrite can dominate and override the effect of the chiral mesostructure and significant spin polarization of CMP.
Figure 4b shows the ee values detected in the NP, SP, and CMP reaction systems when they are subjected to external magnetic fields varying from -1.5 to +1.5 T. The ee values are remarkably consistent, falling within a narrow range of 14.0-16.1%, despite large differences in the catalytic activity, and despite evidence that magnetic fields can induce enantioselectivity in asymmetric reactions through mechanisms such as electron spin polarization[21,55,56] and the magneto-chiral effect[16].
Despite CPL being recognized as an effective CDF for absolute chiral selection[7], no significant differences in the ee values of D-Ala were observed on the four catalysts under irradiation with L- and R-CPL (Figure 4c). Moreover, although chiral molecules and complexes are widely acknowledged as highly effective asymmetric catalysts due to their steric effects[49-52], the addition of three pairs of chiral molecules to the SP reaction system still resulted in D-Ala products with ee values ranging from 14.8 to 16.3% for both enantiomers (Figure 4d).
3.5 Plausible mechanism of resistance of pyrite enantioselectivity for amino acids to CDFs
The key role of inherent enantiomeric selectivity of pyrite, which resists all the CDFs introduced in this work, can be explained in terms of the differences in activation energy for enantiomeric formation of amino acids. As demonstrated in our previous work, on the (200) crystal surface of pyrite, the difference in Gibbs free energies for the formation of D- and L-Ala is 0.344 eV, probably due to the asymmetric factor of the wavy atomic arrangement providing enantioselective affinity (Figure S21)[41].
To investigate the effect of geometrical chirality of mesostructures on enantioselectivity, DFT calculations were performed to determine the adsorption energy of Ala enantiomer on the (200) crystal plane with mirror imaged chiral distortion. The adsorption energy of the Ala enantiomers exhibited a significant difference on oppositely handed screw (200) surfaces. This finding implies that chiral mesostructures would display different enantioselectivity, as the geometric handedness leads to different interactions. Nevertheless, the surface of pyrite seems to exhibit resistance to the enantioselectivity of antipodal chiral distortion (Figure S22).
On the other hand, electron spin polarization has been reported to function as a chiral reagent in asymmetric catalysis[21,44,56,57]. However, in the photocatalytic reductive amination of α-keto acids to amino acids, the effect of spin polarization is predicted to yield only ca. 0.02-0.1 eV energy difference between amino acids enantiomers[21]. Therefore, it could be considered that this energy difference induced by spin polarization cannot surpass that arising from the atomic arrangement of the pyrite surface (0.334 eV). When two energetic effects coexist, the one with the larger magnitude typically dominates kinetically. Thus, the pyrite surface structure exerts a direct and more substantial influence on the activation barrier, rendering spin polarization negligible for enantiocontrol, as its energy contribution is insufficient to meaningfully alter the relative transition state energies or affect the enantiomeric excess. Consequently, pyrite maintains consistent enantioselectivity even in the presence of strong spin polarization produced by chiral mesostructures, magnetic fields, and irradiation with CPL.
For the reaction system containing chiral molecules, the pyrite surface retained its enantioselectivity for D-amino acids, even in the presence of contamination from other chiral species. This expectation can be attributed to several factors. Although the chiral molecules themselves provide chiral bias, they do not alter the enantioselectivity of pyrite. The wavy atomic arrangement of the pyrite surface remains intact, as Fe-chiral molecule complexes could not be formed, and thus the surface is not disrupted by chiral molecules (Table S1).
The low enantioselectivity of pyrite is likely attributed to the presence of several other surfaces among NP, SP, and CMP, such as the {111}, which lack a wavy atomic arrangement[39].
4. Conclusion
In summary, this work provided possible experimental evidence for the random distribution of L- and D-amino acids during the early stages of life’s evolution. Pyrite exhibits a consistent preference for D-amino acids in the photocatalytic reductive amination of α-keto acids, driven by the intrinsic asymmetric factor of the wavy arrangement of atoms on its surface. Under various CDFs, including chiral inorganic mesostructures, magnetic fields, CPL, and the incorporation of chiral molecules, the ee values of D-Ala remained within a narrow range of 14.0 to 16.3%. Given that pyrite is an abundant mineral on primitive Earth and is resistant to chiral selection by various CDFs, these discoveries suggest that pyrite could have interfered with the chiral selection of L-amino acids in the prebiotic world, possibly inducing a random distribution of L- and D-amino acids. Furthermore, chiral driving forces capable of overcoming the interference from pyrite are speculated to be necessary to achieve absolute prebiotic chiral selection of L-amino acids. Pyrite may also influence the enantioselectivity of other minerals, such as calcite. It could facilitate the exposure of crystal facets that preferentially select d-amino acids, thereby broadly interfering with chiral selection.
Supplementary materials
The supplementary material for this article is available at: Supplementary materials.
Authors contribution
Fang Y: Conceptualization, supervision, methodology, writing-original draft, writing-review & editing.
Li R: Investigation, formal analysis, writing-review & editing.
Deng Q, Han L, Duan Y, Wei M: Investigation, formal analysis.
Ouyang T: Methodology.
Conflicts of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 22501180) the Shanghai Natural Science Foundation (Grant No. 25ZR1402587) and the Shanghai Titan Natural Science Development Foundation (Grant No. FF0010561).
Copyright
© The Author(s) 2026.
References
-
1. Mason S. Chemical evolution: Origin of biomolecular chirality. Nature. 1985;314(6010):400-401.[DOI]
-
2. Janoschek R. Theories on the origin of biomolecular homochirality. In: Guijarro A, Yus M, editors. The Origin of Chirality in the Molecules of Life: A Revision from Awareness to the Current Theories and Perspectives of this Unsolved Problem. Berlin: Springer Berlin Heidelberg; 2008. p. 6-20.[DOI]
-
3. Jorissen A, Cerf C. Asymmetric photoreactions as the origin of biomolecular homochirality: A critical review. Orig Life Evol Biosph. 2002;32(2):129-142.[DOI]
-
4. Bada JL. Origins of homochirality. Nature. 1995;374(6523):594-595.[DOI]
-
5. Mejdrová I, Węgrzyn E, Carell T. Step-by-step towards biological homochirality–from prebiotic randomness to perfect asymmetry. Chem. 2025;20:e202401074.[DOI]
-
6. Fuß W. Does life originate from a single molecule? Chirality. 2009;21(2):299-304.[DOI]
-
7. Avalos M, Babiano R, Cintas P, Jiménez JL, Palacios JC, Barron LD. Absolute asymmetric synthesis under physical fields: Facts and fictions. Chem Rev. 1998;98(7):2391-2404.[DOI]
-
8. Deng M, Yu J, Blackmond DG. Symmetry breaking and chiral amplification in prebiotic ligation reactions. Nature. 2024;626(8001):1019-1024.[DOI]
-
9. Tamura K. Origin of homochirality in terrestrial biology. J Biosci. 2025;50:13.[DOI]
-
10. Lee TD, Yang CN. Question of parity conservation in weak interactions. Phys Rev. 1956;104(1):254-258.[DOI]
-
11. Wu CS, Ambler E, Hayward RW, Hoppes DD, Hudson RP. Experimental test of parity conservation in beta decay. Phys Rev. 1957;105(4):1413-1415.[DOI]
-
12. Hazen RM, Sholl DS. Chiral selection on inorganic crystalline surfaces. Nature Mater. 2003;2(6):367-374.[DOI]
-
13. Hazen RM, Filley TR, Goodfriend GA. Selective adsorption ofL- andD-amino acids on calcite: Implications for biochemical homochirality. Proc Natl Acad Sci U S A. 2001;98(10):5487-5490.[DOI]
-
14. Fang Y, Liu X, Liu Z, Han L, Ai J, Zhao G, et al. Synthesis of amino acids by electrocatalytic reduction of CO2 on chiral Cu surfaces. Chem. 2023;9(2):460-471.[DOI]
-
15. Fang Y, Liu X, Ai J, Zhao G, Chen L, Che S, et al. Enantiospecific affinities of chiral Cu films for both D-ribose and L-amino acids. Chem Mater. 2023;35(6):2402-2407.[DOI]
-
16. Rikken GLJA, Raupach E. Enantioselective magnetochiral photochemistry. Nature. 2000;405(6789):932-935.[DOI]
-
17. Guijarro A, Yus M. Chiral physical forces. In: Guijarro A, Yus M, editors. The Origin of Chirality in the Molecules of Life: A Revision from Awareness to the Current Theories and Perspectives of this Unsolved Problem. Berlin: Springer Berlin Heidelberg; 2008. p. 31-71.[DOI]
-
18. Singh G, Chan H, Baskin A, Gelman E, Repnin N, Král P, et al. Self-assembly of magnetite nanocubes into helical superstructures. Science. 2014;345(6201):1149-1153.[DOI]
-
19. Mogi I, Morimoto R, Aogaki R, Watanabe K. Surface chirality induced by rotational electrodeposition in magnetic fields. Sci Rep. 2013;3:2574.[DOI]
-
20. Rosenberg RA, Mishra D, Naaman R. Chiral selective chemistry induced by natural selection of spin-polarized electrons. Angew Chem Int Ed. 2015;54(25):7295-7298.[DOI]
-
21. Ozturk SF, Sasselov DD. On the origins of life’s homochirality: Inducing enantiomeric excess with spin-polarized electrons. Proc Natl Acad Sci U S A. 2022;119(28):e2204765119.[DOI]
-
22. Cui Y, Ai J, Duan Y, Jia M, Ouyang T, Liu A, et al. Enantioselective synthesis of amino acids by photocatalytic reduction of CO2 on chiral mesostructured ZnS. Chem. 2025;11(5):102390.[DOI]
-
23. Bailey J. Astronomical sources of circularly polarized light and the origin of homochirality. Orig Life Evol Biosph. 2001;31(1-2):167-183.[DOI]
-
24. Myrgorodska I, Meinert C, Hoffmann SV, Jones NC, Nahon L, Meierhenrich UJ. Light on chirality: Absolute asymmetric formation of chiral molecules relevant in prebiotic evolution. ChemPlusChem. 2017;82(1):74-87.[DOI]
-
25. Bocková J, Jones NC, Topin J, Hoffmann SV, Meinert C. Uncovering the chiral bias of meteoritic isovaline through asymmetric photochemistry. Nat Commun. 2023;14:3381.[DOI]
-
26. Chan QHS, Zolensky ME, Martinez JE, Tsuchiyama A, Miyake A. Magnetite plaquettes are naturally asymmetric materials in meteorites. Am Mineral. 2016;101(9):2041-2050.[DOI]
-
27. Pizzarello S, Zolensky M, Turk KA. Nonracemic isovaline in the Murchison meteorite: Chiral distribution and mineral association. Geochim Cosmochim Acta. 2003;67(8):1589-1595.[DOI]
-
28. 86th annual meeting of the meteoritical society (2024). Meteorit Planet Sci. 2024;59(S1):i-xlii.[DOI]
-
29. Chen Y, Ma W. The origin of biological homochirality along with the origin of life. PLoS Comput Biol. 2020;16(1):e1007592.[DOI]
-
30. Weller MG. The mystery of homochirality on earth. Life. 2024;14(3):341.[DOI]
-
31. Weber AL, Pizzarello S. The peptide-catalyzed stereospecific synthesis of tetroses: A possible model for prebiotic molecular evolution. Proc Natl Acad Sci U S A. 2006;103(34):12713-12717.[DOI]
-
32. Tamura K, Schimmel P. ChiraL-selective aminoacylation of an RNA minihelix. Science. 2004;305(5688):1253.[DOI]
-
33. Toxvaerd S. Origin of homochirality: The formation and stability of homochiral peptides in aqueous prebiological environment in the Earth’s crust. Symmetry. 2023;15(1):155.[DOI]
-
34. Rickard D, Luther GW. Chemistry of iron sulfides. Chem Rev. 2007;107(2):514-562.[DOI]
-
35. Novikov Y, Copley SD. Reactivity landscape of pyruvate under simulated hydrothermal vent conditions. Proc Natl Acad Sci U S A. 2013;110(33):13283-13288.[DOI]
-
36. Wang W, Li Q, Liu X, Yang Y, Su W. Enhanced photocatalytic performance of ZnS for reversible amination of α-oxo acids by hydrothermal treatment. Orig Life Evol Biosph. 2012;42(4):263-273.[DOI]
-
37. Cody GD, Boctor NZ, Filley TR, Hazen RM, Scott JH, Sharma A, et al. Primordial carbonylated iron-sulfur compounds and the synthesis of pyruvate. Science. 2000;289(5483):1337-1340.[DOI]
-
38. Li R, Deng Q, Han L, Ouyang T, Che S, Fang Y. Prebiotic formation of enantiomeric excess D-amino acids on natural pyrite. Nat Commun. 2024;15:10130.[DOI]
-
39. Crusats J, Moyano A. Absolute asymmetric catalysis, from concept to experiment: A narrative. Synlett. 2021;32(20):2013-2035.[DOI]
-
40. He C, Li Y. Absolute asymmetric synthesis driven by circularly polarized light. Chin Chem Lett. 2023;34(8):108077.[DOI]
-
41. Bloom BP, Lu Y, Metzger T, Yochelis S, Paltiel Y, Fontanesi C, et al. Asymmetric reactions induced by electron spin polarization. Phys Chem Chem Phys. 2020;22(38):21570-21582.[DOI]
-
42. Lv J, Gao X, Han B, Zhu Y, Hou K, Tang Z. Self-assembled inorganic chiral superstructures. Nat Rev Chem. 2022;6(2):125-145.[DOI]
-
43. Mörtberg L. Nonbiotic origin of optical activity. Nature. 1971;232(5306):105-107.[DOI]
-
44. Cronin JR, Pizzarello S. Enantiomeric excesses in meteoritic amino acids. Science. 1997;275(5302):951-955.[DOI]
-
45. Zhang W, Wang Y, Ma Y, Duan Y, Zhang W, Che S, et al. Enantioselective abiotic synthesis of ribose on chiral mesostructured hydroxyapatite. Angew Chem Int Ed. 2025;64(26):e202425581.[DOI]
-
46. Rickard D, Luther GW. Chemistry of iron sulfides. Chem Rev. 2007;107(2):514-562.[DOI]
-
47. Ray K, Ananthavel SP, Waldeck DH, Naaman R. Asymmetric scattering of polarized electrons by organized organic films of chiral molecules. Science. 1999;283(5403):814-816.[DOI]
-
48. Naaman R, Paltiel Y, Waldeck DH. Chiral molecules and the electron spin. Nat Rev Chem. 2019;3(4):250-260.[DOI]
-
49. Yang SH, Naaman R, Paltiel Y, Parkin SSP. Chiral spintronics. Nat Rev Phys. 2021;3(5):328-343.[DOI]
-
50. Zhang W, Ai J, Ouyang T, Yu L, Liu A, Han L, et al. Chiral nanostructured Ag films for multicarbon products from CO2 electroreduction. J Am Chem Soc. 2024;jacs.4c08445.[DOI]
-
51. Duan Y, Che S. Chiral mesostructured inorganic materials with optical chiral response. Adv Mater. 2023;35(51):2205088.[DOI]
-
52. Antonov VN, Germash LP, Shpak AP, Yaresko AN. Electronic structure, optical and X-ray emission spectra in FeS2. Phys Status Solidi B. 2009;246(2):411-416.[DOI]
-
53. Rosenberg RA, Abu Haija M, Ryan PJ. ChiraL-selective chemistry induced by spin-polarized secondary electrons from a magnetic substrate. Phys Rev Lett. 2008;101(17):178301.[DOI]
-
54. Metzger TS, Mishra S, Bloom BP, Goren N, Neubauer A, Shmul G, et al. The electron spin as a chiral reagent. Angew Chem Int Ed. 2020;59(4):1653-1658.[DOI]
-
55. Kumar A, Capua E, Kesharwani MK, Martin JML, Sitbon E, Waldeck DH, et al. Chirality-induced spin polarization places symmetry constraints on biomolecular interactions. Proc Natl Acad Sci U S A. 2017;114(10):2474-2478.[DOI]
-
56. Perdew JP, Burke K, Ernzerhof M. Generalized gradient approximation made simple. Phys Rev Lett. 1996;77(18):3865-3868.[DOI]
-
57. Kresse G, Joubert D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys Rev B. 1999;59(3):1758-1775.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite