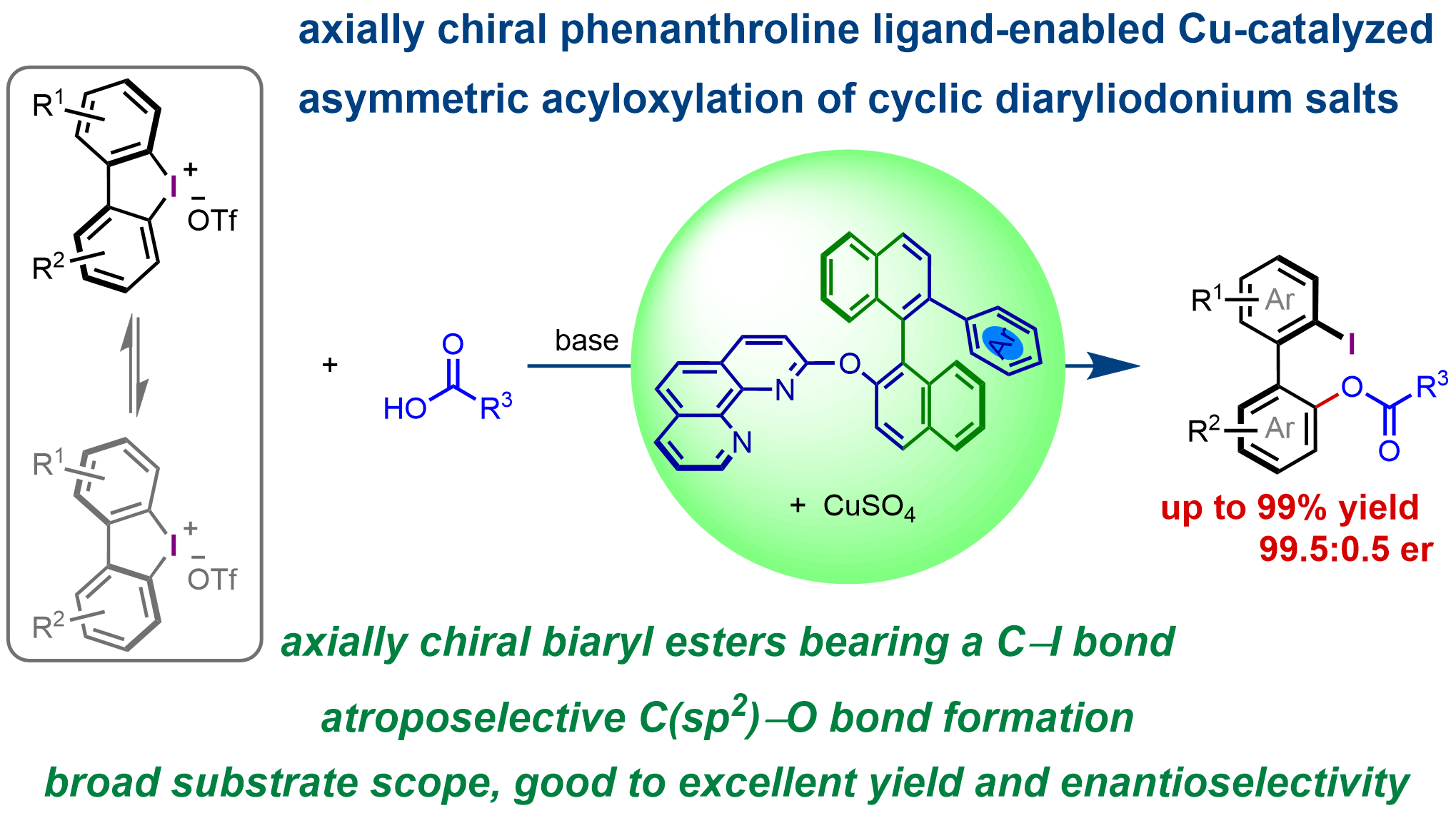
Abstract
Chiral ligands play a crucial role in enantioselective transition-metal catalysis, not only by coordinating to metal centers and modulating their electronic properties, but also by providing the essential chiral environment for efficient enantiodiscrimination. For a given reaction, high enantioselectivities can often be achieved with different chiral ligands, offering valuable enantioinduction models, even when these ligands possess distinct structures and chiral environments. Herein, we report that a class of conformationally flexible C1-symmetric axially chiral phenanthroline ligands enables the formation of chiral copper catalysts capable of promoting the highly enantioselective ring-opening acyloxylation of cyclic biaryliodonium salts. Using this approach, a range of valuable axially chiral biaryl compounds bearing an ester group and a carbon–iodine bond were synthesized in good to excellent yield (up to 99%), with good to excellent enantioselectivity (up to 99.5:0.5 er).
Graphical Abstract
Keywords
1. Introduction
The connection of two aryl units through a sigma bond gives biaryl compounds, which are usually classified as atropisomers or axially chiral compounds if the rotational barrier of the biaryl units at room temperature exceeds 22 kcal/mol (corresponding to a half-life of greater than 1,000 seconds)[1-3]. Axially chiral biaryl compounds, particularly those bearing diverse functional groups, are prevalent in biologically active compounds, functional materials, and chiral catalysts[1-14]. Biaryl-bridged five- to seven-membered cyclic compounds are generally recognized as achiral due to the reduced rotational barriers of their biaryl moieties. In the presence of suitable chiral catalysts, the ring-opening of these normal-sized rings via specific chemical reactions affords axially chiral biaryl compounds. Consequently, the asymmetric ring-opening of conformationally labile biaryl-bridged cyclic compounds provides a very powerful strategy for the synthesis of a wide range of highly useful axially chiral biaryl compounds equipped with various functional groups (Figure 1A)[15-41]. Cyclic biaryliodonium salts contain biaryl-bridged iodine-containing normal-sized rings and typically function as electrophiles. Pioneered by Gu and co-workers, the asymmetric ring-opening of cyclic biaryliodonium salts with various nucleophiles has enabled the synthesis of a series of functionalized biaryls bearing a synthetically valuable carbon–iodine bond (Figure 1B)[26-41]. Copper catalysts derived from classic chiral bis(oxazoline) (Box) and pyridine-bis(oxazoline) ligands have proven to be highly efficient and enantioselective for the asymmetric ring-opening of cyclic biaryliodonium salts with diverse nucleophiles. Gu and co-workers also developed a Cu(I)-anionic chiral cobalt(III) catalyst[40,41] and a Cu(I)-coplanar atropochiral 5H-cyclopenta[2,1-b:3,4-b']dipyridine catalyst[30] for the atroposelective ring-opening of cyclic biaryliodonium salts with bulky anilines and bulky secondary amines, respectively (Figure 1B).
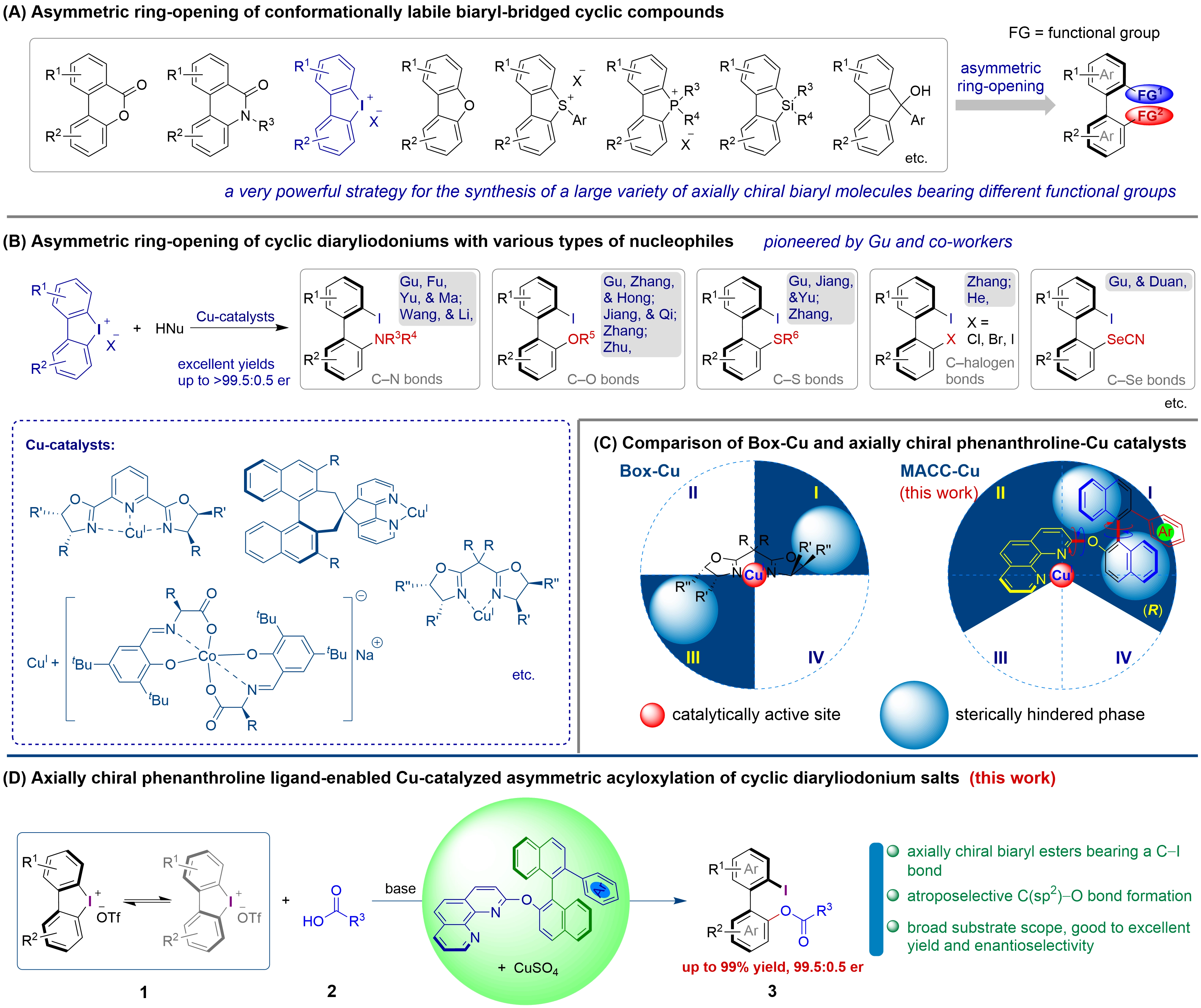
Figure 1. Backgrounds and axially chiral phenanthroline ligand-enabled Cu-catalyzed asymmetric acyloxylation of cyclic biaryliodonium salts.
We have developed a novel type of axially chiral phenanthroline ligands featuring highly tunable binaphthyl chiral units and applied them in the enantioselective synthesis of 1,1'-binaphthyl-2,2'-diamine, 2-amino-2'-hydroxy-1,1'-binaphthyl derivatives[42], axially chiral biaryl δ-amino acid derivatives[17], as well as in the amination and alkylation of allyl acetates and Morita–Baylis–Hillman adducts[43,44]. Regio- and enantioselective ring-opening alkynylation and amination of cyclic biaryliodonium salts have also been achieved through the development of conformationally switchable axially chiral phenanthroline-copper catalysts[45]. The Box-Cu catalyst is structurally rigid due to the formation of a metallacycle upon coordination. In contrast, the axially chiral phenanthroline-Cu catalyst is more flexible, owing to the rotations around several single bonds (Figure 1C). High enantioselectivities have been achieved in the ring-opening of cyclic biaryliodonium salts with various nucleophiles using either type of catalyst, despite their substantial differences in structural rigidity, conformation, and chiral environment. However, whether an axially chiral phenanthroline-Cu catalyst is capable of promoting the asymmetric acyloxylation of cyclic biaryliodonium salts with high enantioinduction remains unknown. Chiral ligands play a crucial role in enantioselective transition-metal catalysis, and studies on asymmetric transformations with different catalytic systems provide valuable insights into catalytic and enantioinduction models. Herein, we investigated the asymmetric acyloxylation of cyclic biaryliodonium salts using an axially chiral phenanthroline-Cu catalyst. Highly enantioselective ring-opening of cyclic biaryliodonium salts with carboxylic acids was achieved using a copper catalyst formed in situ from an axially chiral phenanthroline ligand bearing a bulky aryl group at the 2'-position of the binaphthyl backbone, affording a range of axially chiral biaryl esters containing a carbon–iodine bond in up to 99% yield with enantiomeric ratios (er) up to 99.5:0.5 (Figure 1D).
2. Experimental
Detailed experimental procedures can be found in the Supplementary materials.
3. Results and Discussion
To explore a new axially chiral phenanthroline-Cu catalyst for the asymmetric ring-opening of cyclic biaryliodonium salts with carboxylic acid, substrates 1a and 2a were selected as model reactants. A range of chiral phenanthroline ligands were evaluated under preliminarily optimized reaction conditions (Figure 2). When ligand L1, bearing a phenyl group at the 2'-position of the binaphthyl backbone, was employed, the desired product 3a was obtained in 81% yield with an enantiomeric ratio (er) of 92:8. However, the introduction of a 3,5-dimethylphenyl group at the C9 position of the phenanthroline unit (L2) led to a dramatic decrease in both yield and enantioselectivity, possibly due to steric hindrance that reduces the accessibility of the substrates to the catalytically active copper center. Ligand L3, featuring a 4-methylphenyl group at the 2'-position of the binaphthyl backbone, provided comparable yield and enantioselectivity to L1. Replacement of the methyl group in L3 with a trifluoromethyl group (L4) resulted in a slight decrease in enantioselectivity. Ligands L5–L7, bearing para-substituted phenyl groups at the 2'-position, were also tested. With L5 as ligand, although the er improved to 95.5:4.5, the yield decreased to 62%. L6 and L7 did not provide better yields or enantioselectivities compared to L1. Several ligands with a meta-substituted phenyl group at the 2'-position were subsequently evaluated. With L8, which bears a meta-methylphenyl group, the desired product was obtained in 80% yield with 95.5:4.5 er. Replacement of the methyl group in L8 with a tert-butyl group (L9) led to a decrease in yield, although the enantioselectivity was maintained. L10 exhibited comparable yield and a slightly lower er. When ligand L11, bearing a 3,5-dimethylphenyl group at the 2'-position of the binaphthyl backbone, was used, the acyloxylation product was obtained in 82% yield with 95:5 er. An excellent yield (99%) was obtained with ligand L12, which contains a bulky 2,4,6-trimethylphenyl group at the 2'-position; however, the enantioselectivity obtained was moderate. Replacement of the methyl group in L11 with a tert-butyl group (L13) resulted in decreased yield and er. Interestingly, when L14, bearing a 3,5-diphenylphenyl group at the 2'-position of the binaphthyl backbone, was employed, the product was obtained in 99% yield with 97:3 er. Ligand L15, bearing a very bulky aryl group, did not provide improved yield or enantioselectivity. Therefore, L14 was identified as the optimal ligand. These results also indicated that the substituent at the 2'-position of the binaphthyl backbone significantly influences ligand performance, even though it is expected to point away from the catalytically active center due to steric considerations.
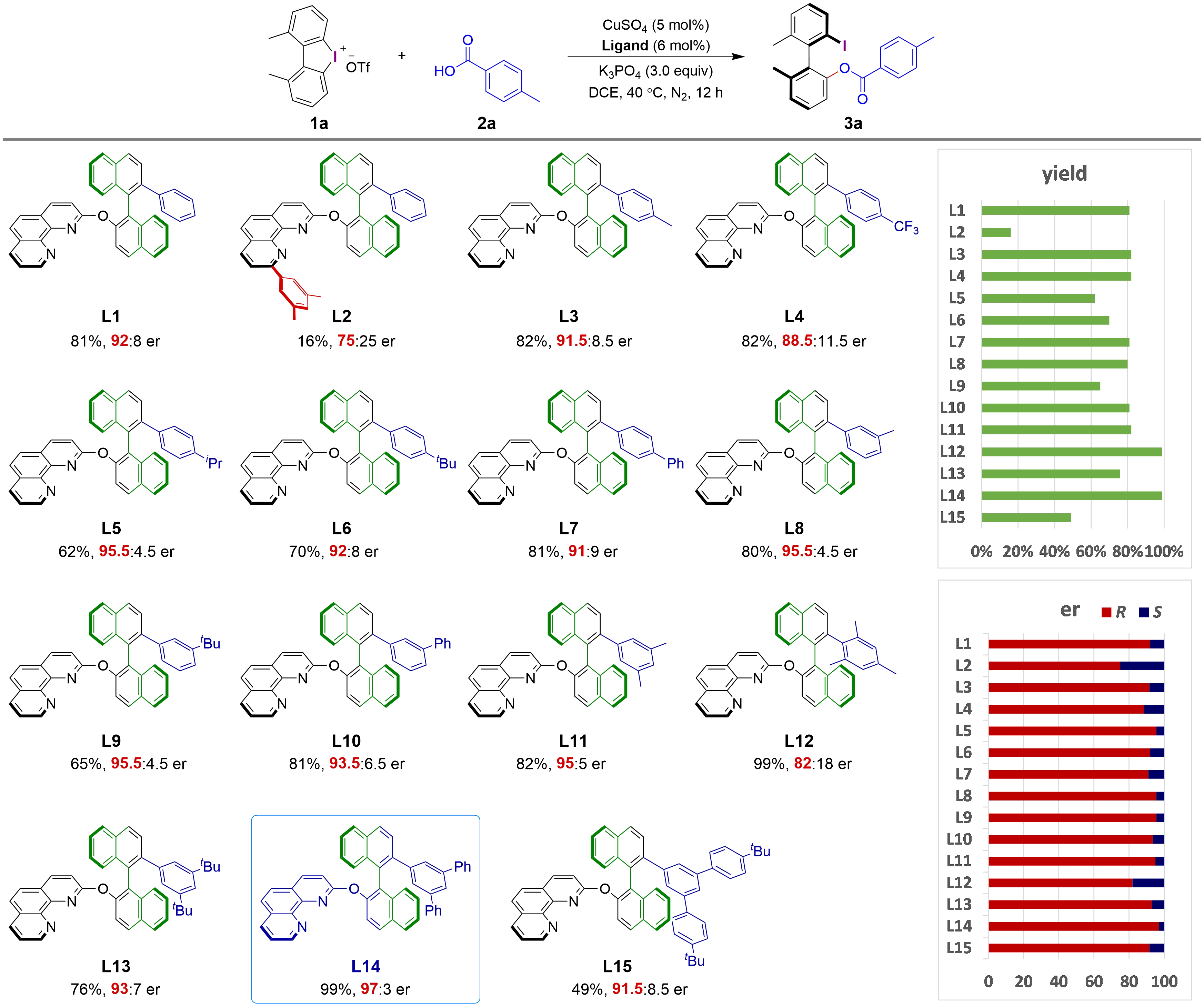
Figure 2. Ligand evaluation for Cu-catalyzed asymmetric acyloxylation of cyclic biaryliodonium saltsa. Reaction conditions: 1a (0.1 mmol), 2a (0.12 mmol), CuSO4 (5 mol%), Ligand (6 mol%), K3PO4 (3.0 equiv.), in DCE (4.0 mL) at 40 °C for 12 h. Yield of the isolated product. Enantiomeric ratios (er) were determined via HPLC analysis on a chiral stationary phase and reported as ratios of (R:S). DCE: 1,2-dichloroethane; HPLC: high-performance liquid chromatography.
With the optimal axially chiral phenanthroline ligand L14 in hand, we next explored the substrate scope of the asymmetric acyloxylation of cyclic biaryliodonium salts (Figure 3). When benzoic acid was employed as the nucleophile to react with cyclic biaryliodonium salt 1a, the corresponding product 3b was obtained in 99% yield with 98.5:1.5 er. Substituted benzoic acids bearing a strong electron-withdrawing trifluoromethyl group or an electron-donating methoxy group were well tolerated, affording axially chiral products 3c and 3d in 99% yield with 97:3 and 97.5:2.5 er, respectively. Sterically more hindered para-phenylbenzoic acid reacted with 1a more slowly to deliver 3e in excellent yield (99%), albeit with a relatively lower er. Acyloxylation of 1a with para-acetylbenzoic acid afforded 3f in 98% yield with 97:3 er. The catalytic system was compatible with para-halogenated (fluoro, chloro, bromo, and iodo) benzoic acids, as well as meta- and ortho-bromobenzoic acids, affording the corresponding halogenated axially chiral biaryls 3g-3l in 96-99% yields with excellent er values (ranging from 96.5:3.5 to 98.5:1.5). Both 1-naphthalenecarboxylic acid and 2-naphthalenecarboxylic acid were tolerated, delivering 3m and 3n in excellent yield with 97:3 and 97.5:2.5 er, respectively. The more challenging quinoline-2-carboxylic acid was also tested; in the presence of a nickel salt, product 3o was obtained in 33% yield with 91:9 er after 36 hours. When pyrrole-2-carboxylic acid was used as the nucleophile, the pyrrole-containing 3p was obtained in 62% yield with 96.5:3.5 er. Furan-2-carboxylic acid and thiophene-2-carboxylic acid reacted with the cyclic biaryliodonium salt to produce heterocyclic axially chiral biaryls 3q and 3r in 94-99% yields with 98:2 er. Aliphatic carboxylic acids were also well tolerated under these conditions, affording the corresponding products 3s-3w in 98-99% yields with excellent er values (ranging from 95.5:4.5 to 99:1). A cyclic biaryliodonium salt bearing two additional methyl groups reacted with benzoic acid to afford 3x in 70% yield with moderate er, while its reaction with para-methylbenzoic acid delivered 3y in 99% yield with 92:8 er. A cyclic biaryliodonium salt bearing a methyl or chloro group at the ortho position relative to the carbon-iodine bond reacted smoothly with benzoic acid to afford 3z and 3aa in 72-77% yields with good er. Phenyl-naphthyl biaryl 3ab was synthesized in 75% yield, albeit with a relatively lower er. A cyclic biaryliodonium salt bearing two additional methyl groups at the meta positions relative to the carbon-iodine bonds was tested with benzoic acid, para-methoxybenzoic acid, para-bromobenzoic acid, and 1-adamantanecarboxylic acid as nucleophiles, delivering the corresponding biaryls 3ac-3af in 63-99% yields with er values up to 99.5:0.5. The reaction of a cyclic biaryliodonium salt bearing two additional methyl groups at the para positions relative to the carbon–iodine bonds with aromatic and aliphatic carboxylic acids also proceeded smoothly, affording the corresponding products 3ag-3aj in excellent yields (up to 99%) with er values ranging from 90.5:9.5 to 94.5:5.5. In addition, a scaled-up reaction was performed under the standard conditions with 1a (456 mg, 1.0 mmol) as substrate, delivering the desired product 3a in 99% yield with 97:3 er after 48 hours (Section S4).
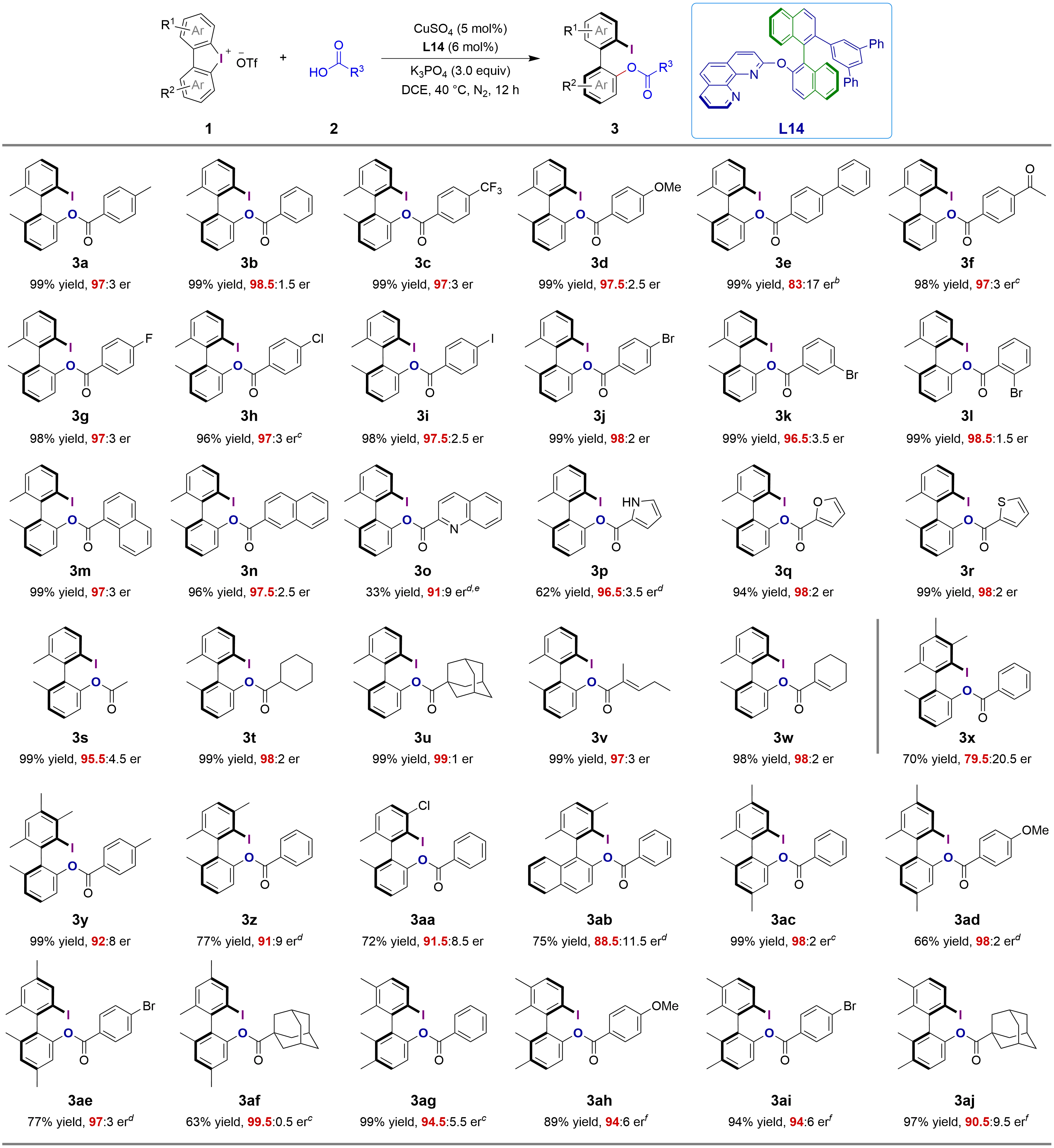
Figure 3. Substrate scope of Cu-catalyzed asymmetric acyloxylation of cyclic biaryliodonium saltsa. Reaction conditions (unless otherwise noted): 1 (0.1 mmol), 2 (0.12 mmol), CuSO4 (5 mol%), L14 (6 mol%) and K3PO4 (3.0 equiv) in DCE (4.0 mL) at 40 °C for 12 h; Yield of the isolated product. Enantiomeric ratios (er) were determined via HPLC analysis on a chiral stationary phase and reported as ratios of (R:S). b: reaction time, 62 h; c: reaction time, 24 h; d: reaction time, 36 h; e: NiCl2(dppe) (10 mol%). f: reaction time, 7 d. DCE: 1,2-dichloroethane; HPLC: high-performance liquid chromatography.
Synthetic transformations of 3a were also conducted to demonstrate the synthetic applications of the obtained products (Figure 4). Hydrolysis of 3a under basic conditions afforded the axially chiral biaryl phenol 4 in 96% yield with retained enantiopurity (97:3 er). The carbon-iodine bond in 4 can be further transformed to phenolic hydroxy group, delivering axially chiral biphenol 5 in 68% yield. Suzuki-Miyaura coupling of 3a with pyridine-3-boronic acid afforded axially chiral biaryl phenol 6. Furthermore, 3a could also be readily converted to a phosphorus-containing chiral biaryl phenol 7 in one step with retained enantiopurity (see Section S6 for details).
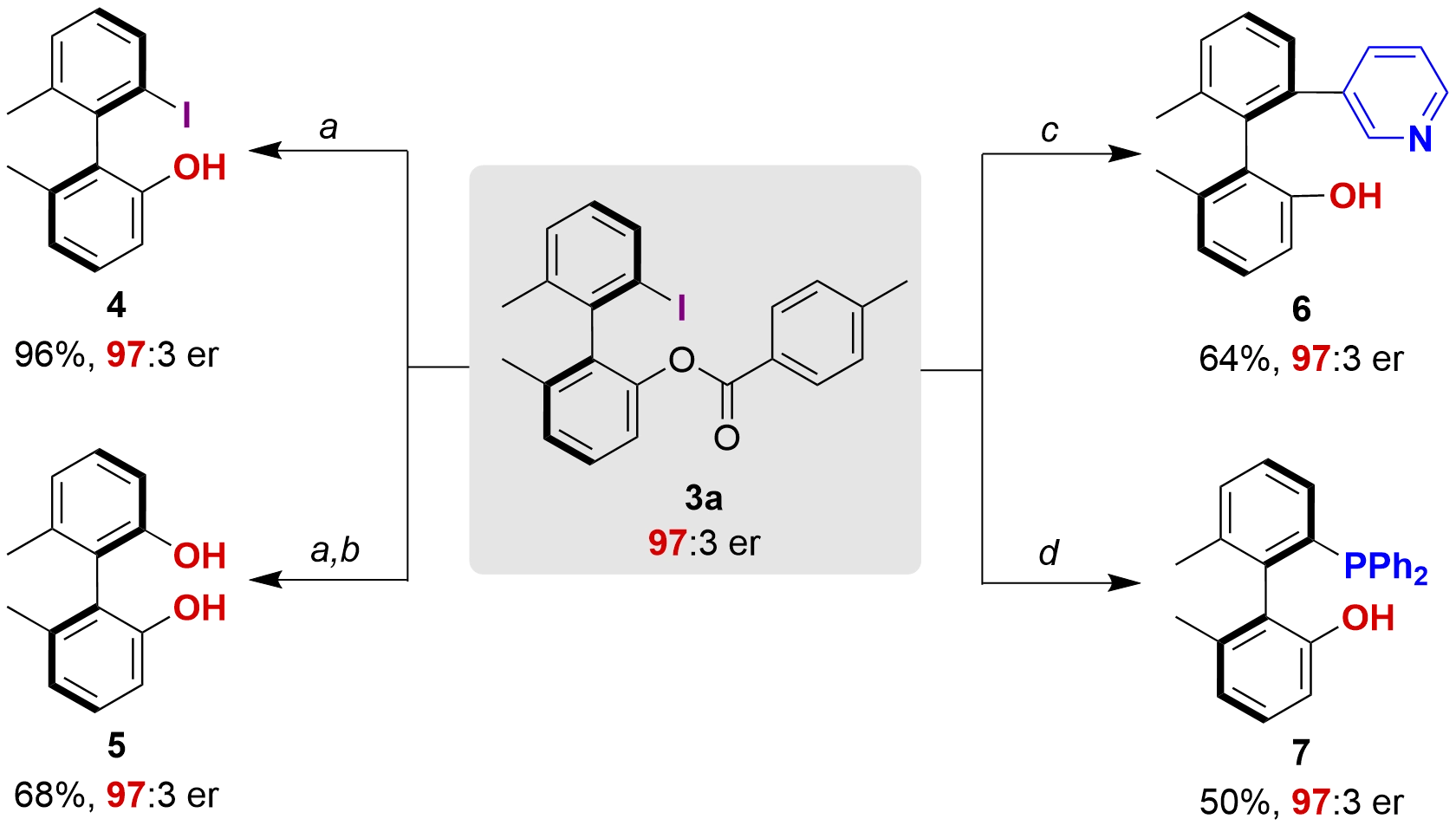
Figure 4. Synthetic transformations of the product 3a. a: LiOH, THF/MeOH/H2O, 45 °C; b: Cu(OAc)2, D-glucose, KOH, DMSO/H2O, 110 °C; c: pyridine-3-boronic acid, Pd(PPh3)4, K2CO3, DMF, 110 °C; d: diphenylphosphine, CuI, Cs2CO3, toluene, 100 °C. Yield of the isolated product. Enantiomeric ratios (er) were determined via HPLC analysis on a chiral stationary phase and reported as ratios of (R:S). DMF: dimethylformamide; HPLC: high-performance liquid chromatography.
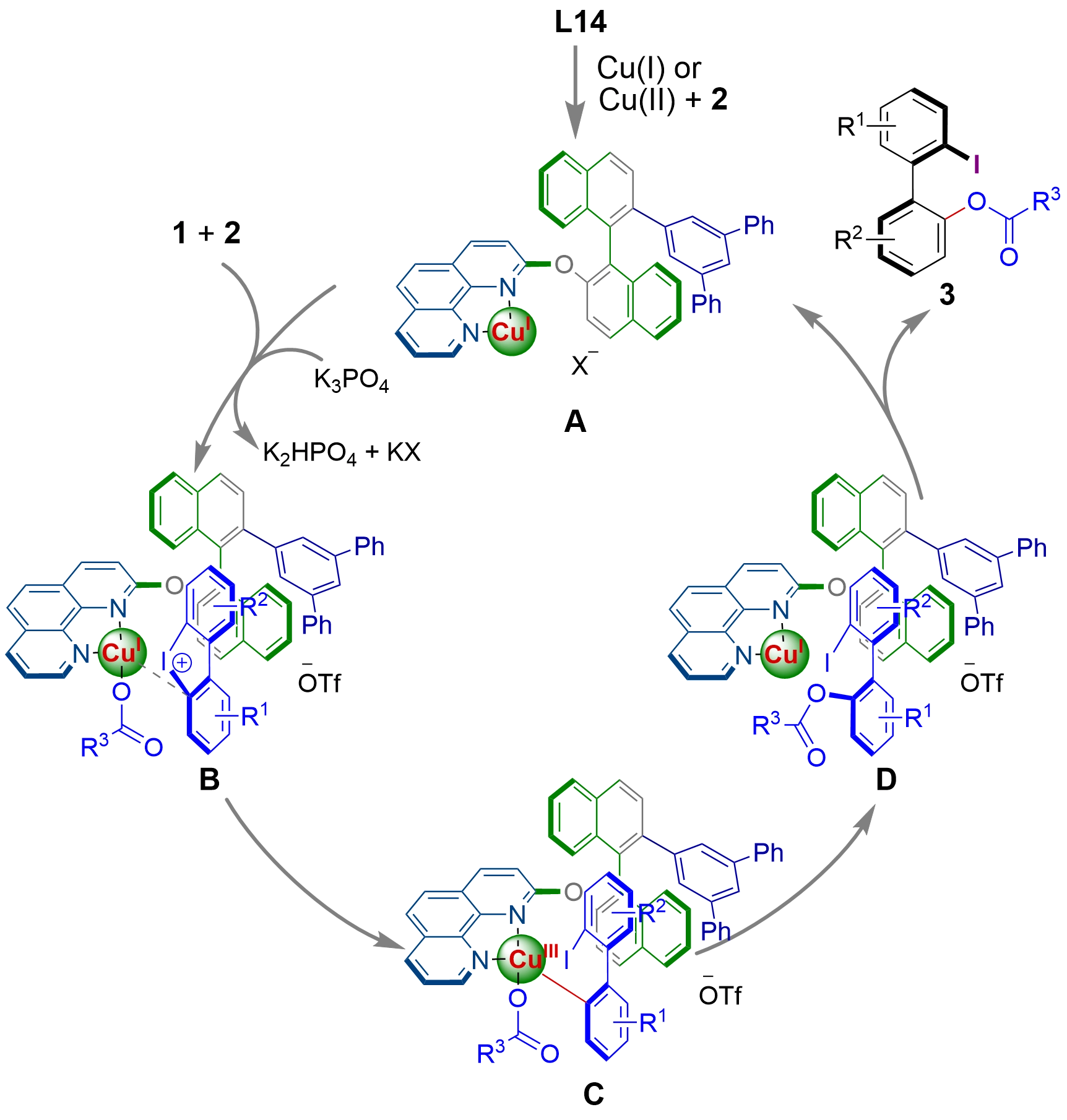
A plausible catalytic cycle for the axially chiral phenanthroline ligand-enabled copper-catalyzed asymmetric acyloxylation of cyclic biaryliodonium salts is illustrated in Figure 5, on the basis of the results obtained in this work and the reported mechanisms for the ring-opening of cyclic biaryliodoniums[26-41,45]. Ligand L14, bearing a bulky aryl group at the 2'-position of the binaphthyl unit, reacted with copper salts under reaction conditions to generate a catalytically active copper species A. The interaction of catalyst A with the reactants in the presence of a base affords intermediate B, which then undergoes oxidative addition of Cu(I) to the C–I bond to give Cu(III) intermediate C. Reductive elimination of the intermediate C produces the desired acyloxylation product and regenerates the catalyst.
4. Conclusion
In summary, a C1-symmetric axially chiral phenanthroline-Cu catalyst, exhibiting significantly different structural rigidity, conformation, and chiral environment compared to the classic Box-Cu catalyst, has been developed for the enantioselective ring-opening acyloxylation of cyclic biaryliodonium salts. A range of axially chiral biaryl compounds bearing an ester substituent and a carbon–iodine bond were synthesized in good to excellent yields (up to 99%), with good to excellent enantioselectivities (up to 99.5:0.5 er).
Supplementary materials
The supplementary material for this article is available at: Supplementary materials.
Acknowledgements
We thank the Research Center of Analysis and Test, East China University of Science and Technology for the help with the characterization.
Authors contribution
Zhang Z: Conceptualization, supervision, formal analysis, writing-original draft.
Cen S, Ma Y, Liu Y: Methodology, investigation, formal analysis, writing-review & editing.
Lin M, Yu Y: Investigation, writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
The data reported in this paper are available in the main text or Supplementary Information, including methods, detailed experimental procedures, characterization data, and copies of NMR and HPLC spectra.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 22371072, 22571082 and 22501081), Natural Science Foundation of Shanghai (Grant No. 24ZR1415000), Fundamental Research Funds for the Central Universities (Grant Nos. 222201717003, JKVJ1251010, SLJ13233303 and YJ0142232), Science and Technology Commission of Shanghai Municipality (Grant No. 24DX1400200), and the Program of Introducing Talents of Discipline to Universities (Grant No. B16017).
Copyright
© The Author(s) 2026.
References
-
1. Ōki M. Recent advances in atropisomerism. In: Allinger NL, Eliel EL, Wilen SH, editors. Topics in stereochemistry. Hoboken: John Wiley & Sons, Inc.; 1983. p. 1-81.[DOI]
-
4. Kozlowski MC, Morgan BJ, Linton EC. Total synthesis of chiral biaryl natural products by asymmetric biaryl coupling. Chem Soc Rev. 2009;38(11):3193.[DOI]
-
5. Bringmann G, Gulder T, Gulder TAM, Breuning M. Atroposelective total synthesis of axially chiral biaryl natural products. Chem Rev. 2011;111(2):563-639.[DOI]
-
7. Cheng JK, Xiang SH, Li S, Ye L, Tan B. Recent advances in catalytic asymmetric construction of atropisomers. Chem Rev. 2021;121(8):4805-4902.[DOI]
-
10. Xiang SH, Ding WY, Wang YB, Tan B. Catalytic atroposelective synthesis. Nat Catal. 2024;7(5):483-498.[DOI]
-
14. Takizawa S, Salem M. Atropisomerism in asymmetric organic synthesis: Challenges and applications. Weinheim: Wiley-VCH GmbH; 2024.[DOI]
-
16. Cai Y, Zhao Y, Tang K, Zhang H, Mo X, Chen J, et al. Amide C–N bonds activation by A new variant of bifunctional N-heterocyclic carbene. Nat Commun. 2024;15:496.[DOI]
-
17. Cen S, Li SS, Zhao Y, Zhao MX, Zhang Z. Catalytic asymmetric synthesis of unnatural axially chiral biaryl δ-amino acid derivatives via a chiral phenanthroline-potassium catalyst-enabled dynamic kinetic resolution. Angew Chem Int Ed. 2024;63(36):e202407920.[DOI]
-
19. Zhang Z, Zhang J, Gao Q, Zhou Y, Yang M, Cao H, et al. Enantioselective alkylative cross-coupling of unactivated aromatic C–O electrophiles. Nat Commun. 2022;13:2953.[DOI]
-
25. Deng R, Xi J, Li Q, Gu Z. Enantioselective carbon-carbon bond cleavage for biaryl atropisomers synthesis. Chem. 2019;5(7):1834-1846.[DOI]
-
26. Zhang X, Zhao K, Gu Z. Transition metal-catalyzed biaryl atropisomer synthesis via a torsional strain promoted ring-opening reaction. Acc Chem Res. 2022;55(12):1620-1633.[DOI]
-
27. Zhao K, Duan L, Xu S, Jiang J, Fu Y, Gu Z. Enhanced reactivity by torsional strain of cyclic diaryliodonium in Cu-catalyzed enantioselective ring-opening reaction. Chem. 2018;4(3):599-612.[DOI]
-
30. Yang H, Sun X, Chen Z, Jiang C, Duan L, Zhao J, et al. Coplanar atropochiral 5H-cyclopenta [2,1-b:3,4-b'] dipyridine ligands: Synthesis and applications in asymmetric ring-opening reaction. Angew Chem Int Ed. 2025;64(4):e202416839.[DOI]
-
31. Duan L, Zhao K, Wang Z, Zhang FL, Gu Z. Enantioselective ring-opening/oxidative phosphorylation and P-transfer reaction of cyclic diaryliodoniums. ACS Catal. 2019;9(11):9852-9858.[DOI]
-
32. Zhu K, Xu K, Fang Q, Wang Y, Tang B, Zhang F. Enantioselective synthesis of axially chiral biaryls via Cu-catalyzed acyloxylation of cyclic diaryliodonium salts. ACS Catal. 2019;9(6):4951-4957.[DOI]
-
34. Kang B, Wang L, Sun X, Liu H, Wen Z, Ren Y, et al. Enantioselective synthesis of axially chiral carbamates and amides with carbon dioxide via copper catalysis. Org Chem Front. 2023;10(20):5231-5241.[DOI]
-
39. Singh PR, Banerjee A, Simlandy AK. Advances in catalytic enantioselective transformations using diaryliodonium reagents. ACS Catal. 2025;15(4):3096-3115.[DOI]
-
42. Cen S, Huang N, Lian D, Shen A, Zhao MX, Zhang Z. Conformational enantiodiscrimination for asymmetric construction of atropisomers. Nat Commun. 2022;13:4735.[DOI]
-
45. Liu Y, Ma Y, Liu PZ, Shen C, Cen S, Zhu YY, et al. Switchable phenanthroline-Cu catalysts for enantioselective alkynylation and amination of cyclic diaryliodonium salts. Angew Chem Int Ed. 2026;65(2):e14745.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite