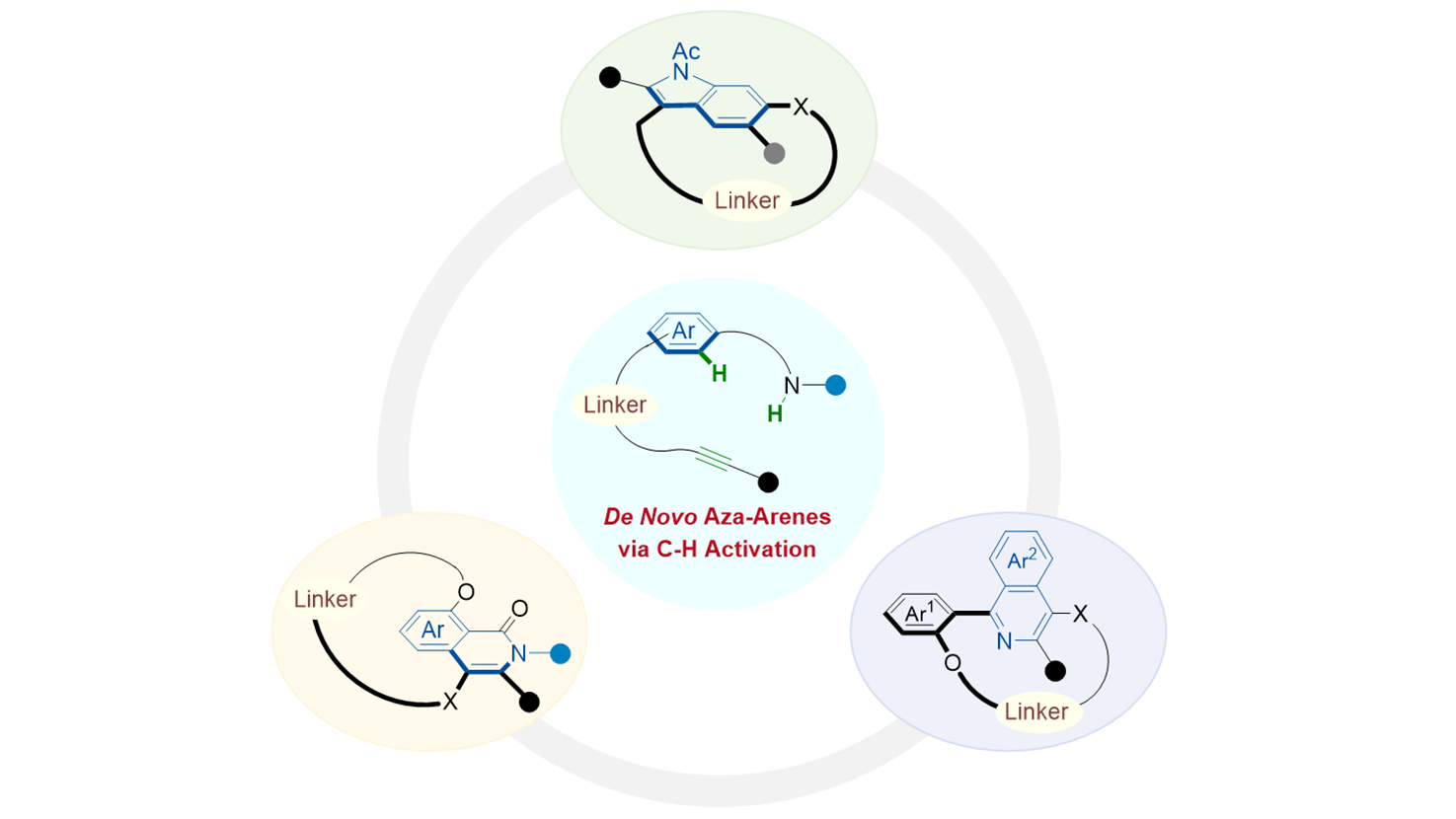
Graphical Abstract
1. Introduction
Planar-chiral macrocycles, characterized by an aromatic ring bridged by an ansa chain, have captivated synthetic chemists for decades owing to their unique stereoelectronic properties and structural complexity. These intriguing chiral architectures are not only embedded in biologically active natural products and pharmaceuticals, but also serve as privileged scaffolds in chiral ligands, catalysts, and functional materials (Figure 1A)[1-5]. Consequently, the development of efficient catalytic methods for atroposelective synthesis of structurally diverse planar-chiral macrocycles has emerged as a topic of significant contemporary interest. Driven by the rapid development of asymmetric catalysis, four catalytic asymmetric synthetic strategies have been established to date (Figure 1B)[6-12]: desymmetrization or (dynamic) kinetic resolution of prochiral or racemic performed cyclophanes via editing of the aromatic ring or ansa chain, and enantioselective macrocyclization involving the formation of either the aromatic ring or ansa chain.
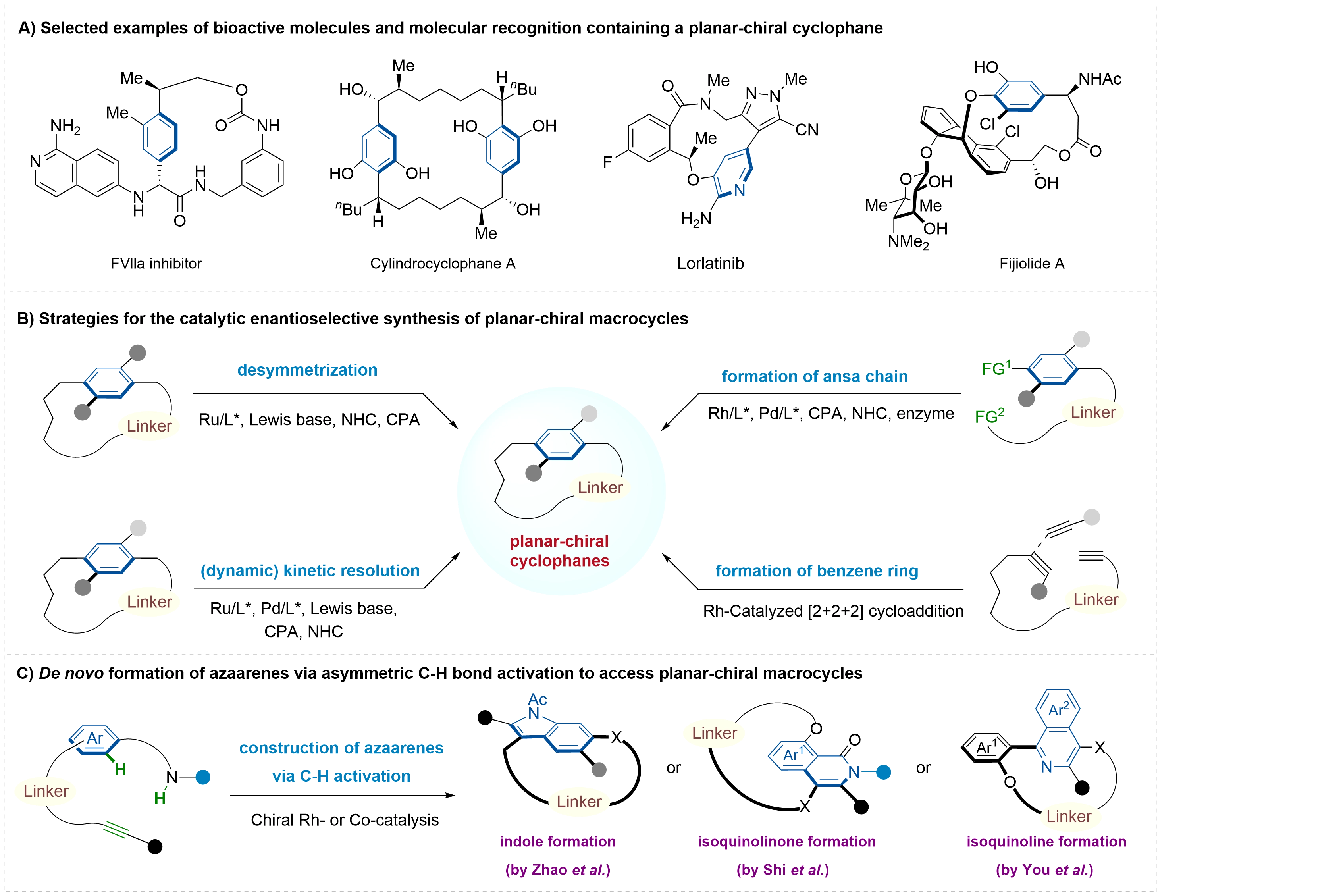
Figure 1. The selected applications and synthetic strategies of planar-chiral macrocycles.
Among these, direct enantioselective macrocyclization, which merges macrocycle formation with the establishment of planar chirality in a single operation, represents the most atom-economical and conceptually attractive approach. Nevertheless, this strategy is impeded by two major challenges: the entropic penalty associated with folding linear precursors into macrocyclic structures often results in low efficiency and competing intermolecular pathways; additionally, achieving stereocontrol during ring closure is complicated by both the conformational flexibility of the linear substrates and the potential racemization of the macrocyclic products[13,14]. Despite these challenges, a variety of elegant methodologies based on catalytic enantioselective macrocyclization have emerged in the past two decades. In contrast to the well-established enantioselective construction of the ansa chain, the de novo formation of the aromatic ring to access planar chirality remains largely unexplored, primarily due to the inherent difficulty in simultaneously controlling regio- and enantioselectivity. In 2007, the Tanaka group pioneered the highly enantioselective synthesis of planar-chiral metacyclophanes via de novo benzene ring formation, employing a cationic rhodium(I)/(R)-H8-BINAP complex-catalyzed [2+2+2] cycloaddition of three alkyne units[15,16]. Shortly thereafter, Shibata and coworkers reported an intramolecular enantioselective [2+2+2] cycloaddition of triynes catalyzed by a cationic chiral Rh-Me-DUPHOS complex, enabling the synthesis of planar-chiral tripodal cage compounds with up to 99% ee[17]. Since then, this de novo benzene ring construction strategy has been extended to the enantioselective synthesis of other planar-chiral cyclophanes, including carba-paracyclophanes[18], bent cyclophanes[19], and [2.2]triphenylenophanes[20]. While great advances have been made, the development of de novo aromatic ring construction pathways to access diverse planar-chiral macrocycles remains a highly desirable yet largely unmet challenge.
In this context, with the flourishing of asymmetric C–H functionalization chemistry, the groups of Zhao[21], Shi[22], and You[23] have independently achieved the enantioselective construction of planar chiral macrocycles by employing transition metal-catalyzed C–H activation/annulation strategies via de novo formation of aza-aromatic rings, including indole[24,25], isoquinoline, and isoquinolinone (Figure 1C). This highlight summarizes these latest advances, discusses the reaction mechanism, advantages and limitations. Furthermore, it outlines the synthetic opportunities and promising directions that remain open for further exploration in this evolving field.
2. De Novo Aza-Arenes Construction for Catalytic Atroposelective Synthesis of Planar-Chiral Macrocycles
Two recent independent studies by the groups of Zhao and You constitute significant progress in the catalytic enantioselective synthesis of planar-chiral macrocycles, achieved through de novo construction of indoles and isoquinoline rings using a Rh-catalyzed intramolecular asymmetric sequential C–H activation/annulation of aniline- or imine-tethered internal alkynes. In 2024, Zhao and coworkers reported the first intramolecular Rh(III)-catalyzed highly atroposelective macrocyclization of aniline-tethered internal alkynes 1, enabling the synthesis of a diverse array of indole-based planar-chiral macrocycles 2 via the indole synthesis pathway (Figure 2A)[21]. By employing 2.5 mol% of You’s chiral spiro-CpRh(III) catalyst (S)-Rh-1[26,27], along with 20 mol% of AgSbF6 as an additive and Cu(OAc)2 as an oxidant, a broad range of indole-based 10- to 18-membered macrocyclic atropisomers 2, including those featuring challenging ones bearing full-carbon ansa chains, were obtained with high to excellent enantioselectivities (75-99% ee). Investigation of substrate scope and conformational stability revealed that both the alkyl chain length and the substituent size are critical for stabilizing macrocyclic atropisomers. This transformation accomplished the construction of the indole core, macrocyclization, and planar chirality control in a single step. Mechanistic studies and density functional theory (DFT) calculations supported a pathway involving Rh(III)-catalyzed C–H activation of aniline, followed by alkyne insertion. The observed stereoselectivity was collectively governed by π–π interactions during the C–H activation step and by minimized steric repulsion in both the C–H activation and intramolecular alkyne insertion events.
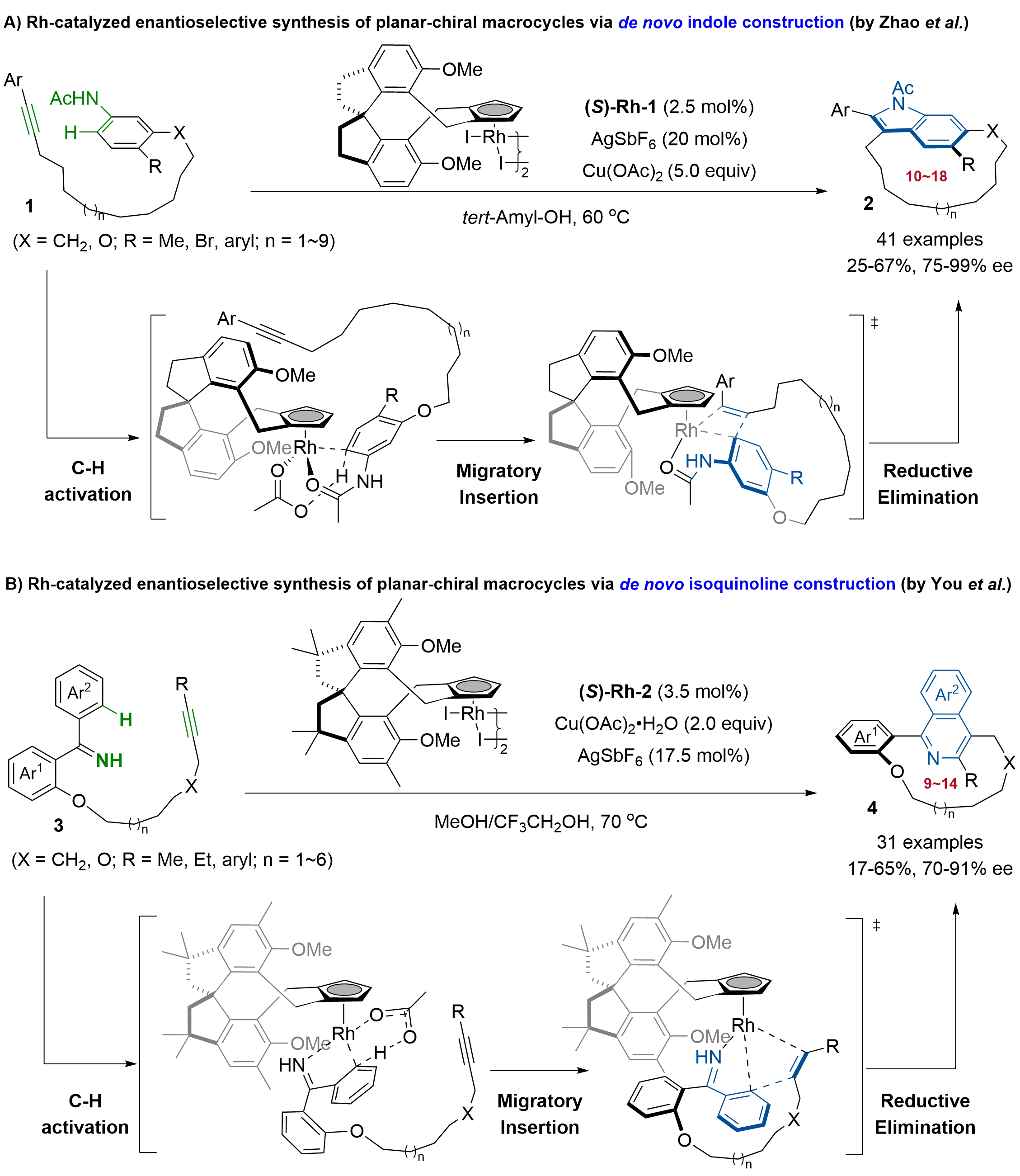
Figure 2. Rh-catalyzed asymmetric synthesis of planar-chiral macrocycles via de novo formation of indole or isoquinoline.
Later, You and co-workers achieved the first enantioselective synthesis of isoquinoline-based planar-chiral macrocyclic atropisomers via de novo formation of the isoquinoline core by a Rh catalyzed asymmetric sequential C–H activation/[4+2] annulation of alkyne-tethered imines (Figure 2B)[23]. The use of their previously developed BCSCpRh catalyst (S)-Rh-2[26] (3.5 mol%), AgSbF6 (17.5 mol%), and Cu(OAc)2·H2O (3.0 equiv) proved to be optimal, which efficiently enabled the intramolecular asymmetric annulation of various alkyne-tethered diaryl ketimines 3, furnishing the corresponding isoquinoline-based 9- to 14-membered planar-chiral macrocycles 4 with 70-91% ee. The synthetic utility of this method was further demonstrated by large-scale synthesis and product elaboration without erosion of enantiopurity. Mechanistic studies, including kinetic isotope effect experiments and DFT calculations, identified the concerted metalation deprotonation step involved in C–H activation of the benzene ring as the rate-determining step, while the migratory insertion of the alkyne moiety into the Rh-C bond is the key enantio-discrimination step in the catalytic cycle.
In parallel to chiral Rh(III)-catalyzed C–H functionalization, Shi and co-workers recently disclosed a cobalt-catalyzed enantioselective macrocyclization that assembles planar-chiral cyclophanes via an intramolecular C–H/N–H [4+2] annulation to form the aromatic isoquinolinone ring (Figure 3).[22] Under the optimized conditions employing Co(OAc)2·4H2O (10 mol%), Salox ligand 6 (20 mol%), Ag₂CO3 (2 equiv), and PivONa·2H2O (2 equiv), a diverse range of linear alkynes 5 bearing a bidentate 8-aminoquinoline directing group underwent smooth macrocyclization. This process efficiently delivered the desired isoquinolinone-containing planar-chiral cyclophanes 7, with ring sizes ranging from 12- to 24-membered, in good to excellent yields and with excellent enantioselectivities. The authors proposed that an in situ generated chiral Salox−cobalt(III) complex templated the linear alkyne into a cyclization-competent chiral conformation, mitigating the entropic penalty and ensuring high facial selectivity. Specifically, the linear substrate underwent a stereoselective self-assembly via C–H metalation with the in situ generated active Co(III) species, forming a key pincer-type cobaltacycle intermediate. This intermediate then preferentially underwent enantio-determining exo-migratory insertion from the sterically less hindered terminus of the alkyne, followed by reductive elimination to deliver the chiral cyclophane product. Two key π–π interactions are proposed to stabilize the precise chiral assembly: one between the phenyl groups of the oxazoline and the quinoline moiety of the directing group, and another between the phenyl group of the alkyne and the quinoline moiety. This precise organization enforced exclusive η2-coordination of the terminal alkyne from the top apical position. The synthesis of chiral crown ethers incorporating planar-chiral cyclophane scaffolds and their application as effective active templates for the diastereoselective synthesis of mechanically planar-chiral rotaxanes further highlighted the practicability of this method. Moreover, the promising fluorescence and circularly polarized luminescence of the resulting planar-chiral cyclophanes exhibited their potential for discovering chiral optics and supramolecular devices.
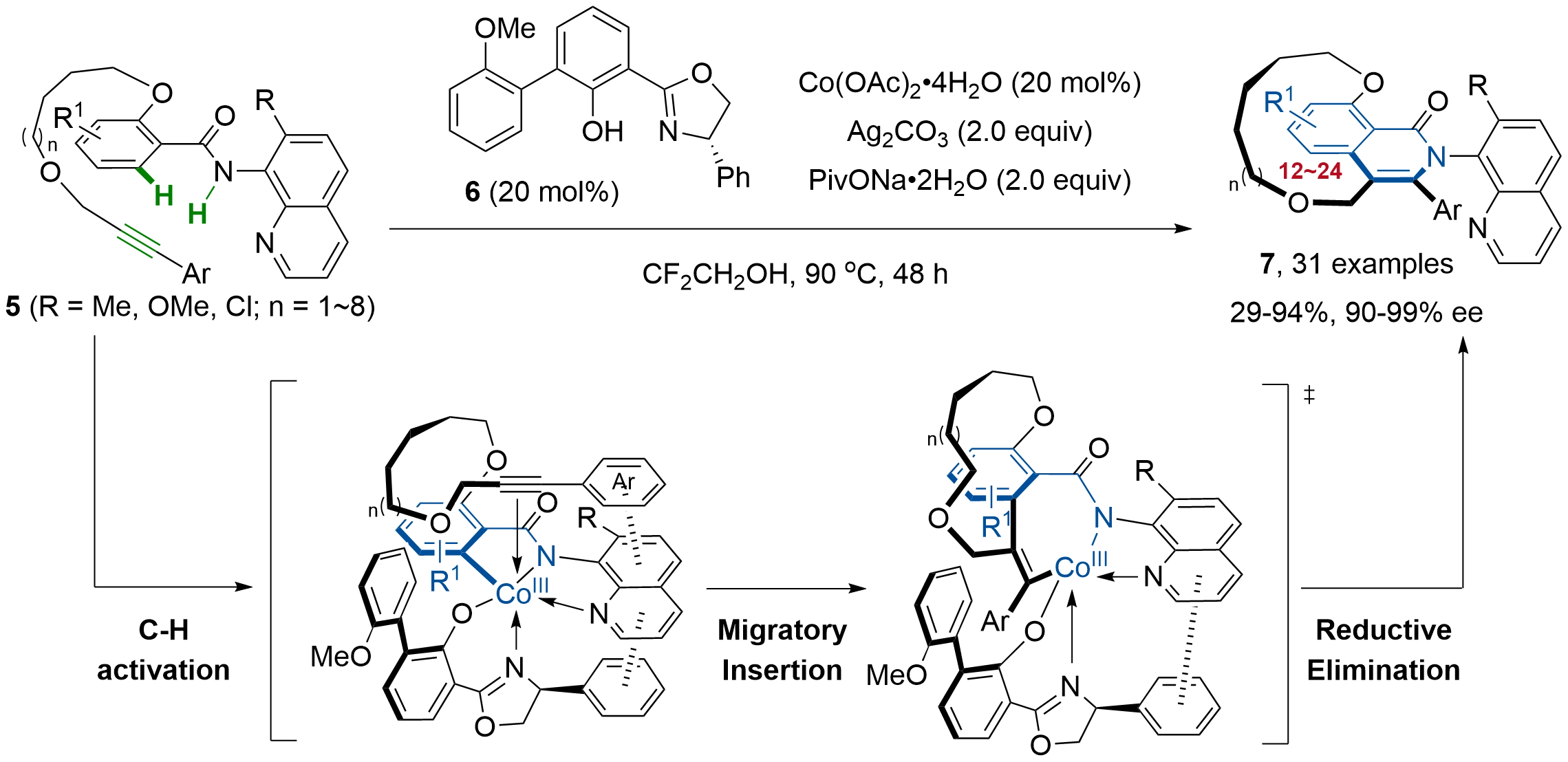
Figure 3. Salox−cobalt(III) complex catalyzed enantioselective C–H macrocyclization toward planar-chiral cyclophanes 7.
3. Conclusion and Outlook
With the rapid evolution of asymmetric catalysis, great advances have been made in the catalytic enantioselective construction of diverse planar-chiral macrocycles via direct asymmetric macrocyclization. However, enantioselective macrocyclization through de novo aromatic ring formation remains underexplored, largely due to the formidable challenge of simultaneously controlling regio- and enantioselectivity during macrocyclization. Recently, employing an asymmetric C–H activation strategy, the groups of Zhao[21], Shi[22], and You[23] have independently developed Rh- or Co-catalyzed intramolecular annulations of functionalized linear alkynes, allowing the enantioselective synthesis of a broad range of planar chiral macrocycles with excellent enantioselectivities. Nevertheless, as an appealing synthetic approach, the de novo construction of diverse aromatic rings to access planar chiral macrocycles is still in its infancy, and awaits further exploration. First, expanding the repertoire of reaction types for the de novo formation of aromatic rings, and the diversity of the aromatic rings themselves is highly desirable. To date, only two strategies have been reported: the [2+2+2] annulation of triynes for benzene rings, and the sequential C–H activation/intramolecular annulation of alkynes with arenes for indole, isoquinoline, or isoquinolinone. Second, the development of new chiral catalyst systems based on novel chiral ligands and/or diverse metal catalysts (e.g. Fe, Ni, Cu, etc.) for asymmetric C–H macrocyclization represents a promising yet underexplored direction. Third, a deeper mechanistic understanding of the origins of stereocontrol in these C–H activation/macrocyclization cascades is urgently needed, as such insight would greatly facilitate broader application of this strategy to the diverse synthesis of planar-chiral macrocycles. Furthermore, the exploration of more direct asymmetric macrocyclizations to access planar-chiral macrocycles featuring diverse ansa chains, particularly those incorporating silicon or boron atoms or other unique functional groups, would be another important direction. More importantly, the exploration of functional applications of planar-chiral macrocycles in fields such as medicinal and agrochemical discovery, chiral ligands/catalysts in asymmetric catalysis, attractive candidates for chiral recognition, and functional chiroptical materials, as well as the design and synthesis of planar-chiral macrocycles with tailored functions, is of great significance and awaits further exploitation. With the ongoing advent of novel synthetic strategies, catalyst systems, and reactions, we are confident that a wide array of direct enantioselective macrocyclization, especially those through de novo aromatic ring formation, will emerge as a powerful and general platform for accessing diverse, function-oriented planar-chiral macrocycles in the near future.
Authors contribution
Ye ZT, Xiao Q: Investigation, visualization, writing-original draft.
Chen G: Supervision, writing-review & editing.
Yu JS: Conceptualization, project administration, funding acquisition, resources, supervision, writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 22471073), the Technology Innovation Project of Shanghai Municipal Agricultural Committee (Grant No. HNK T2023302), and the Ministry of Education (PCSIRT) and the Fundamental Research Funds for the Central Universities (This program does not have a grant number).
Copyright
© The Author(s) 2026.
References
-
3. Glunz PW. Recent encounters with atropisomerism in drug discovery. Bioorg Med Chem Lett. 2018;28(2):53-60.[DOI]
-
4. Roy I, David AHG, Das PJ, Pe DJ, Stoddart JF. Fluorescent cyclophanes and their applications. Chem Soc Rev. 2022;51(13):5557-5605.[DOI]
-
5. Han XN, Han Y, Chen CF. Fluorescent macrocyclic arenes: Synthesis and applications. Angew Chem Int Ed. 2025;137(12):e202424276.[DOI]
-
6. Tanaka K. Catalytic enantioselective synthesis of planar chiral cyclophanes. Bull Chem Soc Jpn. 2018;91(2):187-194.[DOI]
-
7. Dong Z, Li J, Zhao C. Catalytic enantioselective macrocyclization for the synthesis of planar-chiral cyclophanes: Recent updates. Eur J Org Chem. 2024;27(47):e202400841.[DOI]
-
8. Zhu K, Yang L, Yang Y, Wu Y, Zhang F. Recent advances toward the catalytic enantioselective synthesis of planar chiral cyclophanes. Chin Chemical Lett. 2025;36(7):110678.[DOI]
-
11. Dong Z, Lv K, Yuan C, Wang L, Wang H, Li J, et al. Syntheses, mechanism insights, and anti-inflammatory activities of conformationally defined [n] metacyclophanes. ACS Catal. 2026;16(3):2182-2193.[DOI]
-
12. Gagnon C, Godin É, Minozzi C, Sosoe J, Pochet C, Collins SK. Biocatalytic synthesis of planar chiral macrocycles. Science. 2020;367(6480):917-921.[DOI]
-
14. Garnes-Portolés F, Leyva-Pérez A. Macrocyclization reactions at high concentration (≥0.2M): The role of catalysis. ACS Catal. 2023;13(14):9415-9426.[DOI]
-
16. Tanaka K, Sagae H, Toyoda K, Hirano M. Enantioselective synthesis of planar-chiral metacyclophanes through cationic Rh(I)/modified-BINAP-catalyzed inter- and intramolecular alkyne cyclotrimerizations. Tetrahedron. 2008;64(5):831-846.[DOI]
-
21. Zhai H, Lv K, Li J, Wang J, Liu T, Zhao C. Rhodium(III)-catalyzed atroposelective indolization to access planar-chiral macrocycles. J Am Chem Soc. 2024;146(42):29214-29223.[DOI]
-
22. Wang BJ, Jiang TY, Jiang YL, Wu X, Yao QJ, Shi BF. Catalytic enantioselective synthesis of planar-chiral cyclophanes via a chiral octahedral cobalt(III)-templated C−H macrocyclization. J Am Chem Soc. 2025;147(49):45785-45792.[DOI]
-
24. Tao P, Jia Y. Rhodium-catalyzed intramolecular annulation via C–H activation leading to fused tricyclic indole scaffolds. Chem Commun. 2014;50(55):7367-7370.[DOI]
-
25. Zhou B, Yang Y, Tang H, Du J, Feng H, Li Y. Rh(III)-catalyzed intramolecular redox-neutral or oxidative cyclization of alkynes: Short, efficient synthesis of 3, 4-fused indole skeletons. Org Lett. 2014;16(15):3900-3903.[DOI]
-
26. Yang H, Zhang R, Zhang SZ, Gu Q, You SL. Synthesis of hexamethyl-1, 1′-spirobiindane-based chiral spiro cp ligands and their application in rhodium-catalyzed enantioselective aryl C–H addition to nitroalkenes. ACS Catal. 2023;13(13):8838-8844.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite