Jun Xuan, Anhui Province Key Laboratory of Chemistry for Inorganic/Organic Hybrid Functionalized Materials, College of Chemistry & Chemical Engineering, Anhui University, Hefei 230601, Anhui, China; Key Laboratory of Structure and Functional Regulation of Hybrid Materials, Ministry of Education, Anhui University, Hefei 230601, Anhui, China. E-mail: xuanjun@ahu.edu.cn
Abstract
Over the past few decades, transition-metal-free chiral Brønsted acid or Lewis
Graphical Abstract
Keywords
1. Introduction
Diazo compounds serve as useful and versatile synthons in organic synthesis due to their diverse reactivity, acting as nucleophiles, carbene precursors, or radical precursors[1-3]. Over the past decades, the transition-metal-free chiral Brønsted acid or Lewis
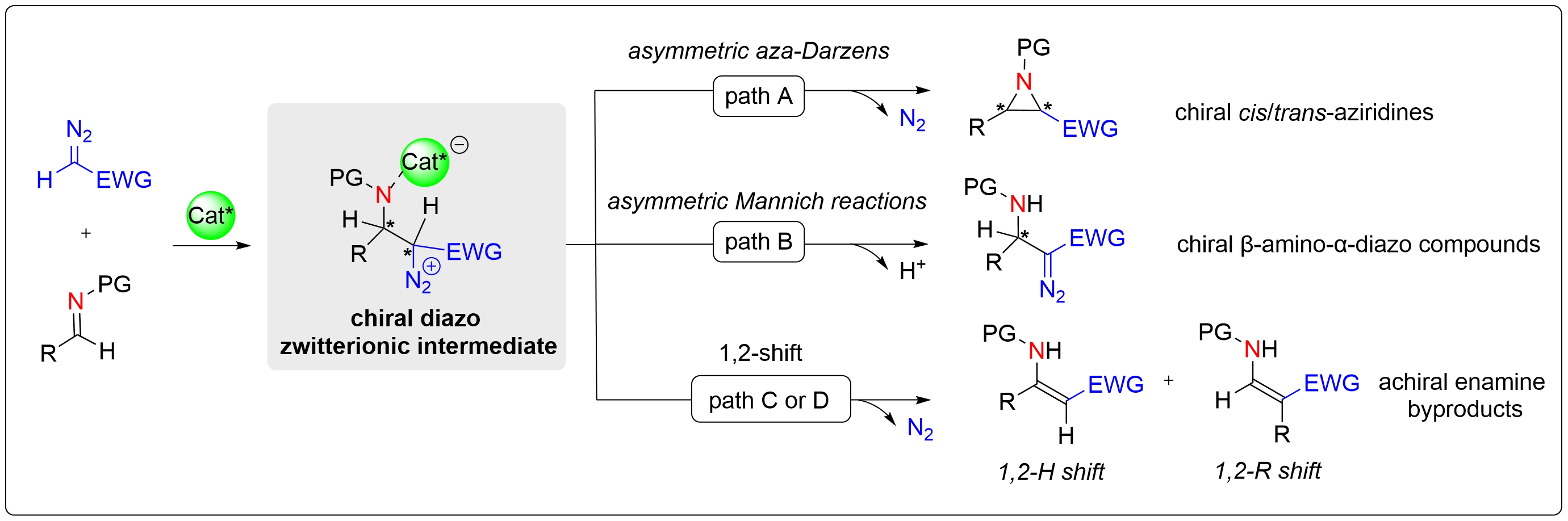
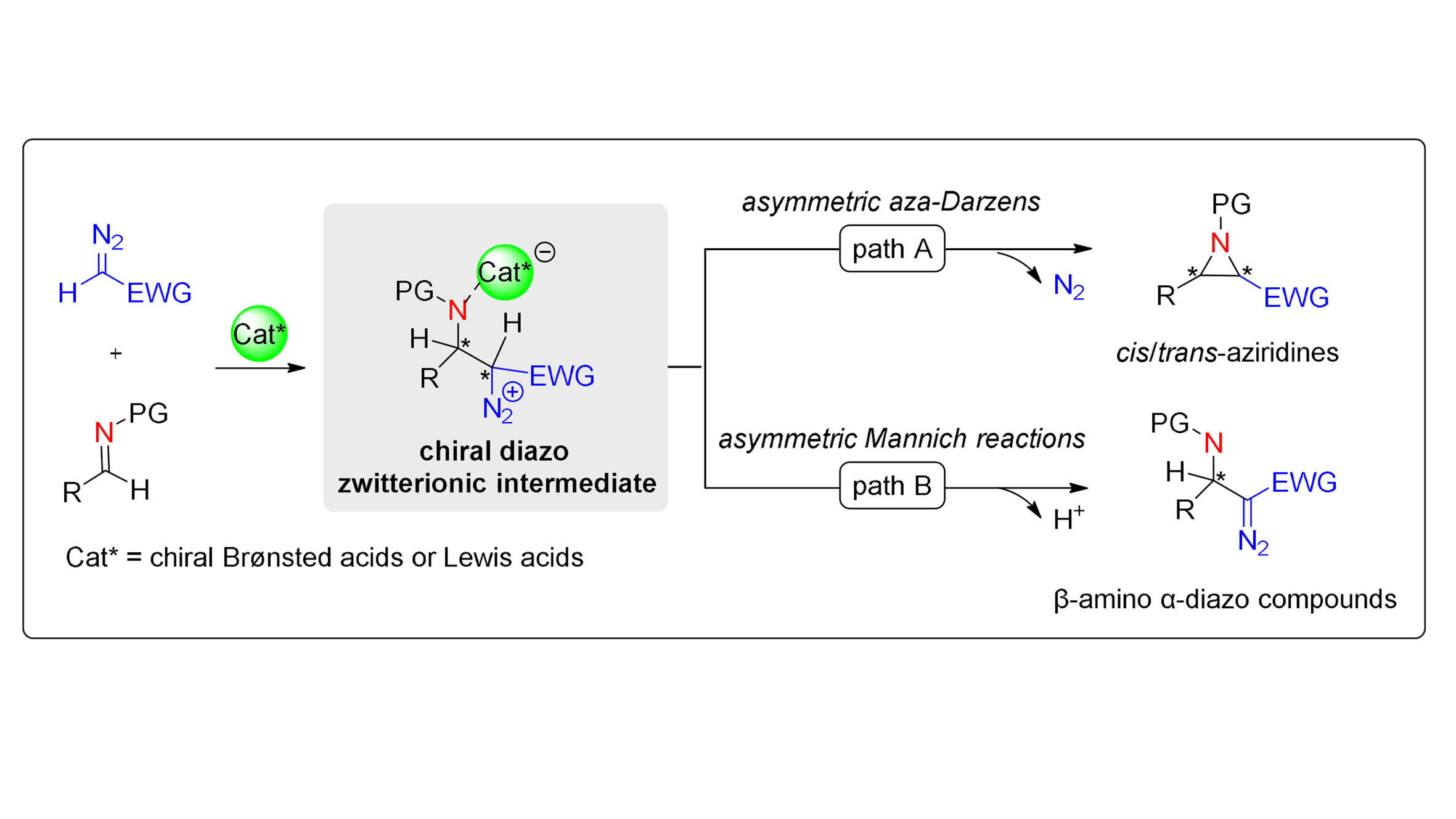
Figure 1. Chiral Brønsted/Lewis acid-catalyzed asymmetric diazo nucleophile-imine reactions.
2. Asymmetric Aza-Darzens Reaction
Aziridines are privileged structural motifs in organic synthesis, attributed to their inherent ring strain and versatile reactivity
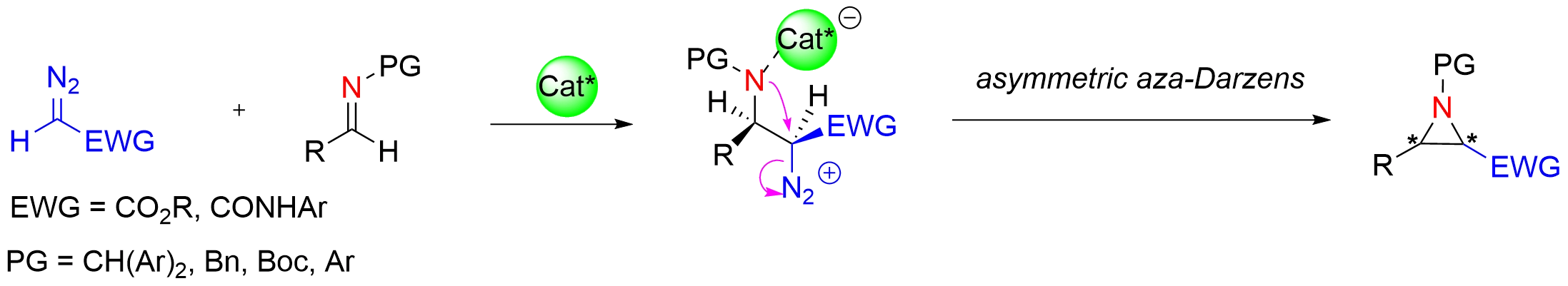
Figure 2. Chiral Brønsted/Lewis acid-catalyzed asymmetric aza-Darzens reaction of diazo nucleophile with imine.
2.1 Chiral boroxinate-catalyzed asymmetric aza-Darzens reaction
In a pioneering 1999 report, the Wulff group described a boroxinate (BOROX) Brønsted acid catalyst derived from
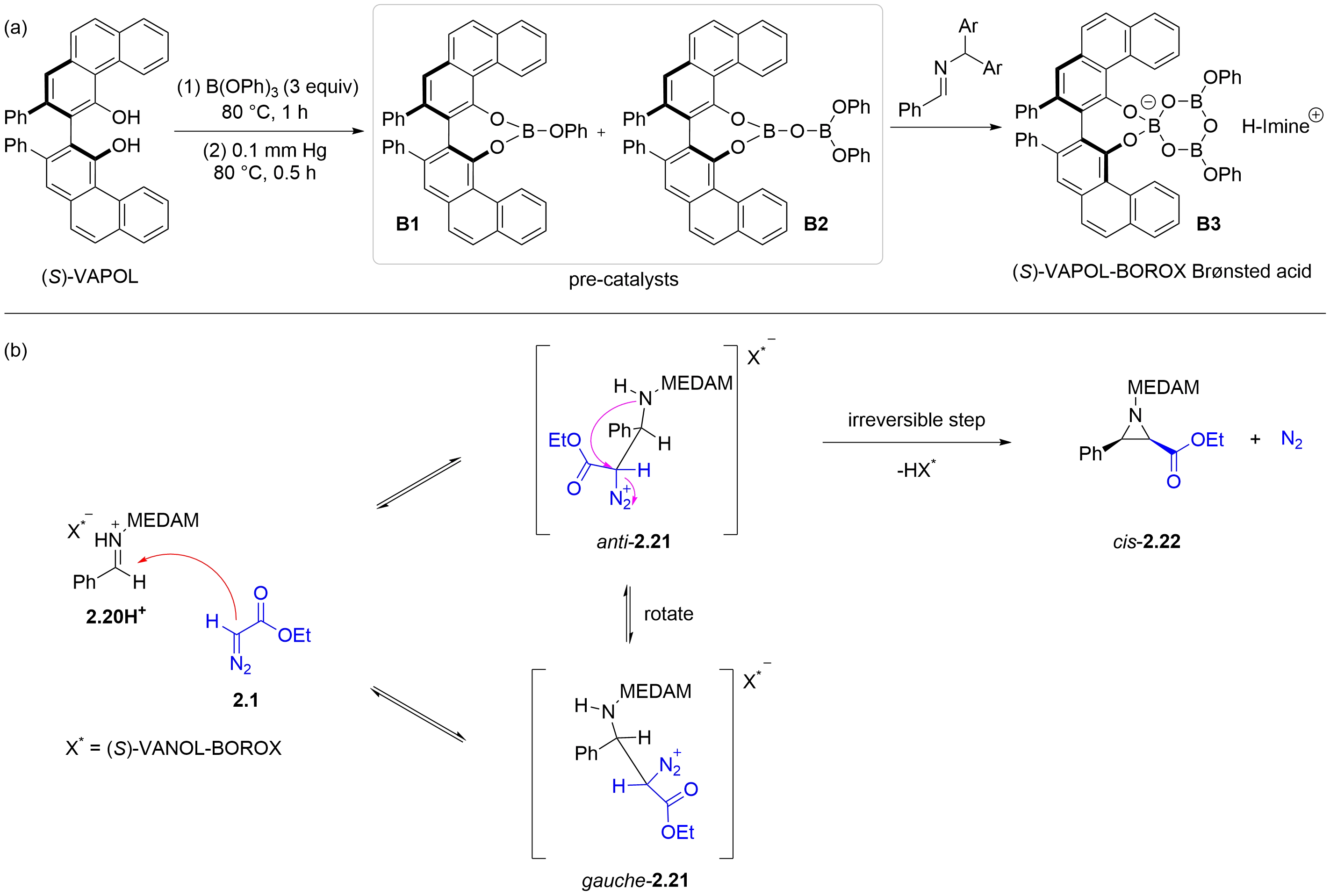
Figure 3. Mechanism of chiral BOROX-catalyzed asymmetric aza-Darzens reaction. BOROX: boroxinate.
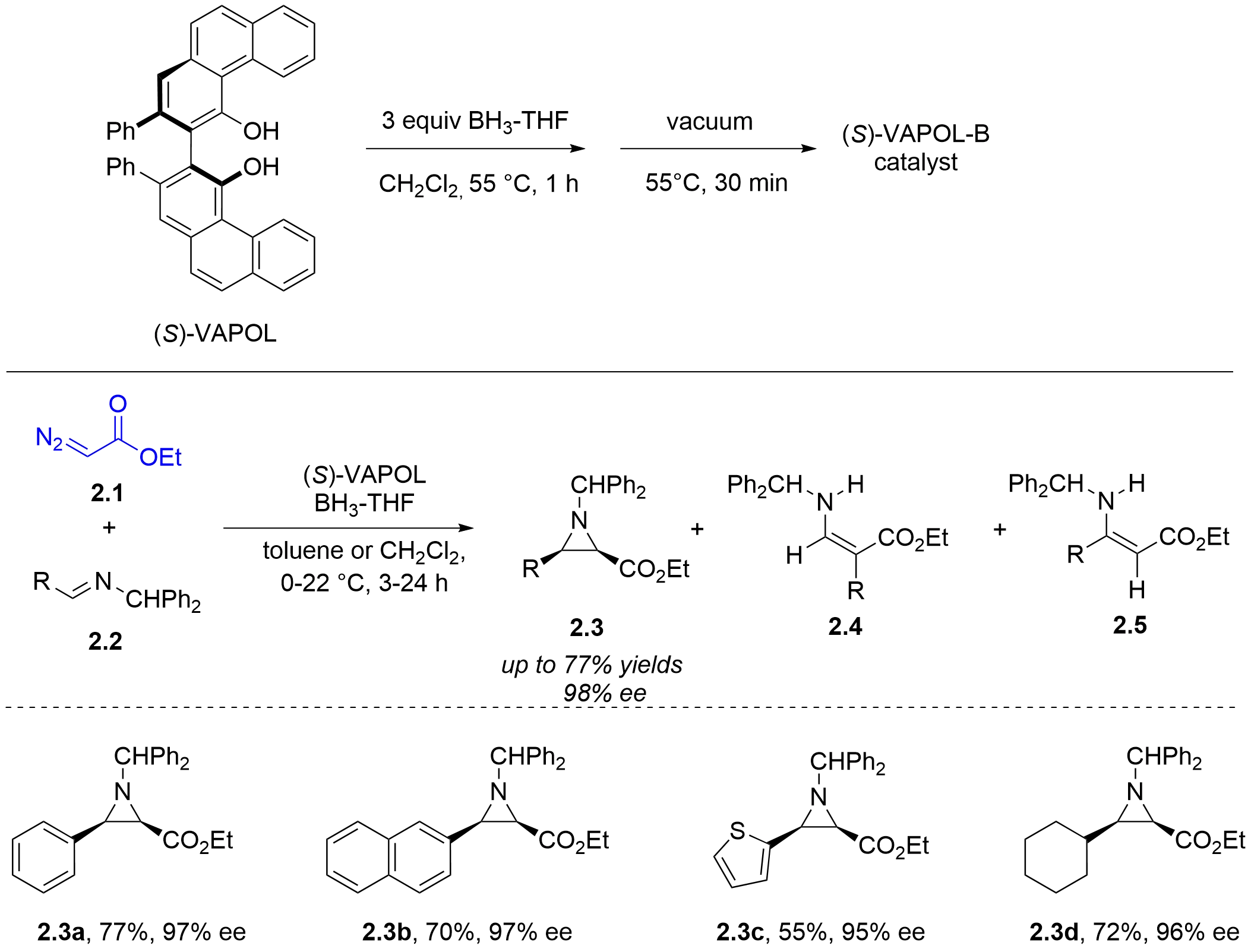
Figure 4. Chiral boroxinate-catalyzed asymmetric aza-Darzens reaction of α-diazoacetates with N-diphenylmethyl imines.
In 2007, the Wulff group reported an asymmetric aza-Darzens reaction of N-dianisylmethyl aldimines 2.6 with ethyl α-diazoacetate 2.1 (Figure 5)[12]. Compared to the diphenylmethyl protecting group, the dianisylmethyl (DAM) group could be more easily removed under acidic conditions to afford the unprotected N-H-aziridines without competitive ring-opening.
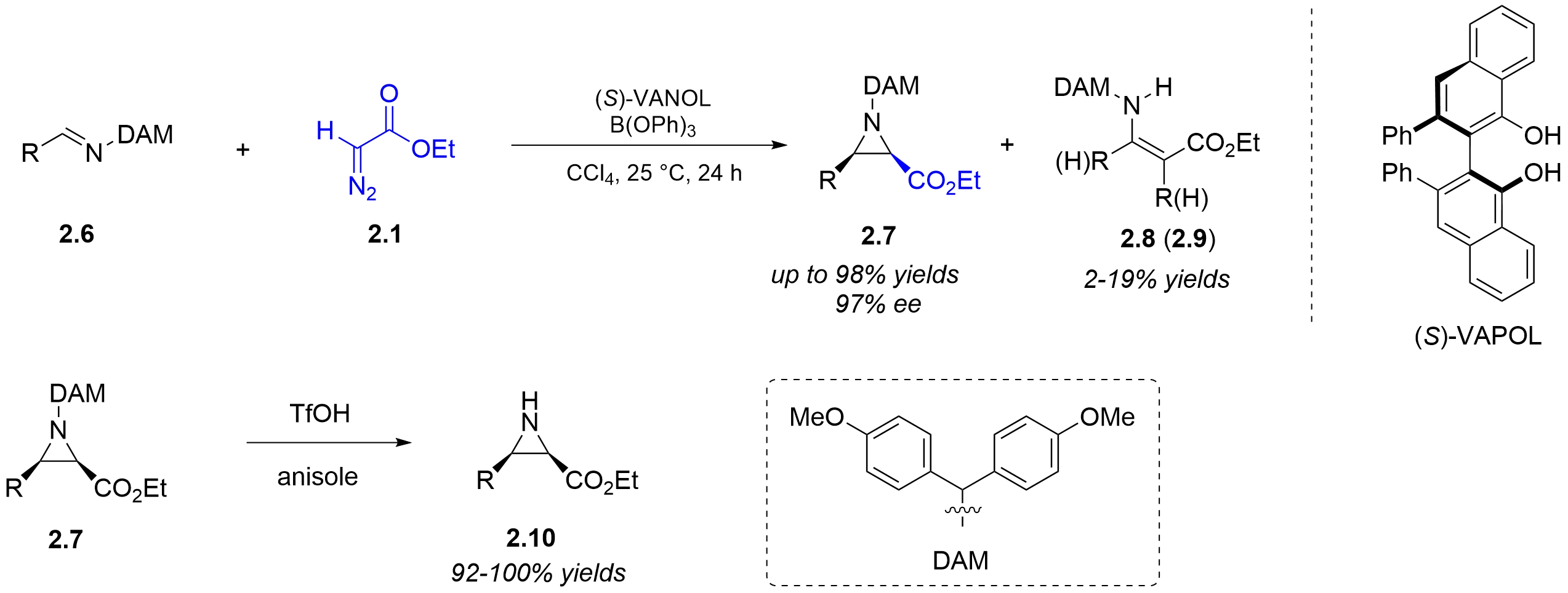
Figure 5. Chiral BOROX-catalyzed asymmetric aza-Darzens reaction of α-diazoacetates with N-DAM-imines. BOROX: boroxinate; DAM: dianisylmethyl.
The following year, the Wulff group discovered that the tetra-tert-butyldianisylmethyl (BUDAM) protecting group could further enhance the efficiency and enantioselectivity of the asymmetric aza-Darzens reaction of aldimines 2.11 with ethyl α-diazoacetate 2.1 under chiral BOROX Brønsted acid catalysis (Figure 6)[13]. Compared with aryl aldimines, alkyl aldimines deliver slightly lower yields and ee values.
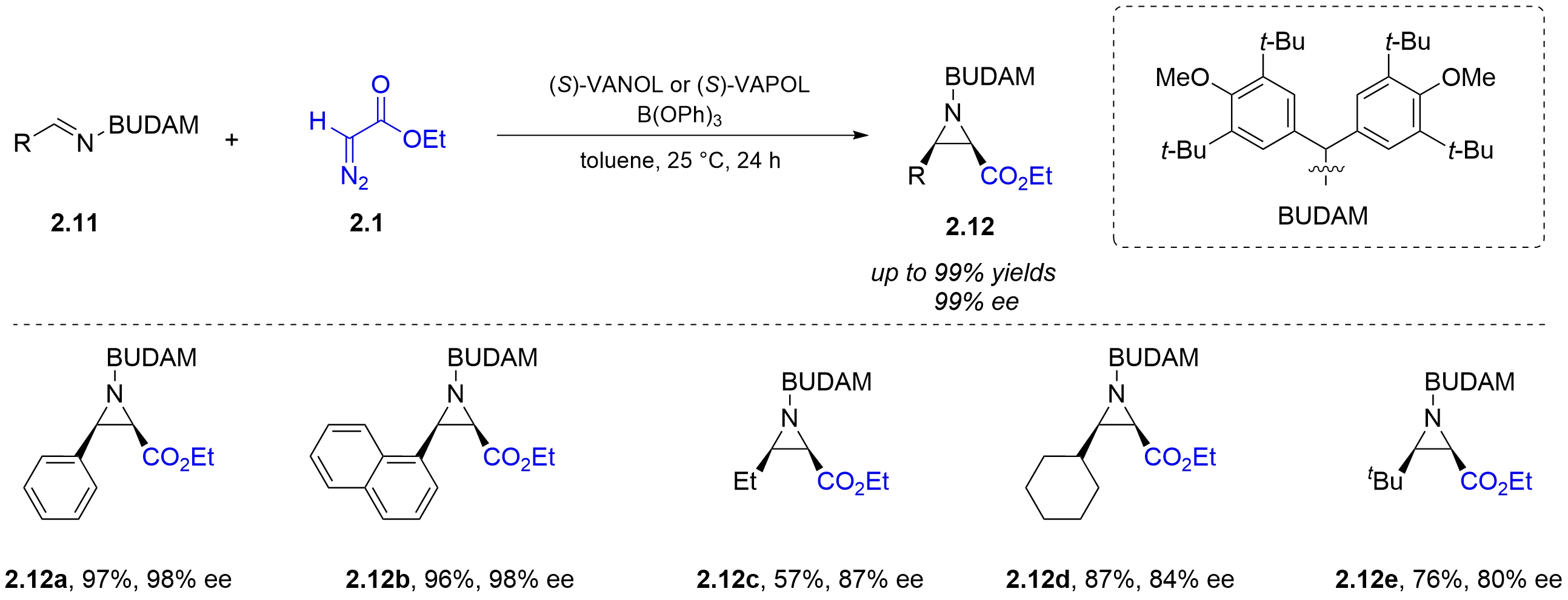
Figure 6. Chiral BOROX-catalyzed asymmetric aza-Darzens reaction of α-diazoacetates with N-BUDAM-imines. BOROX: boroxinate; BUDAM: tetra-tert-butyldianisylmethyl.
In terms of innovation in reaction substrates, in 2007, Wulff and colleagues achieved the synthesis of cis-aziridine ketones from the reaction of diphenylmethyl aldimines 2.2 with vinyl diazoketones 2.13, catalyzed by chiral BOROX generated in situ from chiral
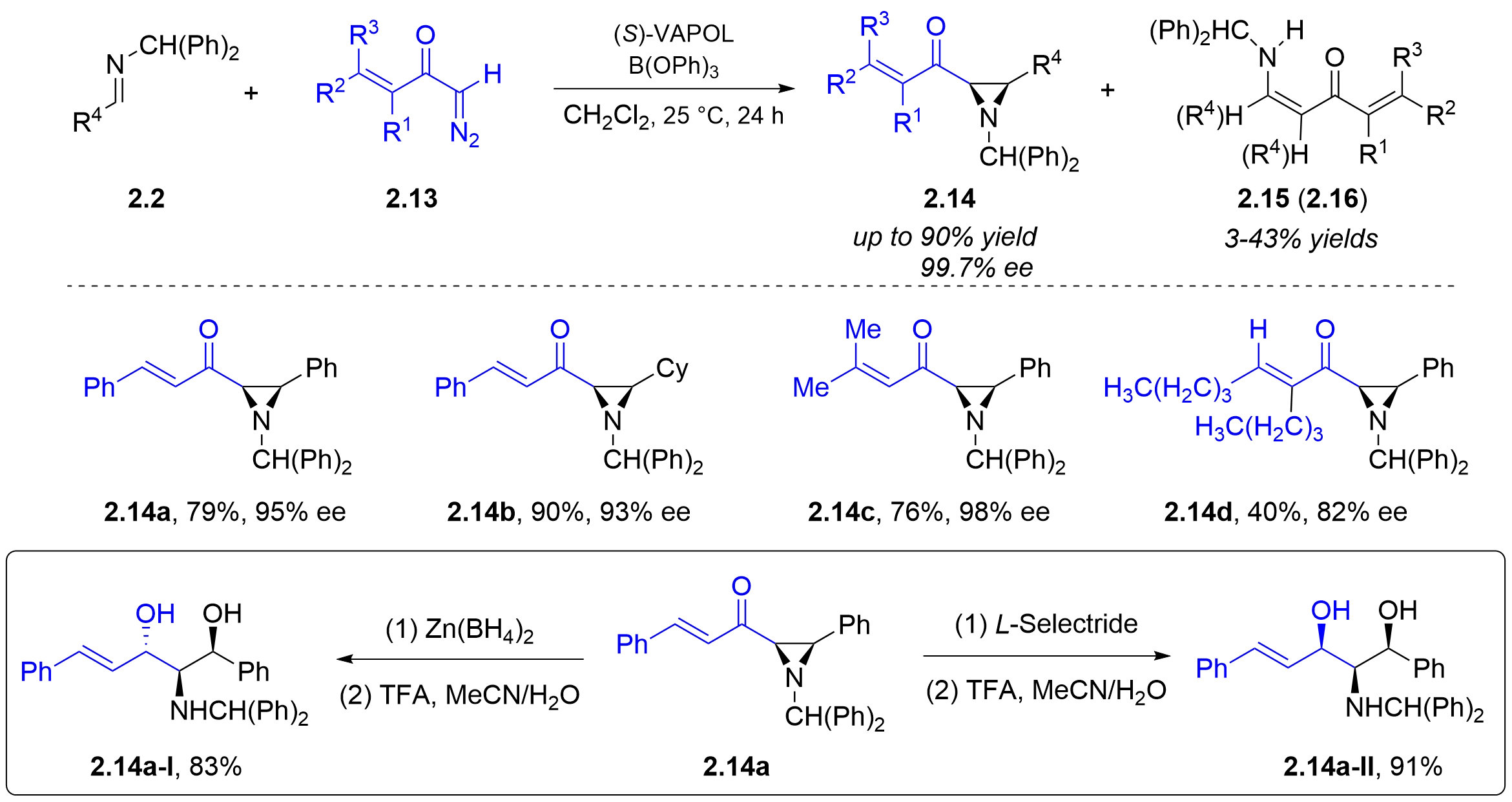
Figure 7. Chiral BOROX-catalyzed asymmetric aza-Darzens reaction of vinyl diazoketones with diphenylmethyl mines. BOROX: boroxinate.
In 2011, the Wulff group achieved a three-component asymmetric aza-Darzens reaction under this BOROX catalysis. Aldehydes 2.17 and amines 2.18 bearing a bis(dimethylanisyl)methyl group (MEDAM) generated imines in situ in the reaction system, which were then immediately captured by the catalyst and reacted with ethyl α-diazoacetate 2.1 to produce chiral cis-aziridine compounds 2.19
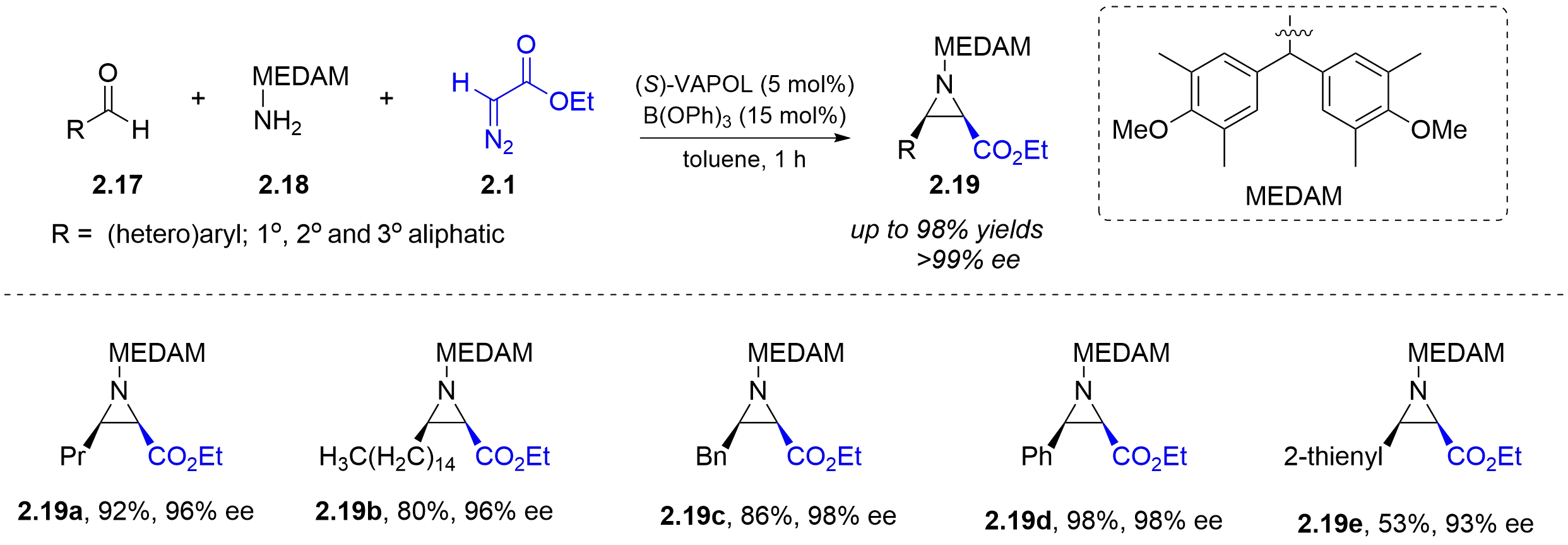
Figure 8. A three-component asymmetric aza-Darzens reaction under chiral BOROX catalysis. BOROX: boroxinate.
In 2012, the Wulff group demonstrated that the mesoborate (B1) and pyroborate (B2) generated from VAPOL and triphenyl borate act as precatalysts. The actual catalytic species is the boroxinate (B3) formed upon reaction of these precatalysts with imines[16]. Furthermore, the mechanism of the chiral BOROX Brønsted acid catalyzed aziridination reaction of imines and ethyl α-diazoacetate 2.1 was investigated using a combination of experimental kinetic isotope effects and theoretical calculations. Specifically, the process initiates with reversible nucleophilic addition of ethyl α-diazoacetate 2.1 to the intermediate 2.20H+ activated by the catalyst, generating a chiral diazonium intermediate 2.21. The gauche-2.21 can undergo rotation around the C-C bond to give the anti-2.21. An intramolecular irreversible SN2-like ring-closure step then occurs, accompanied by nitrogen extrusion, ultimately yielding the
2.2 Chiral dicarboxylic acid-catalyzed asymmetric aza-Darzens reaction
In contrast to the well-developed asymmetric cis-aziridination of diazo compounds with imines, the trans-selective variant has remained largely elusive. In 2008, the Maruoka group reported the trans-selective asymmetric aziridination of diazoacetamides 2.24 and N-Boc aldimines 2.23 catalyzed by axially chiral dicarboxylic acids (Figure 9)[18]. α-Diazoacetamide is critical for selective
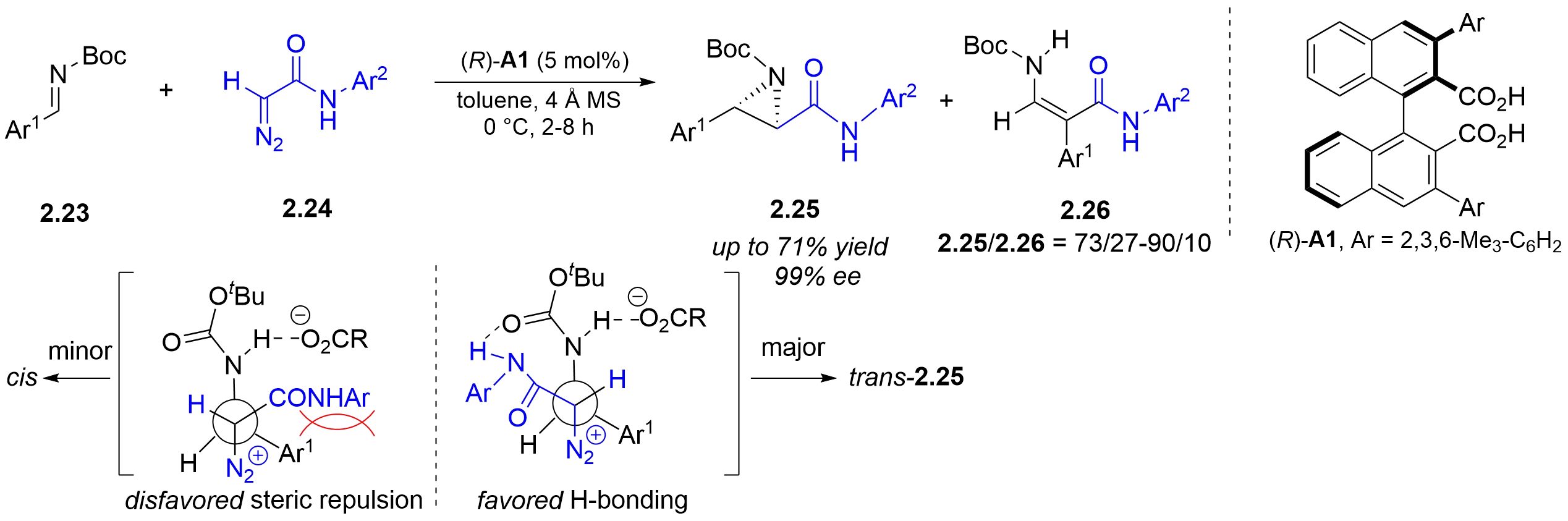
Figure 9. Chiral dicarboxylic acid-catalyzed asymmetric aza-Darzens reaction of α-diazoacetamides with N-Boc-imines.
2.3 Chiral phosphoric acid/N-triflylphosphoramide-catalyzed asymmetric aza-Darzens reaction
Chiral phosphoric acid could lower the lowest unoccupied molecular orbital (LUMO) energy of the C=N bond to activate an imine group. In 2009, the Akiyama group reported a chiral phosphoric acid-catalyzed asymmetric aza-Darzens reaction of ethyl diazoacetate 2.1 and N-p-methoxybenzyl (PMB) aldimines derived from aryl glyoxals 2.27, which furnished cis-aziridine carboxylates 2.29 (Figure 10)[19]. Phenyl, 4-biphenyl, 1-naphthyl, and 2-thienyl groups are all compatible in this catalytic reaction system.
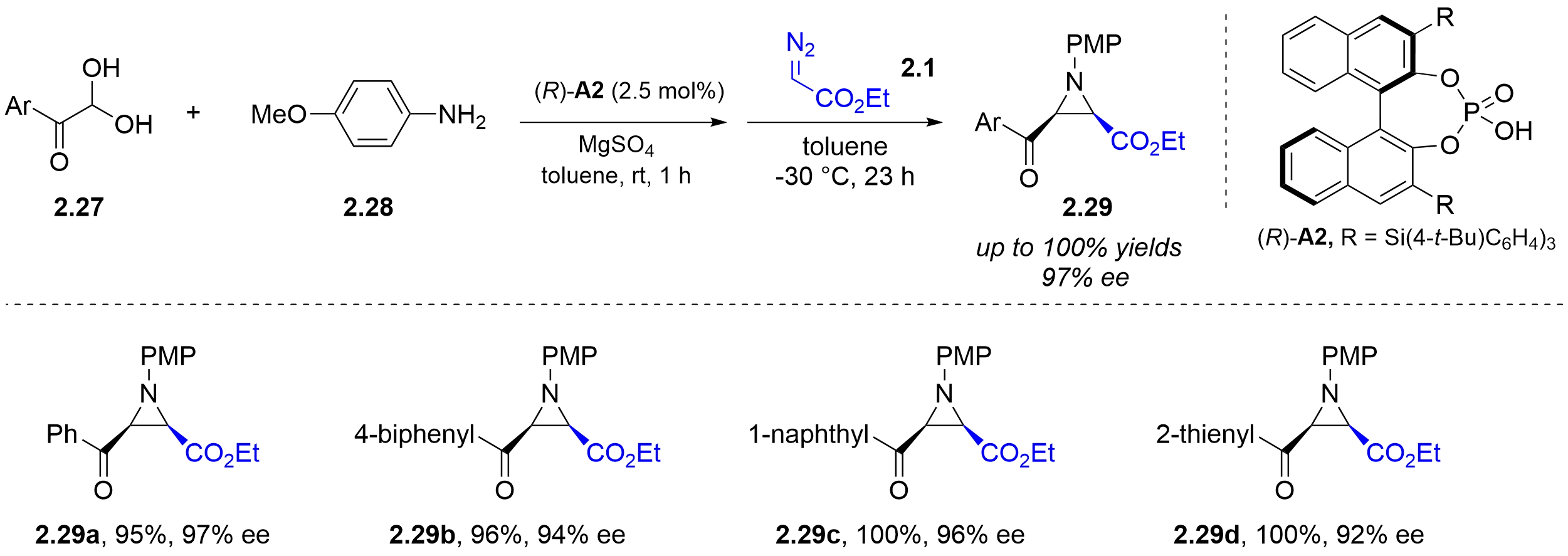
Figure 10. Chiral phosphoric acid-catalyzed asymmetric three-component aziridination reaction of α-ethyl diazoacetate with aryl glyoxals and 4-methoxyaniline.
In the same year, the Zhong group reported a chiral phosphoric acid-catalyzed asymmetric aza-Darzens reaction of diazoacetamides 2.24 and N-Boc aldimines 2.23 (Figure 11)[20]. Both the reaction yields and chemoselectivities were improved significantly compared with those obtained using the axially chiral dicarboxylic acid [(R)-A1] shown in Figure 9. A wide range of aryl-substituted substrates afford satisfactory results in this catalytic reaction.
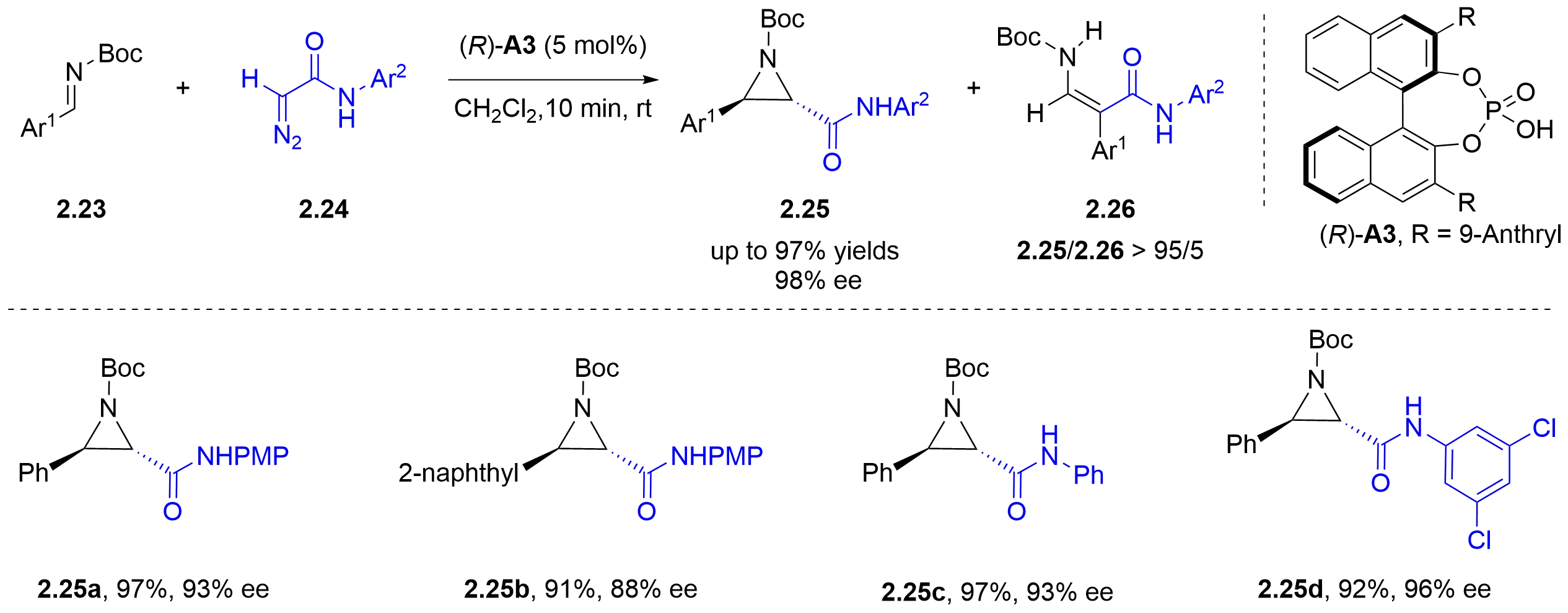
Figure 11. Chiral phosphoric acid-catalyzed asymmetric aza-Darzens reaction of α-diazoacetamides with N-Boc-imines.
Regarding the expansion of new types of diazo substrates for such asymmetric aza-Darzens reactions, the Cahard group in 2012 achieved the diastereo- and enantioselective synthesis of CF3-substituted aziridines 2.31 catalyzed by chiral phosphoric acids
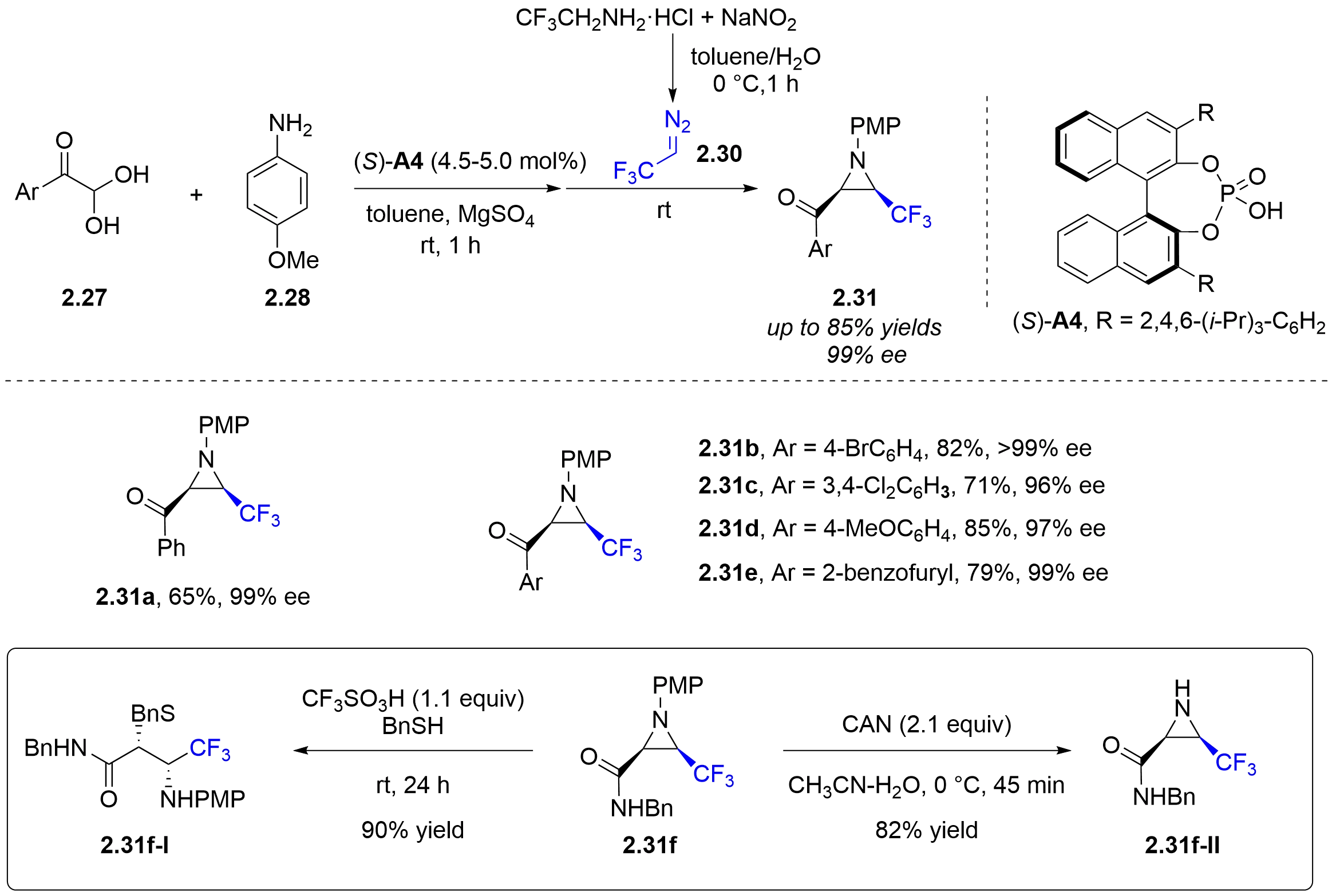
Figure 12. Chiral phosphoric acid-catalyzed asymmetric multicomponent aza-Darzens reaction of in situ generated CF3CHN2 with aryl glyoxal-derived N-PMB imines.
Despite significant advances in chiral Brønsted acid-catalyzed asymmetric aza-Darzens reactions between imines and diazo nucleophiles for aziridine synthesis, these transformations have been largely restricted to acyclic imine substrates, leaving cyclic imines virtually unexplored. In 2026, the Xuan group reported a chiral phosphoric acid-catalyzed asymmetric aza-Darzens reaction of quinoxalin-2(1H)-ones 2.32 with α-diazoacetates 2.33 (Figure 13)[22]. A proposed mechanism for this catalytic asymmetric aza-Darzens reaction was shown in Figure 13a. First, protonation of quinoxalin-2(1H)-one 2.32a with chiral phosphoric acid (R)-A5 affords the activated iminium INT I with a H-bonding interaction between the amidic hydrogen and an oxygen atom of the chiral counterion. Then, an enantio- and diastereo determining nucleophilic addition of diazo acetate 2.33a to the iminium carbon occurs, generating the chiral anti-diazonium intermediate INT II. Finally, an intramolecular SN2 cyclization between the antiperiplanar amine and diazonium group, affords cis-aziridine carboxylate 2.34a with N2 release. The stereochemical outcome can be rationalized by the approach of the diazo nucleophile 2.33a through its Si face (2.33a Si) toward the Re face of the activated iminium intermediate INT I. This trajectory minimizes steric clashes between the bulky anthracen-9-yl substituent of the CPA catalyst [(R)-A5] and the tert-butyl ester moiety of 2.33a Si (Figure 13b).
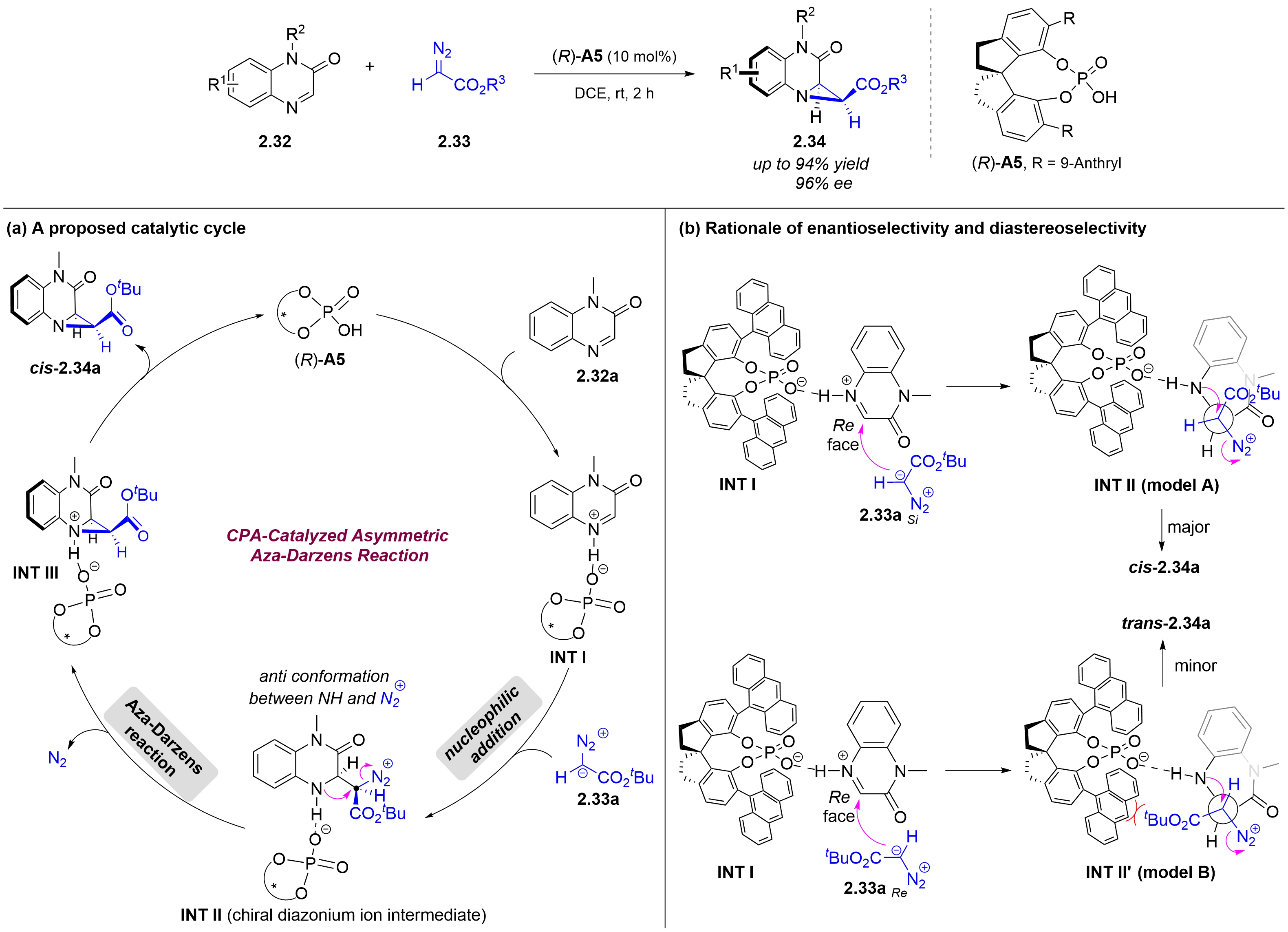
Figure 13. Chiral phosphoric acid-catalyzed asymmetric aza-Darzens reaction of quinoxalin-2(1H)-ones with α-diazoacetates.
Compared with chiral phosphoric acids, chiral N-triflylphosphoramides are more acidic. In 2017, the Bew group reported a chiral
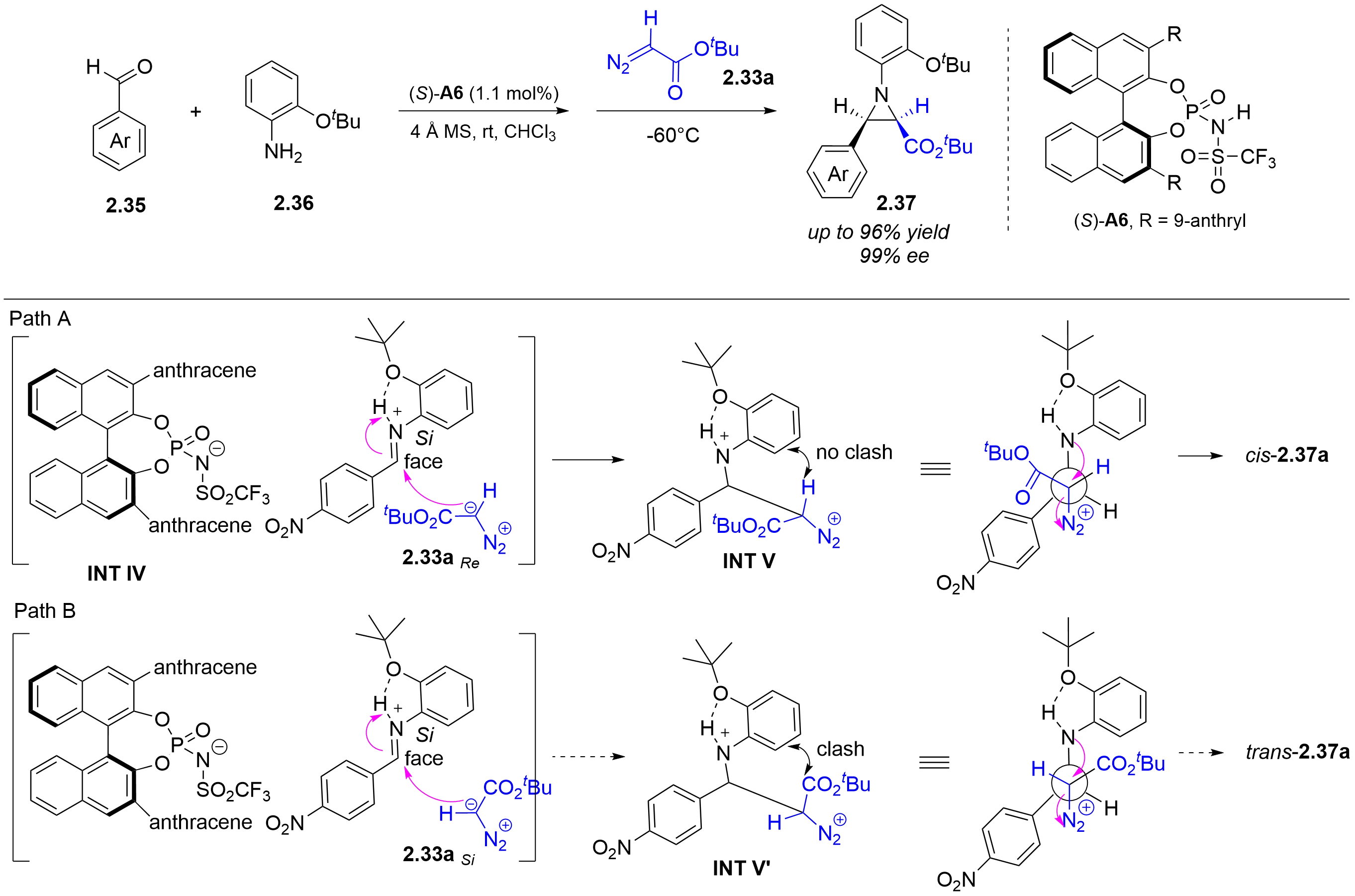
Figure 14. chiral N-triflylphosphoramide-catalyzed three component asymmetric aza-Darzens reaction of (hetero)aryl aldehydes with 2-(tert-butoxy)aniline and tert-butyl
2.4 Combined strong Brønsted acid-catalyzed asymmetric aza-Darzens reaction
Typical chiral Brønsted acids comprise a proton and a chiral conjugate base. As the proton is structurally invariant, the catalyst acidity, reactivity, and chirality are governed solely by the conjugate base. In 2013, the Maruoka group developed a new chiral Brønsted acid catalyst composed of two independent organic molecules, a chiral diol as a ligand and 2-boronobenzoic acid as an achiral Brønsted acid[24]. This in situ assembled catalyst proved efficient in an asymmetric aza-Darzens reaction of N-Boc and
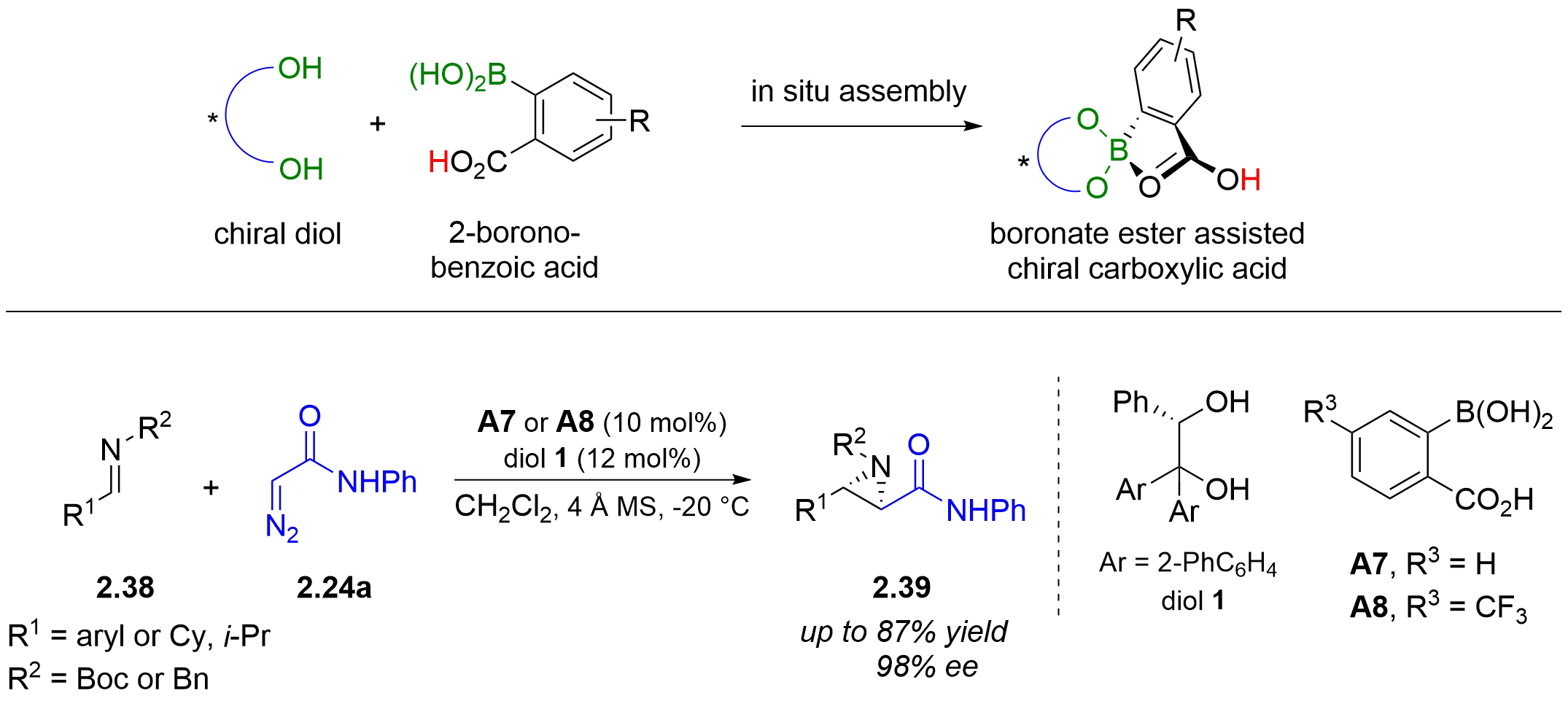
Figure 15. In-situ-assembled boronate ester assisted chiral carboxylic acid-catalyzed asymmetric aza-Darzens reaction.
Inspired by prior studies of arylboronic acid in combination with chiral diols or chiral aminoalcohols, in 2020, the Ma/Zhang group further achieved the synthesis of chiral CF2-functionalized aziridines 2.40 in the three-component asymmetric aza-Darzens reaction of aryl glyoxals 2.27 and 4-methoxyaniline 2.28 with difluorodiazoethyl phenyl sulfone 2.40, using a combined strong Brønsted acid system comprising a chiral disulfonimide (S)-A9 and 2-carboxyphenylboronic acid A7 (Figure 16)[25]. A series of derivatization experiments were conducted to demonstrate the synthetic utility of this method. For example, selective oxidative cleavage of the PMP protecting group in 2.41a using ceric ammonium nitrate proceeded smoothly, affording the unprotected aziridine 2.41a-I in 81% yield. Reduction of the ketone functionality with either sodium borohydride or lithium aluminum hydride furnished the
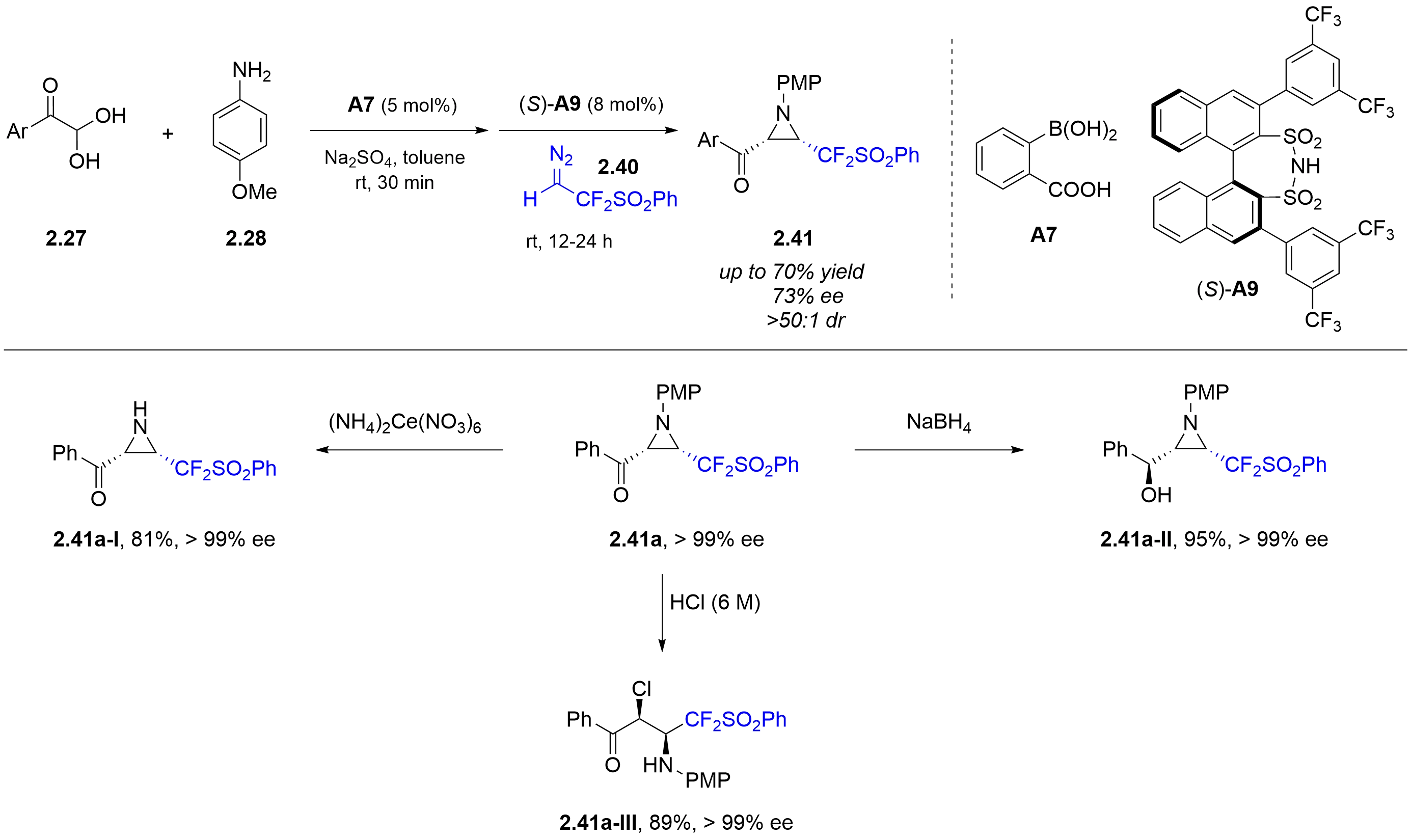
Figure 16. Asymmetric synthesis of CF2-functionalized aziridines by combined strong Brønsted acid catalysis.
3. Asymmetric Mannich-Type Reaction
The diverse reactivity of diazo groups renders their incorporation into molecular core skeletons highly advantageous for subsequent functionalization and structural elaboration[26-29]. Chiral Brønsted/Lewis acid-catalyzed asymmetric Mannich-type reactions provide an efficient strategy for introducing diazo groups into organic molecules. Mechanistically, nucleophilic addition of diazo compounds to the catalyst-activated imine carbon generates a chiral diazo zwitterionic intermediate. Subsequent intramolecular deprotonation of this intermediate by the basic moiety of the catalyst delivers chiral β-amino α-diazo compounds (Figure 17).
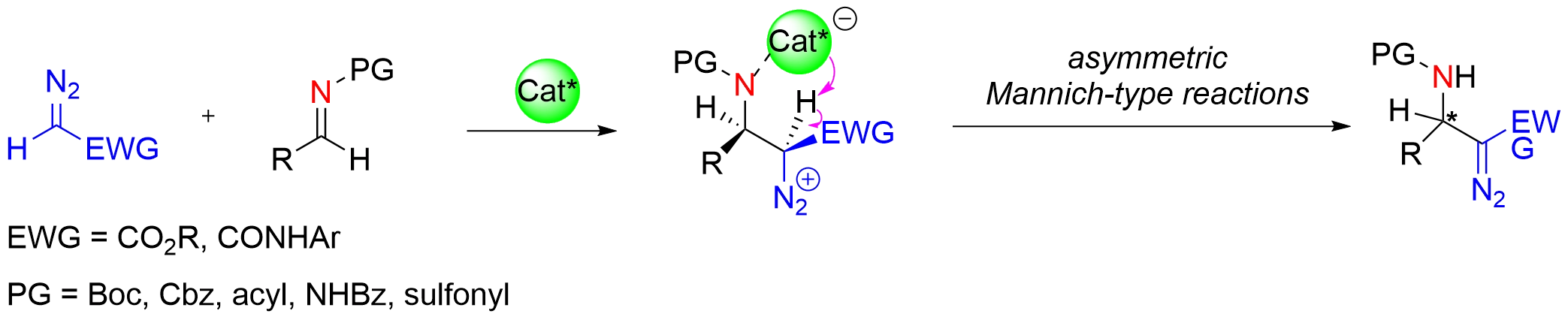
Figure 17. Chiral Brønsted/Lewis acid-catalyzed asymmetric Mannich-type reaction of diazo nucleophile with imine.
3.1 Chiral Brønsted acid-catalyzed asymmetric Mannich-type reaction
In 2005, the Terada group reported the first chiral phosphoric acid-catalyzed asymmetric Mannich-type reaction of ethyl diazoacetate 2.33 and N-acyl aldimines 3.1, affording chiral β-amino-α-diazo esters 3.2 (Figure 18)[30]. The key process was proposed to involve the intracomplex deprotonation of the chiral diazo zwitterionic intermediate by the phosphoryl oxygen through C-H bond cleavage.
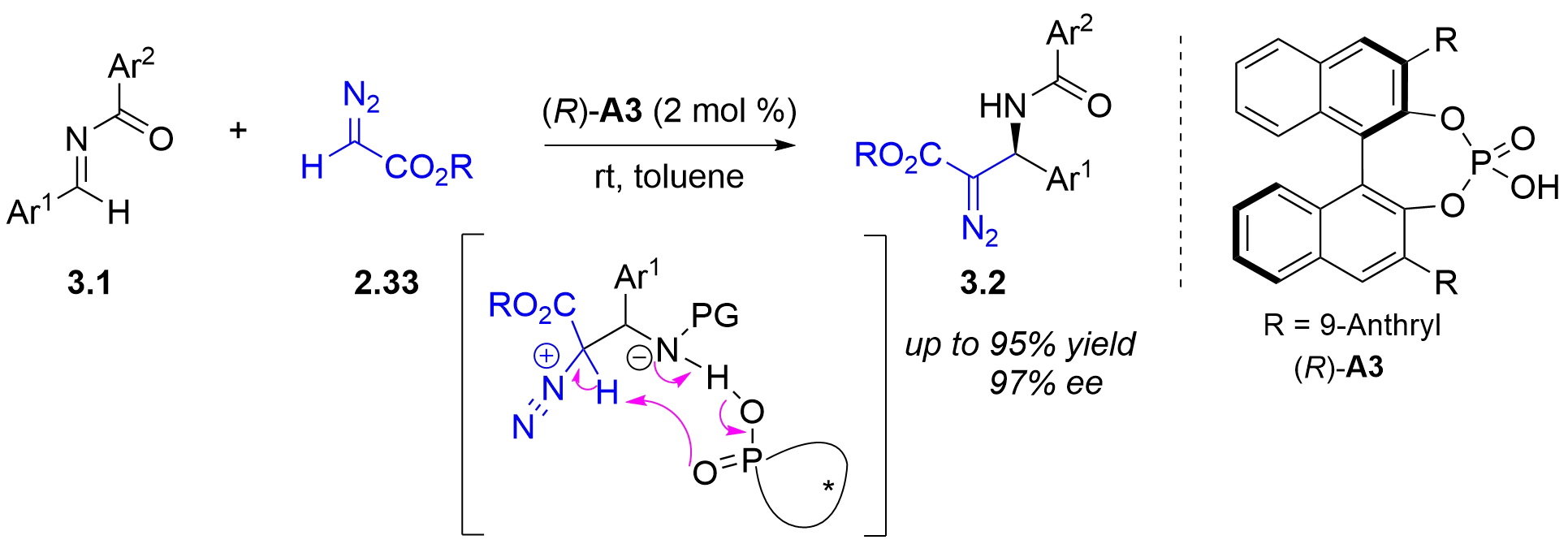
Figure 18. Chiral phosphoric acid-catalyzed asymmetric Mannich-type reaction of α-diazo esters with N-acyl imines.
In contrast to the well-established α-diazo carbonyl chemistry, α-diazomethylphosphonates generally exhibit lower reactivity, which can be attributed to both electronic effects and the greater steric bulk of the phosphonate group. In 2012, the Peng group first applied chiral phosphoric acids to the asymmetric Mannich-type reaction of dialkyl diazomethylphosphonates 3.4 with N-carbamoyl aldimines 3.3 (Figure 19)[31]. The reaction afforded β-N-Boc amino-α-diazophosphonates 3.5 with good yields, diastereoselectivities, and enantioselectivities at a catalyst loading as low as 0.1 mol%. A reasonable transition state model was established to illustrate the catalytic pathway. Phenyl, 2-naphthyl and 2-thienyl are all compatible in this catalytic reaction. The chiral phosphoric acid (R)-A4 serves as a bifunctional Brønsted acid–base catalyst: its acidic proton activates the imine nitrogen via hydrogen bonding, and the nucleophilic carbon of diazomethylphosphonate attacks the imine from the Si-face. The phosphoryl oxygen then acts as a Brønsted basic site to mediate intramolecular deprotonation.
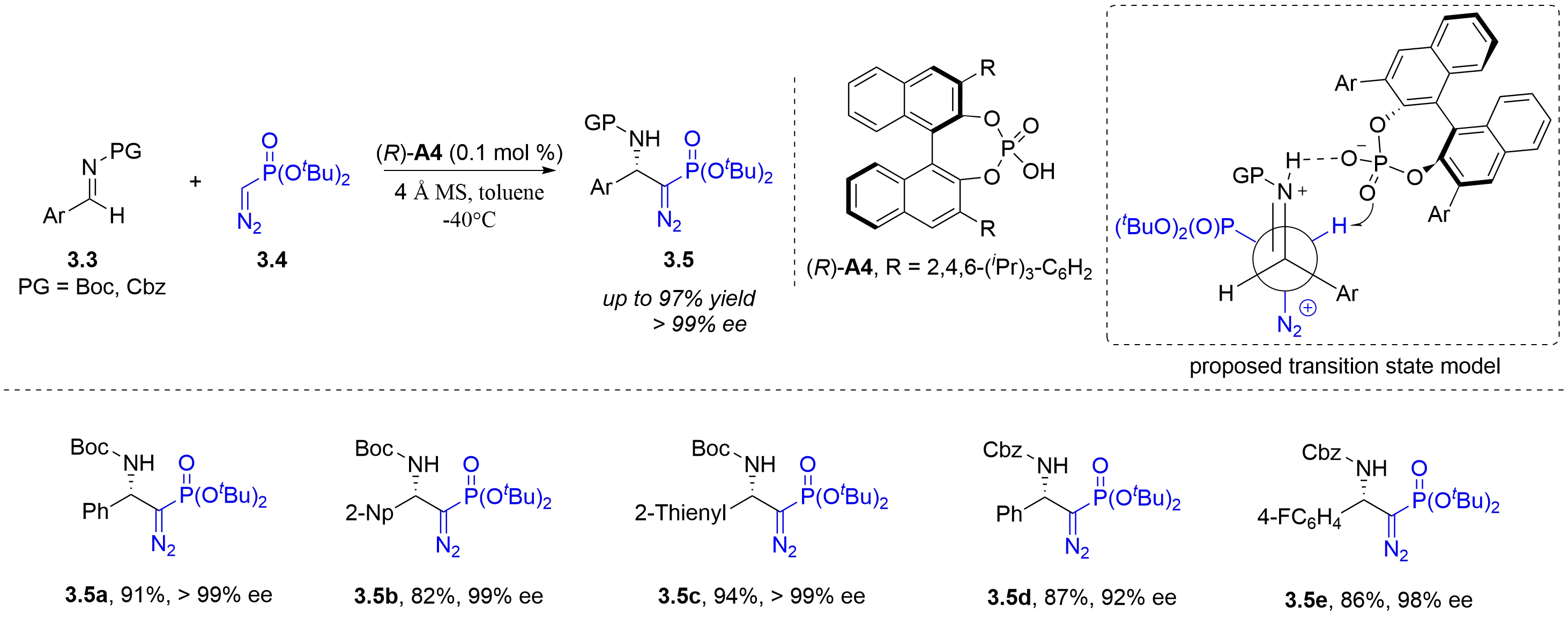
Figure 19. Chiral phosphoric acid-catalyzed asymmetric Mannich-type reaction of dialkyl diazomethylphosphonates with N-carbamoyl imines.
In 2019, the Singh group reported a chiral phosphoric acid-catalyzed tandem Mannich–lactamization reaction (Figure 20)[32]. A reasonable transition-state model was proposed based on experimental results to explain the stereochemical outcome. Chiral phosphoric acid acts as a bifunctional catalyst with both Brønsted acid and Brønsted base sites. In this process, acidic protons activate the in situ generated imines and ester groups via hydrogen bonding. Nucleophilic α-diazo esters 2.33 then attack the Si face of the imines. The α-diazo esters are subsequently deprotonated by the phosphoryloxy group, which serves as the Brønsted base site. The desired products are finally obtained through intramolecular lactamization.
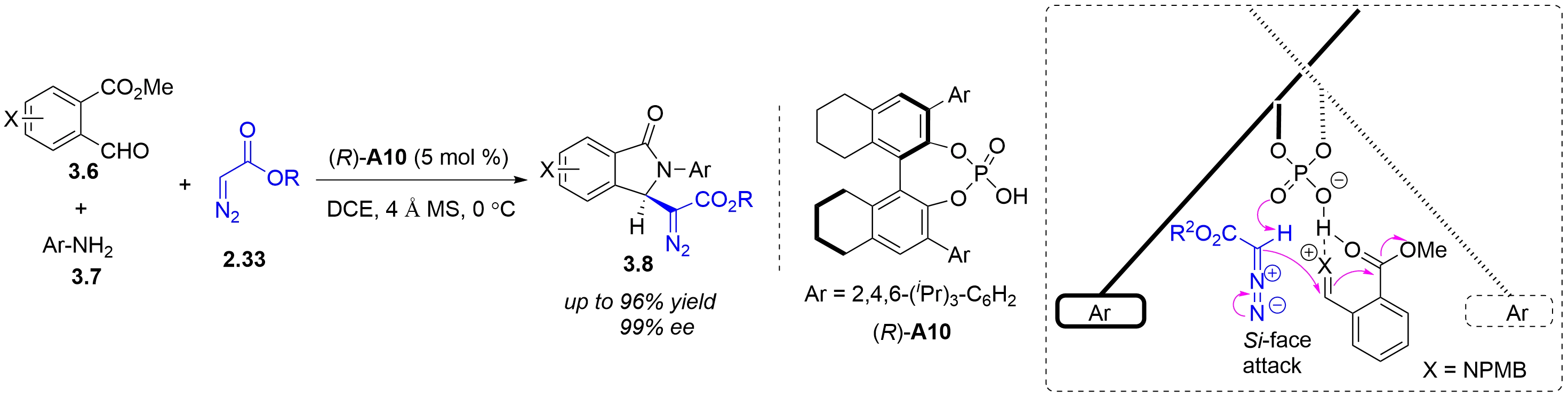
Figure 20. Chiral phosphoric acid-catalyzed Tandem Mannich–lactamization reactions.
In 2019, Ma et al. developed a facile, and chemodivergent, and stereoselective reaction of N-Boc aldimines with PhSO2CF2CHN2
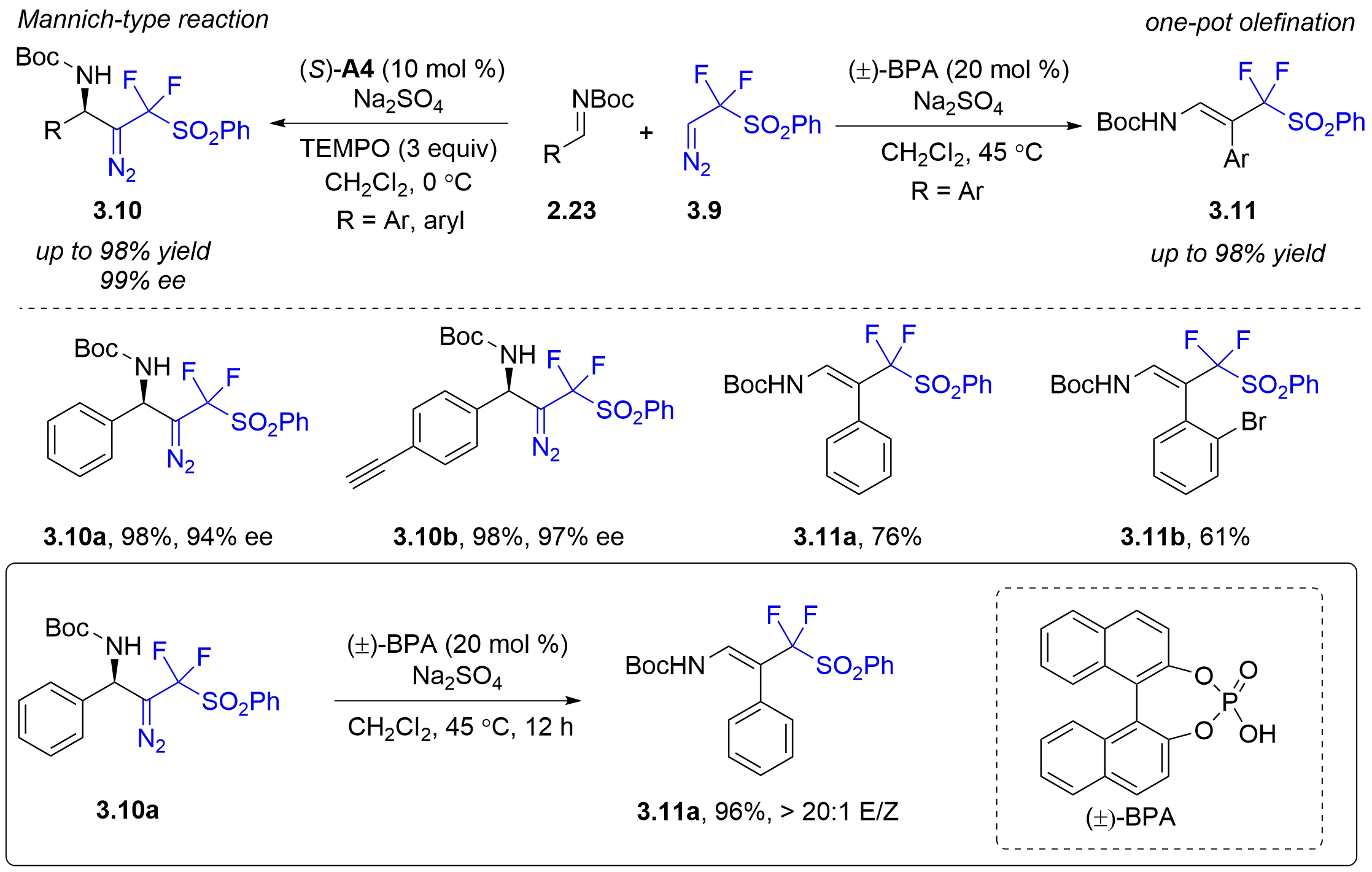
Figure 21. Chiral phosphoric acid-catalyzed chemodivergent and stereoselective reaction of N-Boc aldimines with PhSO2CF2CHN2.
In 2020, the Peng group reported a chiral phosphoric acid-catalyzed asymmetric acyl-Mannich reaction of (diazomethyl)phosphonate 3.14 or ethyl α-diazoacetate 2.1 with isoquinolines (Figure 22)[34]. This highly atom-economical reaction begins with the isoquinoline reacting with diethyl dicarbonate 3.13 to generate intermediate 3.16. Protonation of the 1-ethoxy group in 3.16 by chiral phosphoric acid, followed by loss of EtOH, furnishes a 2-ethoxycarbonylisoquinolinium ion. This cation forms an ion pair with the chiral phosphate anion, which directs the enantioselective nucleophilic addition of 3.14 to the Si-face of the iminium. Finally, the phosphoryl oxygen acts as a Brønsted base to abstract the α-hydrogen from the α-(diazomethyl)phosphonate, affording product 3.15.
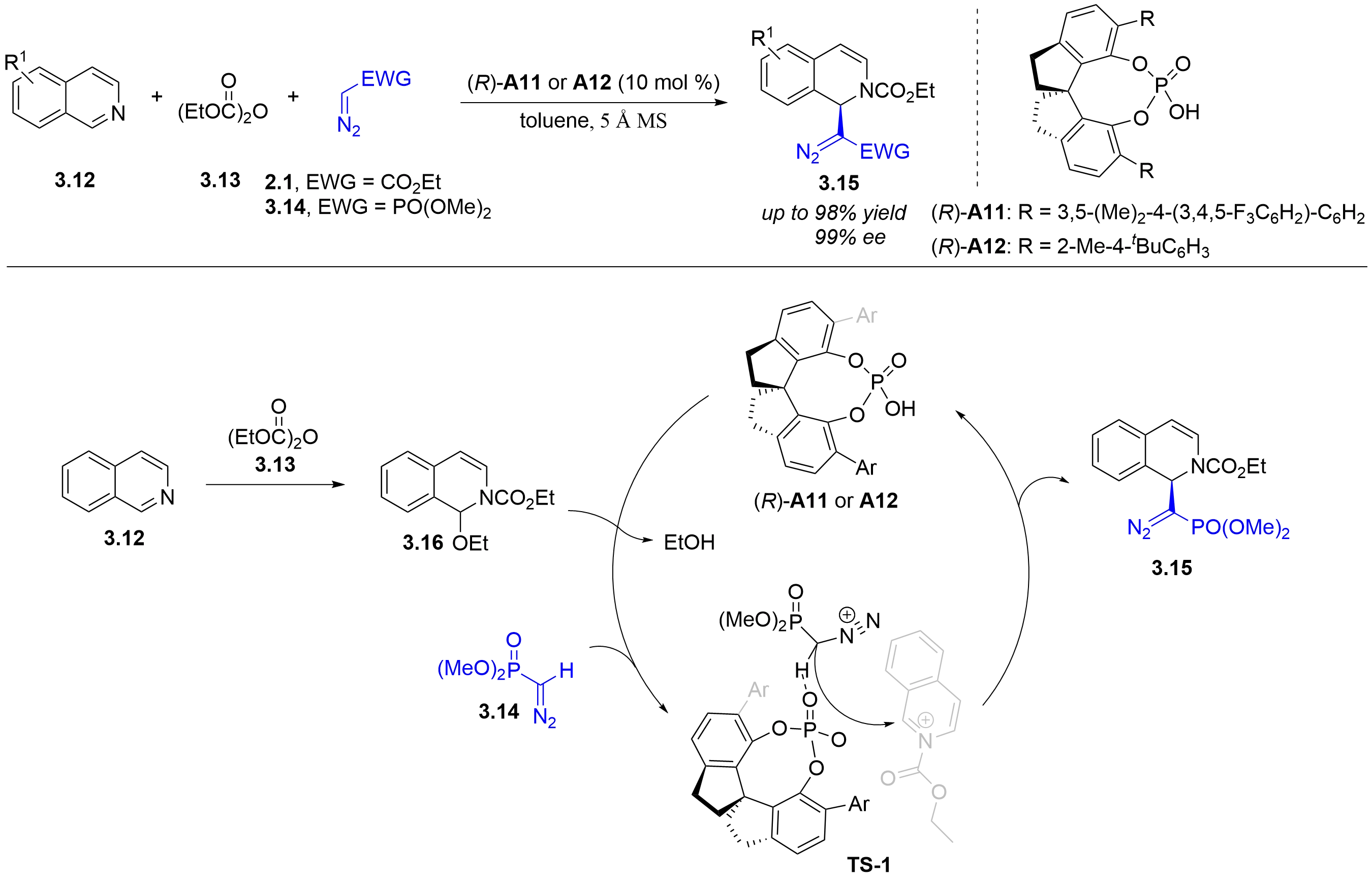
Figure 22. Chiral phosphoric acid-catalyzed asymmetric acyl-Mannich reaction of isoquinolines with α-diazomethyl)phosphonate and ethyl α-diazoacetate.
In 2018, the Singh group reported a chiral phosphoric acid-catalyzed asymmetric Mannich-type reaction of α-diazoacetate with in situ generated N-acyl ketimines, derived from 3-hydroxyisoindolinones[35]. Inspired by this work, the Zhao group further expanded the substrate scope from 3-hydroxyisoindolinones to 2-aryl-3H-indol-3-ones in 2025 (Figure 23)[36]. The reaction is initiated by the interaction of ethyl diazoacetate 2.1 and 2-aryl-3H-indol-3-one 3.17a with chiral phosphoric acid via hydrogen bonding to generate intermediate INT VI. In INT VI, the hydroxyl group of the chiral phosphoric acid protonates the imine unit of 3.17a through hydrogen bonding, forming a chiral indolin-3-one iminium ion. Simultaneously, the α-hydrogen of diazoacetate 2.1 interacts with the phosphoryl oxygen of the chiral phosphoric acid, enhancing its nucleophilicity. Subsequently, nucleophilic attack of the activated 2.1 onto the Si-face of the indolin-3-one iminium ion occurs via transition state TS-2, affording intermediate INT VII. Finally, intramolecular deprotonation of INT VII yields the final product 3.18a.
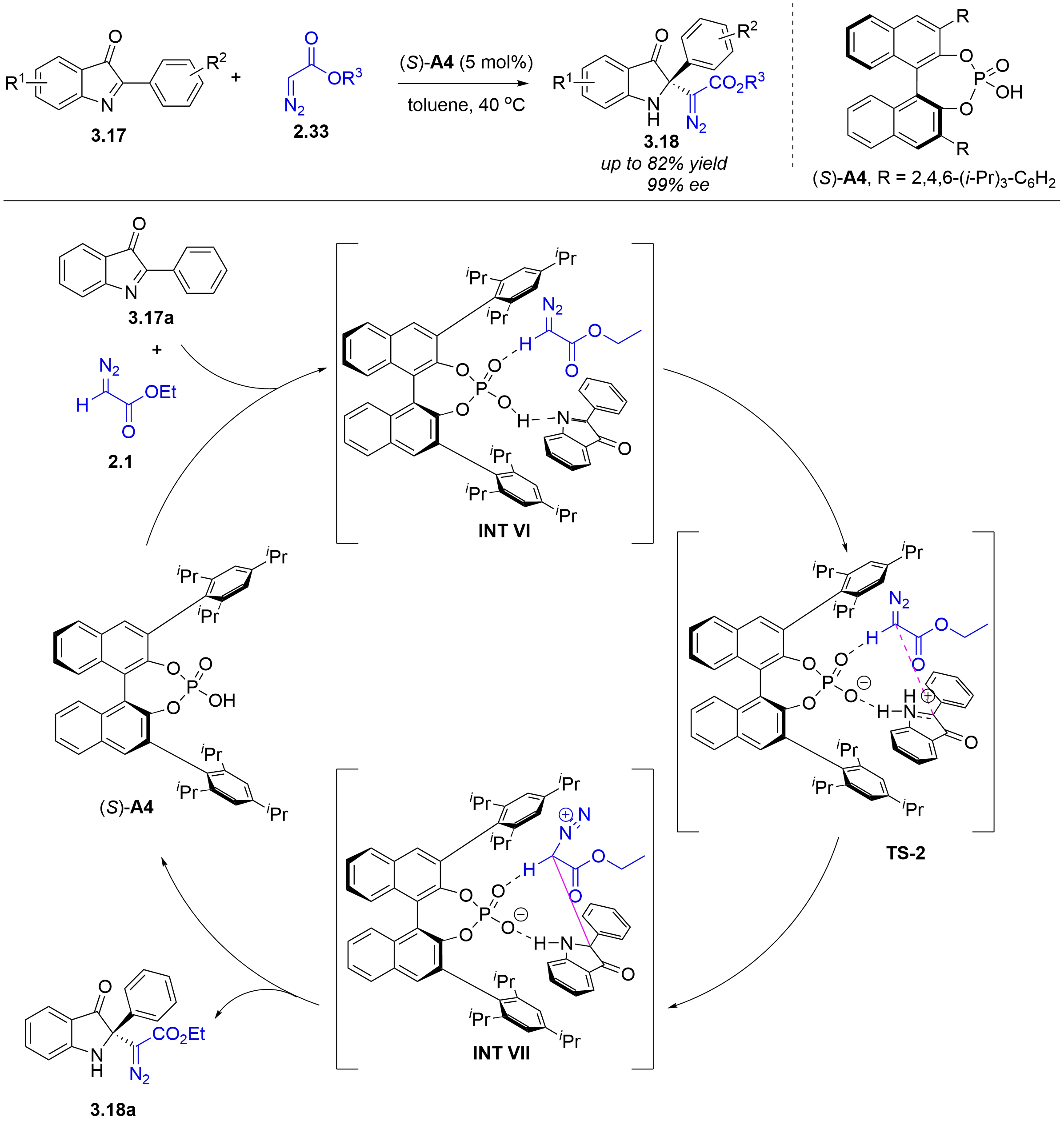
Figure 23. Chiral phosphoric acid-catalyzed asymmetric Mannich-type reaction of α-diazoacetate with 2-aryl-3H-indol-3-ones.
3.2 Chiral dicarboxylic acid-catalyzed asymmetric Mannich-type reaction
By combining two carboxylic acids and an axially chiral binaphthyl moiety, the Maruoka group designed a new type of axially chiral binaphthalenedicarboxylic acid catalyst in 2007, which was designed to address the issues of poor reactivity of carboxylic acid and difficulty in building an effective chiral recognition site around them. It was successfully applied to the asymmetric Mannich-type reaction of arylaldehyde N-Boc imines 2.23 with tert-butyl 2-diazoacetate 2.33a or dimethyl diazomethylphosphonate 3.14
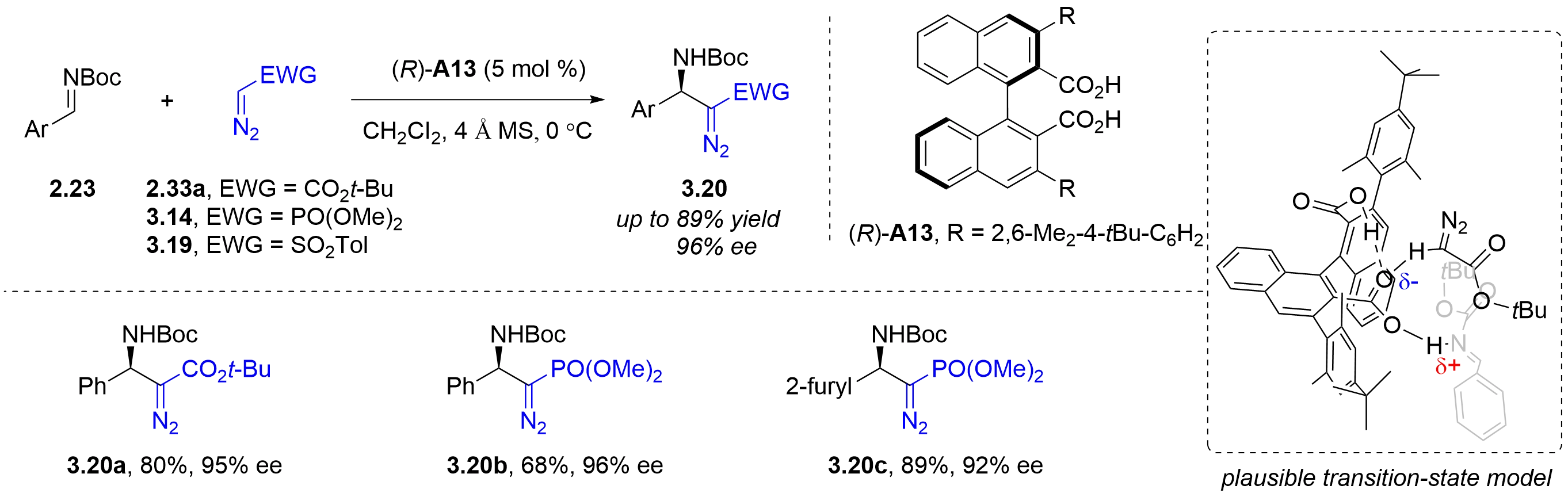
Figure 24. Chiral dicarboxylic acid-catalyzed asymmetric Mannich-type reaction of aromatic aldehyde N-Boc imines with diazo compounds.
While cyclic azomethine imines were widely utilized in asymmetric additions, acyclic azomethine imines remained underdeveloped. In 2011, the Maruoka group employed acyclic azomethine imines as novel prochiral electrophiles, which were generated in situ from the reaction of aldehydes 3.21 with N′-benzylbenzoylhydrazide 3.22 (Figure 25)[39]. Using an axially chiral dicarboxylic acid catalyst
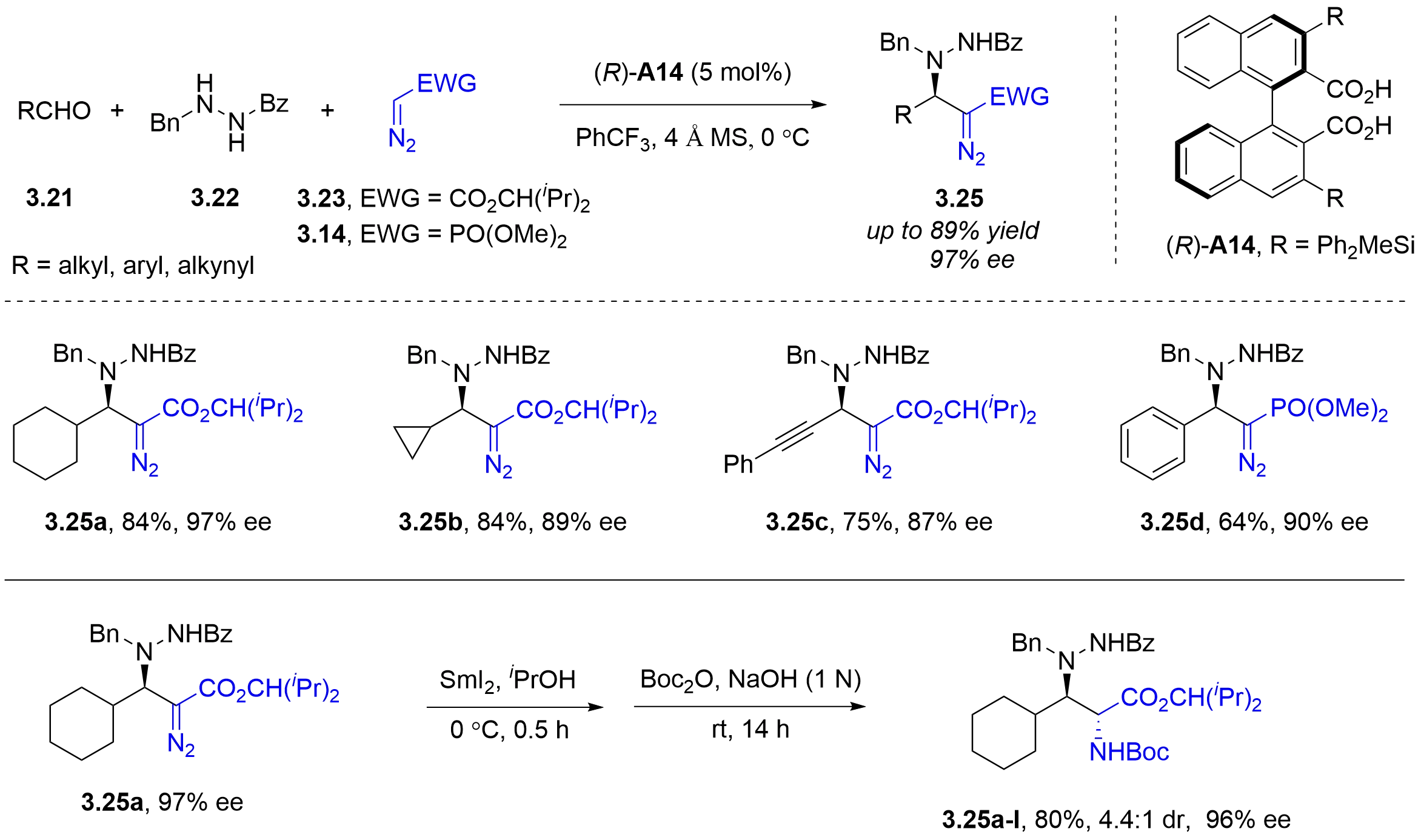
Figure 25. Chiral dicarboxylic acid-catalyzed asymmetric three-component Mannich-type reaction of aldehydes and N′-benzylbenzoylhydrazide with diazo compounds.
3.3 Chiral Lewis acid-catalyzed asymmetric Mannich-type reaction
The lower reactivity of α-diazomethylphosphonates has largely confined asymmetric Mannich-type reactions to aldimines. Their reactions with less reactive ketimines remain highly challenging. In 2016, the Peng group reported a chiral silver
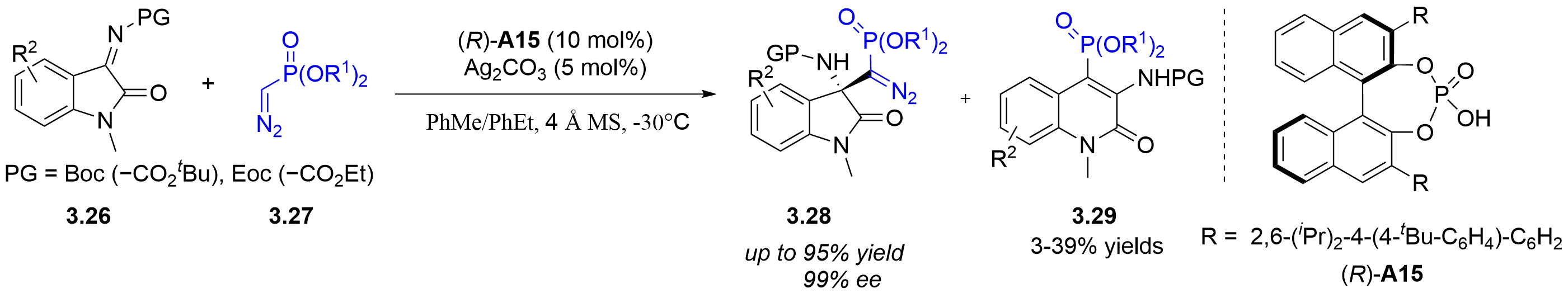
Figure 26. Chiral silver phosphate-catalyzed asymmetric Mannich-type reaction of isatin-derived ketimines with α-diazomethylphosphonates.
In 2018, the Wang/He group reported a Lewis acidic gold(I)-catalyzed asymmetric Mannich-type reaction of α-diazoacetates 2.33 with N-sulfonyl cyclic ketimines 3.30 (Figure 27)[41]. This reaction was catalyzed by a chiral phosphine ligand [(S)-BINAP] with an achiral
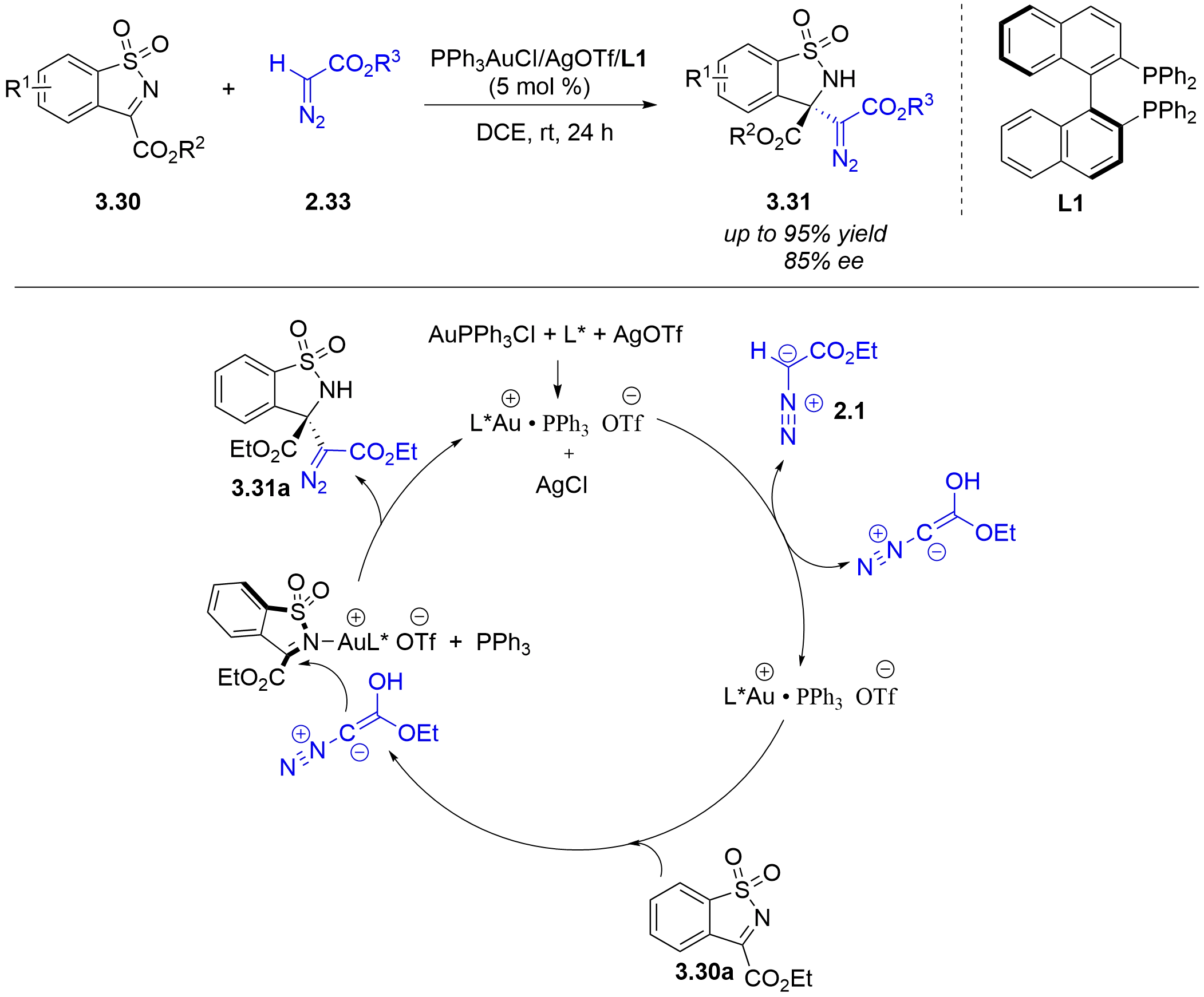
Figure 27. Chiral Au(I)/(S)-BINAP asymmetric Mannich-type reaction of N-sulfonyl cyclic ketimines with α-diazoacetates. BINAP: 2,2′-diphenylphosphino-1,1′-binaphthyl.
In 2020, the Dixon group reported a silver(I)-catalyzed asymmetric Mannich-type reaction of α-diazoacetates with N-Boc aldimines
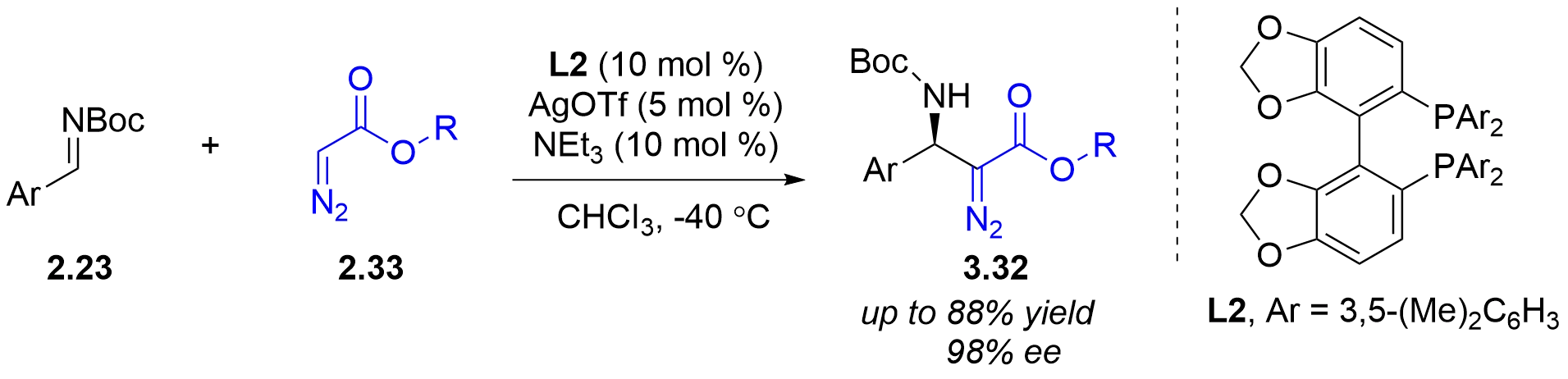
Figure 28. Chiral Ag(I)/(R)-DM-SEGPHOS asymmetric Mannich-type reaction of N-Boc aldimines with α-diazoacetates.
In 2021, the Wang group reported a chiral silver imidodiphosphate-catalyzed asymmetric tandem reaction of o-alkynylbenzaldehydes 3.33, aromatic amines 3.7, and ethyl α-diazoacetate 2.1 or α-diazomethylphosphonates 3.14 (Figure 29)[43]. Various chiral
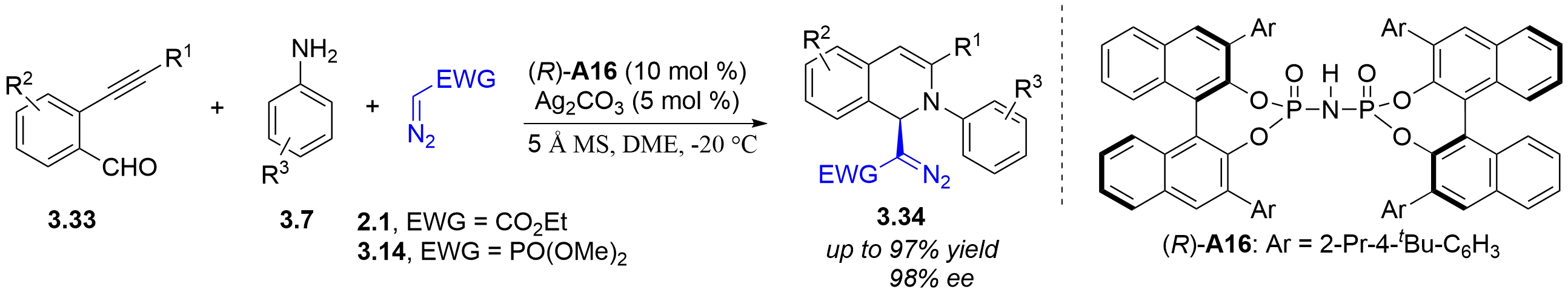
Figure 29. Chiral silver imidodiphosphate-catalyzed asymmetric tandem reaction of o-alkynylbenzaldehydes, aromatic amines and diazo compounds.
4. Conclusion and Perspective
In this review, recent developments in transition-metal-free chiral Brønsted/Lewis acid-catalyzed asymmetric diazo
| imine | diazo | cis-aziridine | trans-aziridine | β-amino-α-diazo | catalyst | Refs. |
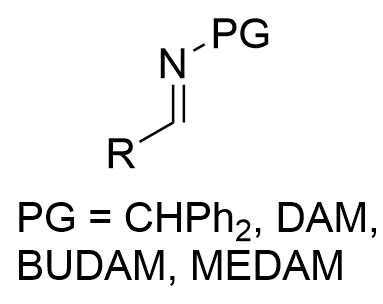 | 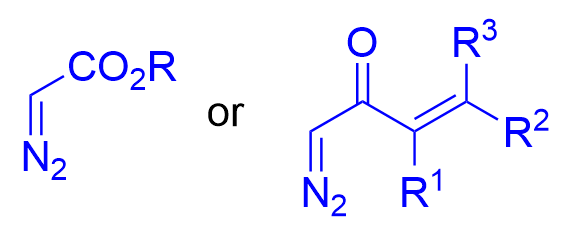 | √ | VANOL/VAPOLB(OPh)3 | [10-15] | ||
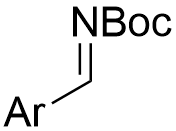 | 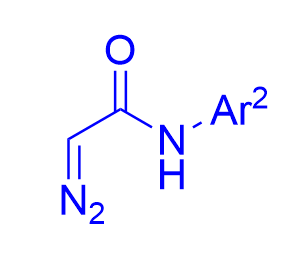 | √ | chiral dicarboxylic acid | [18] | ||
| chiral phosphoric acid | [20] | |||||
| combination chiral diol with2-boronobenzoic acid | [24] | |||||
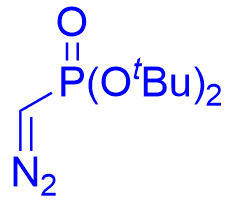 | √ | chiral phosphoric acid | [31] | |||
 | √ | chiral phosphoric acid | [33] | |||
 | √ | chiral dicarboxylic acid | [37,38] | |||
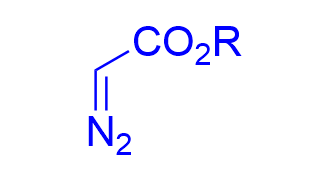 | √ | Ag(I)(R)-DM-SEGPHOS | [42] | |||
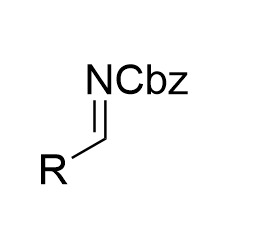 | 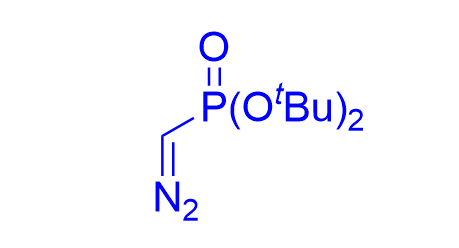 | √ | chiral phosphoric acid | [31] | ||
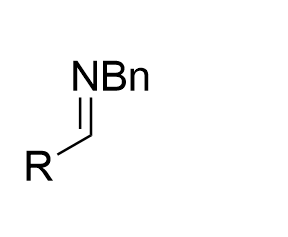 | 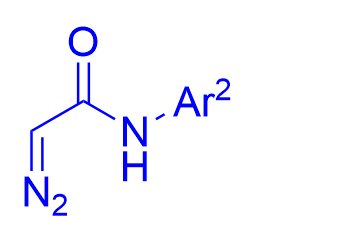 | √ | combination chiral diol with2-boronobenzoic acid | [24] | ||
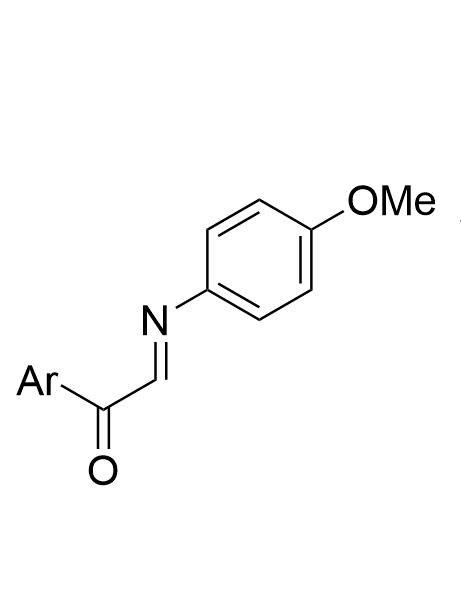 | 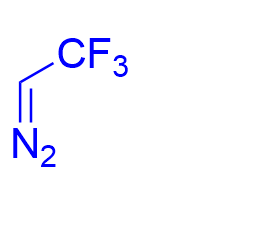 | √ | chiral phosphoric acid | [21] | ||
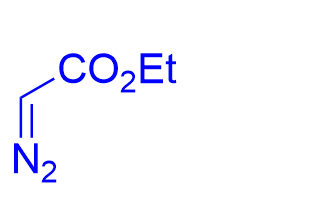 | √ | chiral phosphoric acid | [19] | |||
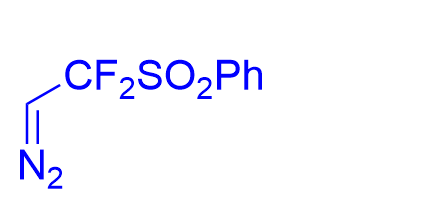 | √ | combinationchiral disulfonimide with2-boronobenzoic acid | [25] | |||
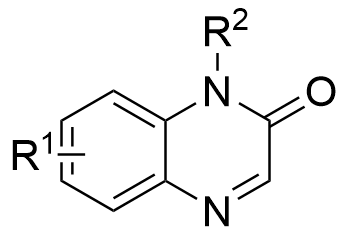 | 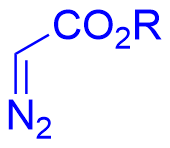 | √ | Chiral phosphoric acid | [22] | ||
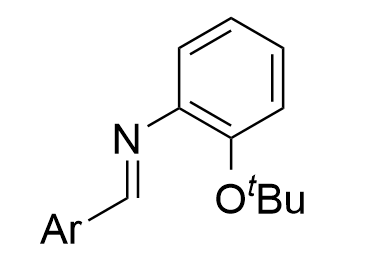 | 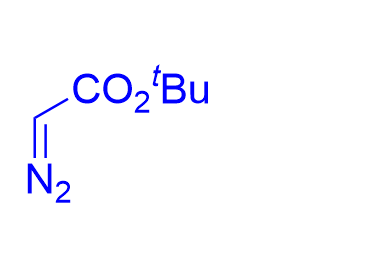 | √ | chiral N-triflylphosphoramide | [23] | ||
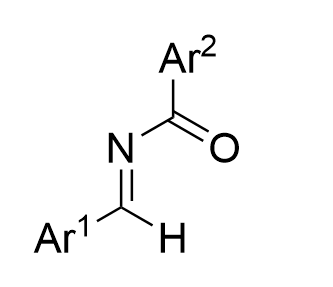 | 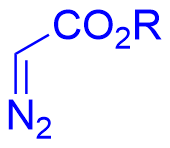 | √ | Chiral phosphoric acid | [30] | ||
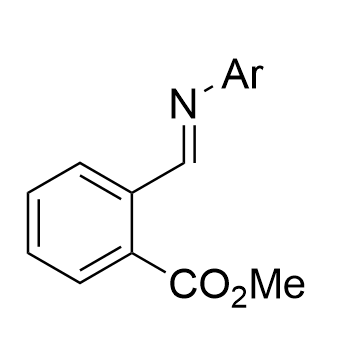 | 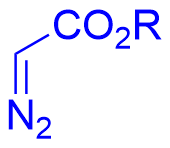 | √ | Chiral phosphoric acid | [32] | ||
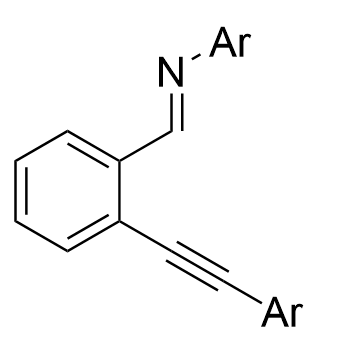 | 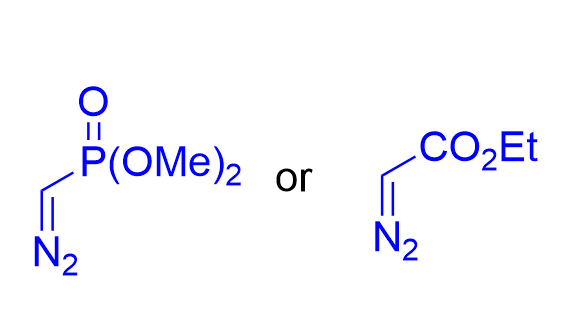 | √ | chiral silver imidodiphosphate | [43] |
Despite the remarkable progress achieved to date, several critical challenges remain to be addressed. First, the enantioselectivity of products derived from aliphatic imine substrates still needs further improvement. Second, high catalyst loadings (5-10 mol %) are typically needed, and thus catalytic efficiency must be enhanced. Third, chiral Lewis acid catalytic systems remain underdeveloped, with most relying on noble metals such as Au and Ag. Earth-abundant, cost-effective first-row transition metals, including Fe, Ni, Sc, and Cu, offer promising alternatives worthy of systematic exploration in asymmetric diazo nucleophile–imine reactions. Fourth, much attention is currently concentrated on the construction of central chirality, so the design and synthesis of planar, axial, helical, or inherently chiral compounds is in great demand. It is worth mentioning that the enamines generated via 1,2-migration are ubiquitous byproducts across various catalytic systems. Optimizing the catalytic system or rationally designing substrates to achieve controlled synthesis of enamines, particularly chiral enamines, might represent an important direction in this field. It is our hope that this review will stimulate further investigations to address these outstanding synthetic challenges and foster continued growth in the development of catalytic asymmetric diazo nucleophile-imine reactions.
Authors contribution
Xie PR, Luo YP, Xie ZY: Literature search, writing-original draft.
Pan C: Conceptualization, literature search, writing-original draft, writing-review & editing.
Xuan J: Conceptualization, writing-review & editing.
Conflict of interests
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Nos. 22201002, 22571002 and 21971001), the Distinguished Young Research Project of Anhui Higher Education Institution (Grant No. 2023AH020003), and the Natural Science Foundation of Anhui Province (Grant No. 2408085MB041).
Copyright
© The Author(s) 2026.
References
-
1. Carey JS, Laffan D, Thomson C, Williams MT. Analysis of the reactions used for the preparation of drug candidate molecules. Org Biomol Chem. 2006;4(12):2337-2347.
-
2. Nugent T, El-Shazly M. Chiral amine synthesis–recent developments and trends for enamide reduction, reductive amination, and imine reduction. Adv Synth Catal. 2010;352(5):753-819.[DOI]
-
3. Wang Z, Yang C, Chen J, Yang F, Khan R, Yang Y, et al. Asymmetric transfer hydrogenation of trifluoromethylated imines to chiral α-trifluoromethylated amines with alcohol as the hydrogen source. Asian J Org Chem. 2021;10(6):1530-1535.[DOI]
-
5. Pan C, Gu Z. Synthesis of atropisomers via transition-metal-catalyzed enantioselective carbene transformations. Trends Chem. 2023;5(9):684-696.[DOI]
-
6. Ahmad Khan H, Szostak M, Sivasankar C. Diazo compounds: Synthesis, carbene generation and reactivity. Org Biomol Chem. 2026;24(4):734-766.[DOI]
-
7. Olaizola I, Ochoa de Retana AM, de los Santos JM. Recent developments in catalytic asymmetric aziridination. Top Curr Chem. 2025;383(4):40.[DOI]
-
8. Kaur N. Synthesis of aziridines and oxaziridines from imines. Amsterdam: Elsevier; 2024.[DOI]
-
10. Antilla JC, Wulff WD. Catalytic asymmetric aziridination with a chiral VAPOL–boron lewis acid. J Am Chem Soc. 1999;121(21):5099-5100.[DOI]
-
11. Antilla JC, Wulff WD. Catalytic asymmetric aziridination with arylborate catalysts derived from VAPOL and VANOL ligands. Angew Chem Int Ed. 2000;39(24):4518-4521.[DOI]
-
14. Deng Y, Lee YR, Newman CA, Wulff WD. Aziridinyl vinyl ketones from the asymmetric catalytic aziridination reaction. Eur J Org Chem. 2007;2007(13):2068-2071.[DOI]
-
22. Xie ZY, Xu CL, Zhu Y, Tai JP, Liu Y, Duan HT, et al. Enantioselective synthesis of aziridine-fused polycyclic architectures via a catalytic asymmetric aza-Darzens reaction and reductive cyclization. Org Chem Front. 2026;13(2):440-446.[DOI]
-
27. Zhang Z, Gevorgyan V. Visible light-induced reactions of diazo compounds and their precursors. Chem Rev. 2024;124(11):7214-7261.[DOI]
-
28. Cai B, Xuan J. Visible light-promoted transformation of diazo compounds via the formation of free carbene as key intermediate. Chin J Org Chem. 2021;41(12):4565.[DOI]
-
29. Xie Y, Xuan J. Photocatalytic reactions involving diazo compounds as radical precursors. Chin J Org Chem. 2022;42(12):4247.[DOI]
-
39. Hashimoto T, Kimura H, Kawamata Y, Maruoka K. Generation and exploitation of acyclic azomethine imines in chiral Brønsted acid catalysis. Nat Chem. 2011;3(8):642-646.[DOI]
-
41. Zhou M, Su Q, Addepalli Y, He Y, Wang Z. An asymmetric Mannich reaction of α-diazocarbonyl compounds and N-sulfonyl cyclic ketimines catalyzed by complexes generated from chiral and achiral phosphines with gold(i). Org Biomol Chem. 2018;16(16):2923-2931.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite