Abstract
Magnesium alloys are primarily composed of magnesium, with the additions of elements such as calcium, yttrium, and zinc. In the human physiological environment, they gradually degrade, and their degradation products can be absorbed, exhibiting excellent biocompatibility, mechanical properties comparable to bone tissue, and degradability; thus, they hold broad prospects in orthopedics. Nanotechnology involves the design and manufacture of materials, devices, and systems with unique physical, chemical, and biological properties by controlling the arrangement and interactions of atoms, molecules, or nanostructural units at the nanoscale (1-100 nm). The integration of these two technologies shows exceptional potential for orthopedic regenerative repair. Nanotechnology significantly enhances the mechanical performance, bioactivity, antibacterial properties, and controlled degradation of biodegradable magnesium alloys through various approaches, while biodegradable magnesium alloys provide an ideal biomaterial carrier for nanotechnology, enabling the better exertion of its advantages in bone tissue repair. This review summarizes the innovations arising from the fusion of magnesium alloys and nanotechnology in bone repair, aiming to advance the evolution of orthopedic medical devices, promote a shift in clinical treatment paradigms toward personalized and precise therapy, and ultimately deliver superior and more efficient therapeutic options for patients with orthopedic conditions, thereby improving human health and quality of life.
Keywords
1. Introduction
Traditional bone repair faces numerous challenges and technical limitations, yet the clinical demands for efficient bone repair remain urgent. The limitations of conventional approaches severely restrict the efficacy of bone defect treatment, highlighting the clinical significance of developing novel bone repair materials and technologies[1,2].
Therefore, this review aims to systematically summarize and critically evaluate the recent advancements in leveraging nanotechnology to overcome the inherent limitations of biodegradable Mg alloys for bone repair. We focus on the principal strategies of nano-integration, including nano-alloying, nanostructured coatings, and nanocomposites, and elucidate their synergistic effects on modulating degradation behavior, enhancing mechanical integrity, and promoting osteogenesis. Furthermore, this review discusses the molecular mechanisms underlying these enhancements and explores future perspectives, such as intelligent implants and personalized medicine, heralded by this convergent technology.
2. Challenges of Traditional Bone Repair
2.1 The dilemma of autologous bone grafting as the “gold standard”
Autologous bone grafting has long been regarded as the “gold standard” for bone repair; however, its limitations cannot be ignored. Donor site morbidity is a major concern, with potential complications such as pain, infection, and fractures at the harvesting sites. Additionally, the limited supply of autologous bone fails to meet the demands of large bone defect repair, and the risk of secondary surgery increases patient suffering and raises medical costs[3-5]. A direct comparison between autologous bone grafting and conventional artificial materials highlights the clinical dilemma, as illustrated in Table 1. Kim et al.[6] conducted a retrospective cohort study to analyze the efficacy and complications of autologous iliac crest bone grafting in the treatment of large bone defects of the lower extremities, further confirming the clinical value of autologous bone grafting and the potential donor-site risks, such as pain and fractures at the bone harvest site.
| Traditional Method | Advantages | Disadvantages |
| Autologous bone grafting | Excellent osteoconductivity, osteoinductivity, and bioactivity | Donor site morbidity, limited supply, need for secondary surgery |
| Conventional artificial materials | Abundant sources, mass production capability | Insufficient mechanical support, poor degradation matching, weak osteoinductivity |
2.2 Limitations of conventional artificial bone materials
Traditional artificial bone materials, including calcium-based ceramics, inert metals, and polymers, exhibit significant deficiencies in mechanical support, degradation matching, and osteoinductivity[7-9]. The main types of conventional artificial bone materials, along with their key characteristics and application limitations, are summarized in Table 2. Andrussow et al.[10] conducted a review and analysis of biodegradable metallic biomaterials and traditional inert metallic materials, concluding that while traditional inert metallic materials (e.g., stainless steel and cobalt-chromium alloys) possess favorable mechanical properties, they exhibit poor bioactivity, fail to form adequate osseointegration with bone tissue, and may induce inflammatory reactions with long-term in vivo implantation.
| Material Type | Mechanical Support | Degradation Matching | Osteoinductivity | Application Limitations |
| Calcium-based ceramics | High brittleness, low strength | Slow degradation rate | Weak | Unable to meet complex bone defect repair needs |
| Inert metals | High strength, good stability | Non-degradable | None | May cause stress shielding and foreign body reactions |
| Polymers | Good plasticity, biocompatibility | Poor mechanical properties, potentially toxic degradation products | Insufficient | Poor long-term stability |
3. Characteristics of Biodegradable Magnesium Alloys
This review focuses on calcium(Ca), yttrium(Y), and zinc(Zn), three extensively investigated alloying elements with well-established biocompatibility, which serve as representative elements widely utilized in biomedical magnesium alloys.
Magnesium alloys are primarily magnesium-based, with the addition of alloying elements such as Ca, Y, and Zn. The addition of these elements can significantly affect the microstructural characteristics of magnesium alloys, such as crystal structure and phase composition, thereby influencing their mechanical properties and degradation behavior. As magnesium is an essential element in the human body that participates in numerous physiological processes, it endows magnesium alloys with good biocompatibility. Magnesium alloys possess degradability, which is a key characteristic distinguishing them from traditional bone repair materials. Magnesium alloys offer new hope for addressing the limitations of traditional bone repair materials and are therefore referred to as “revolutionary metals”[11-14].
3.1 Advantages of magnesium alloys
Magnesium alloys exhibit excellent biodegradability and biocompatibility, along with suitable mechanical properties and an elastic modulus close to that of human bone, which effectively reduces the stress-shielding effect. Moreover, magnesium is an essential nutrient for the body, participating in nearly all metabolic processes such as protein synthesis, enzyme activation, and the regulation of neuromuscular and central nervous system activities[15-18].
Biologically, magnesium ions activate the AKT/β-catenin signaling pathway, thereby promoting osteoblast proliferation, differentiation, and mineralization and accelerating new bone formation (Figure 1)[19,20].
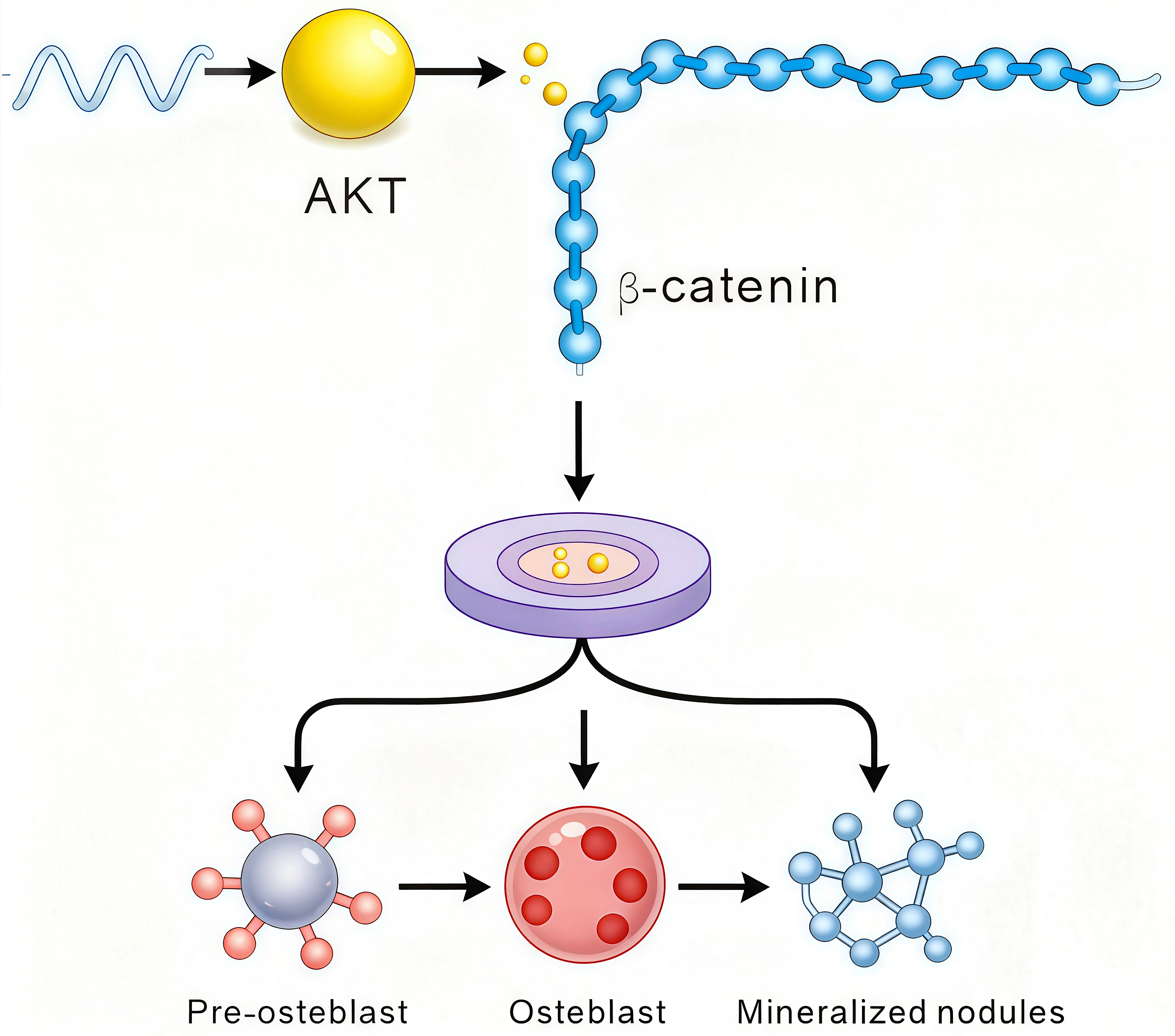
Figure 1. Schematic diagram of the mechanism by which magnesium ions activate the AKT/β-catenin signaling pathway to promote the proliferation, differentiation, and mineralization of osteoblasts.
3.2 Clinical application bottlenecks of magnesium alloys
Studies have confirmed that magnesium alloys degrade both in vitro and in vivo, releasing magnesium ions that provide new insights into bone repair and osteonecrosis treatment. However, rapid degradation rates may lead to insufficient mechanical support before bone healing is complete, and pH fluctuations during degradation can affect cellular activity, potentially triggering inflammatory responses and tissue damage[14,21-23].
To modulate the degradation rate and improve the performance of biomedical magnesium alloys, several key methods have been established. These include surface modification (e.g., electrochemical deposition, ion implantation, and chemical conversion), alloy composition optimization, and extrusion process control .
When a magnesium alloy is subjected to a specific voltage in an electrolyte, the high-temperature micro-arc discharge on its surface forms a porous oxide layer through a method known as micro-arc oxidation (MAO) or plasma electrolytic oxidation (PEO)[24]. Studies have reported that the inclusion of CeO2 nanoparticles in the PEO coating greatly improved the corrosion resistance and biocompatibility of a Mg-Zn-Y-Nd alloy[25].
4. Nanotechnology Upgrading Bone Repair Materials
Nanotechnology provides new opportunities for the precise regulation and functional enhancement of bone repair materials by manipulating their structures and properties at the nanoscale, thereby simulating natural bone characteristics, increase the specific surface area and surface activity, and strengthen interactions with biological tissues.
4.1 Applications of nanotechnology in bone repair materials
Nanocoating technology can improve the surface properties of materials and enhance their biocompatibility and osteoinductive capacity[26-29]. Nanofiber technology can be used to construct a biomimetic extracellular matrix, providing a favorable microenvironment for cell adhesion, proliferation, and differentiation[29].
4.2 Precision construction of nanoscale biomimetic structures
Ultralow-temperature deposition 3D printing technology enables not only precise control of macroscopic porosity but also
In addition, compared with traditional high-temperature printing technologies, LTD 3D printing technology has significant advantages. The mechanical strength of bone repair materials printed using this technology can reach 52.01 MPa, which can meet the mechanical requirements during bone repair[33,34].
4.3 Synergistic effects of nanoscale components
The nanoscale composite design of a biodegradable polymer (poly lactic-co-glycolic acid, PLGA), a bioceramic (β-tricalcium phosphate, β-TCP), and metallic magnesium achieves synergistic enhancement among its components. PLGA possesses good biocompatibility and biodegradability; however, it generates acidic products during degradation that may exert adverse effects on surrounding tissues. In contrast, magnesium releases alkaline substances during degradation, which can neutralize the acidic degradation products of PLGA and maintain the stability of the local microenvironment. β-TCP is a bioactive ceramic with excellent osteoinductivity, capable of promoting osteoblast adhesion, proliferation, and differentiation[35]. This nanoscale composite design provides new insights and methods for bone repair materials and is expected to significantly improve the efficacy of bone repair[36,37].
5. Integration of Biodegradable Magnesium Alloys and Nanotechnology
The integration of biodegradable magnesium alloys and nanotechnology has significantly enhanced the performance of bone repair materials. Nanotechnology modifies magnesium alloys to precisely control degradation rates and mechanical properties, increase tissue contact area, and enhance bioactivity. Consequently, magnesium alloys serve as ideal carriers for nanomaterial
5.1 Approaches of integration of magnesium alloys with nanotechnology
5.1.1 Nanoscale alloying element doping
By incorporating nanoscale alloying elements into magnesium alloys, the high reactivity and diffusivity of the nanoparticles enable their uniform distribution within the magnesium matrix during alloy preparation. Nanoscale alloying can be achieved using techniques such as mechanical alloying and smelting. Nanoscale alloying element doping significantly refines the grain structure of magnesium alloys, thereby enhancing their mechanical properties, including yield strength and hardness, via the Hall-Petch relationship. It also inhibits dislocation movement to improve deformation resistance, modifies electrochemical properties to enhance corrosion resistance, and slows degradation rates[40]. Nanoparticles can achieve dispersion strengthening by pinning grain boundaries and impeding dislocation motion, while their uniform distribution also facilitates the formation of a more stable passive film, thereby slowing local corrosion and improving overall degradation uniformity.
The nanostructuring process may influence the distribution patterns and release kinetics of rare earth elements within the magnesium matrix, thereby modulating their multiple biological effects such as antioxidant, anti-inflammatory, and osteogenic activities.
5.1.2 Construction of nanostructured surface coatings
Nanostructured coatings are deposited on the surface of degradable magnesium alloys using techniques such as physical vapor deposition and chemical vapor deposition. Bioactive materials, including nano-hydroxyapatite (nHA) and titanium dioxide nanoparticles (nano-TiO2), can be selected as coating materials.
Nanostructured coatings enhance the bioactivity and osteogenic inductive capacity of degradable magnesium alloys. Their high specific surface area and abundant active sites enable the adsorption of more extracellular matrix components, thereby providing a favorable growth microenvironment for osteoblasts. Additionally, these coatings improve the corrosion resistance of magnesium alloys and slow their degradation rate[41,42].
5.1.3 Preparation of nanocomposite magnesium alloy materials
Nanomaterials, such as nanobioceramic particles or carbon nanotubes, are compounded with a magnesium alloy matrix via processes including powder metallurgy and stir casting. In composite design, factors such as the content, size, and distribution of nanomaterials must be comprehensively considered for their impact on the composite’s[43,44].
The key performance indicators of nanocomposite degradable magnesium alloy materials, including mechanical properties, antibacterial performance, and osteogenic induction capacity, were significantly improved. Nanobioceramic particles enhance the composite’s hardness, modulus, wear resistance, and fatigue resistance, while carbon nanotubes provide mechanical reinforcement, thereby increasing its tensile and yield strengths. The incorporation of nanomaterials also endows the composite with antibacterial properties, potentially through interactions between active components on the nanomaterial surface and bacterial cell walls, which disrupt bacterial cell structures and inhibit bacterial growth and reproduction. Additionally, nanobioceramic particles can release bioactive ions, thereby promoting the activation of osteogenesis-related signaling pathways and inducing bone tissue regeneration in the body.
For fixation implants (such as screws), nanostructuring can significantly enhance their surface bioactivity, corrosion resistance, and early-stage osseointegration capacity. Particularly in high-stress environments, nanostructures can improve interfacial bonding strength and reduce the risk of loosening.
5.1.4 Comparative analysis of different nanoscale integration strategies
Different nanoscale integration strategies are suitable for varying clinical needs and types of implants. Table 3 summarizes the advantages and limitations of nano-alloying, nanocoatings, and nanocomposite materials in terms of mechanical enhancement, degradation control, bioactivity, and fabrication processes.
| Strategy | Advantages | Disadvantages | Applicable Scenarios |
| Nano-alloying | Enhanced strength, refined grain structure | Difficulties in achieving uniform dispersion | High-load-bearing implants |
| Nanostructured Coatings | Improved bioactivity, antibacterial properties | Long-term adhesion stability under physiological stress | Surface-functionalized implants |
| Nanocomposites | Excellent comprehensive performance, high designability | Complex manufacturing processes, high cost | Complex structured bone scaffolds |
5.2 Synergistic effects of magnesium alloys and nanotechnology
5.2.1 Dynamic balance of mechanics-degradation-regeneration
Materials integrating degradable magnesium alloys with nanotechnology ingeniously achieve a balance between “complete degradation within 6-9 months” and “mechanical support in the early stage of osteogenesis”. In the early stage of osteogenesis, through rational structural design and precise regulation via nanotechnology, the materials possess sufficient mechanical strength to stably withstand impact forces during surgical procedures, thereby providing reliable mechanical support to bone tissue[45,46].
Over time, these materials begin to degrade at a predetermined rate. Within 6-9 months, they are completely degraded and fully absorbed by the human body. The magnesium ions released during degradation participate in new bone formation and normal physiological metabolism, thereby accelerating bone repair[10,45]. Degradation products are primarily excreted via urine, leaving no in vivo residues and thus avoiding foreign body reactions caused by material retention. Nanocoatings or nanocomposite designs enable precise regulation of material degradation rates, allowing them to align with the bone healing cycle and thereby preventing functional failure caused by premature or delayed degradation.
5.2.2 Dual breakthroughs in biocompatibility and repair efficiency
Traditional materials, such as Ca-based ceramics, inert metals, and polymeric materials, have numerous limitations in terms of biocompatibility and repair efficiency, which may induce clinical complications (e.g., immune rejection and foreign body reactions). In contrast, integrated innovative materials, leveraging their unique design and properties, significantly improve the success rate of bone repair, effectively alleviate patient suffering and complications, and bring new hope to bone defect repair[46,47].
5.3 Molecular mechanisms of the integration of magnesium alloys and nanotechnology
5.3.1 Activating osteogenesis-related signaling pathways
Magnesium alloys degrade in the physiological environments to release magnesium ions, which activate the AKT/β-catenin signaling pathway[48]. AKT is a crucial protein kinase that promotes cell survival and proliferation. After accumulating intracellularly, β-catenin translocates into the nucleus, regulating gene transcription, enhancing osteoblast proliferation, differentiation, and mineralization, accelerating new bone formation, and providing robust biological support for bone defect repair (Figure 2).
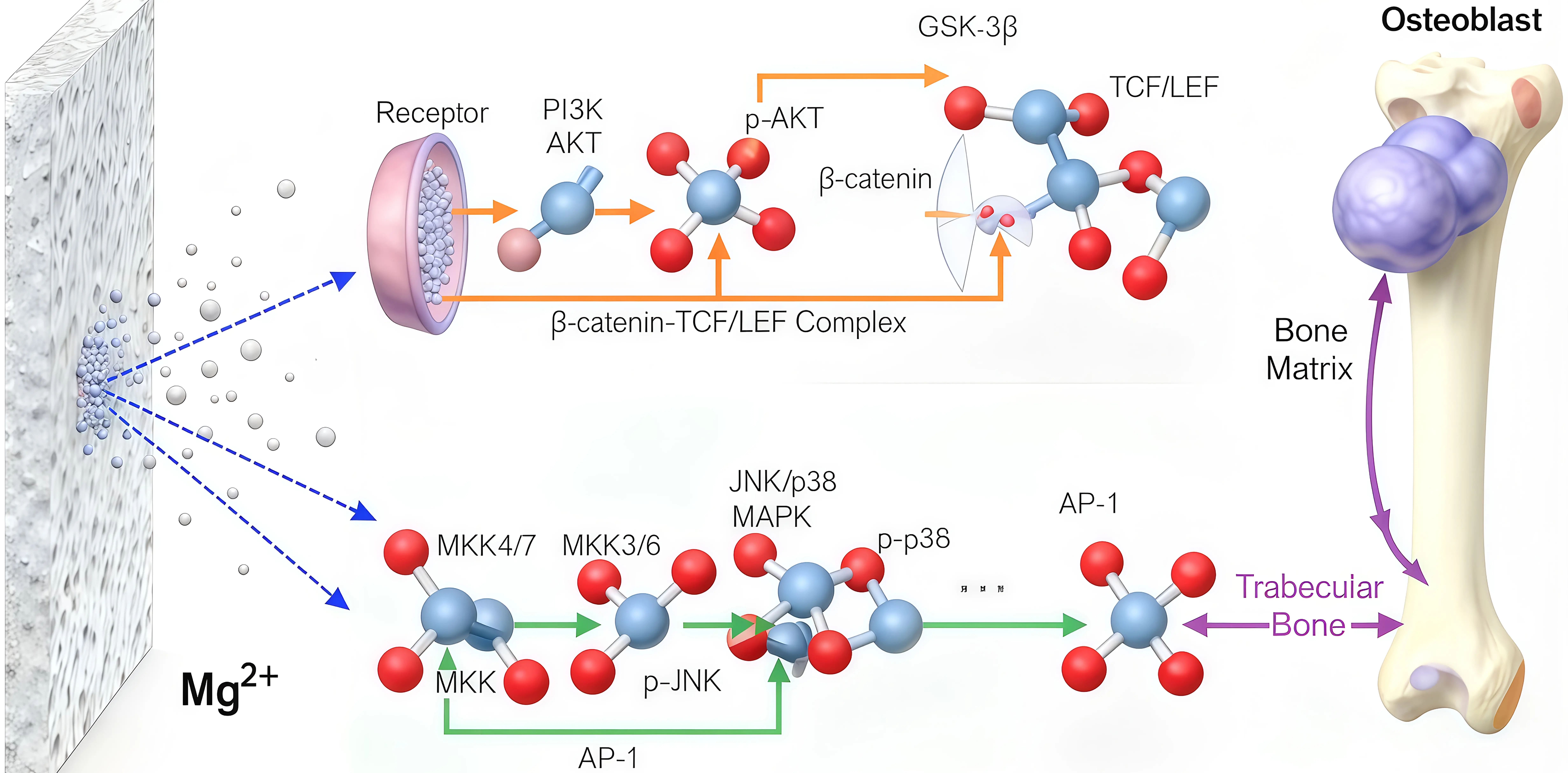
Figure 2. Signaling pathway diagram showing magnesium ions activating the AKT/β-catenin and MAPK signaling pathways to regulate osteoblast function and bone tissue formation.
Magnesium ions can also activate the mitogen-activated protein kinase (MAPK) signaling pathway. The MAPK signaling pathway plays a key role in cellular responses to external stimuli and regulates cell growth, differentiation, and apoptosis. Activation of the MAPK signaling pathway promotes the differentiation and maturation of osteoblasts, increases bone tissue formation, and simultaneously inhibits osteoclast activity to reduce bone resorption, thereby facilitating bone repair (Figure 2)[49].
Nanostructured surfaces can modulate the localized release kinetics of Mg2+, thereby influencing the activation efficiency of the PI3K/AKT signaling pathway. For instance, nanoporous coatings can delay the release of Mg2+, enabling it to maintain effective concentrations over an extended period and thus persistently promote the expression of osteogenic genes.
5.3.2 Interaction between nanomaterials and cells
The high specific surface area and abundance of active sites in nHA coatings enable the adsorption of more extracellular matrix components, such as fibronectin and laminin. These components bind to integrin receptors on the osteoblast surface, activating intracellular signal transduction and promoting osteoblast adhesion, proliferation, and differentiation. Concurrently, ions such as Ca and phosphorus released from nHA coatings participate in the mineralization of bone tissue, thereby enhancing its hardness and strength[50,51].
Nano-titanium dioxide (nano-TiO2) coatings exhibit excellent bioactivity and antibacterial properties. Their antibacterial mechanism primarily involves the generation of reactive oxygen species (ROS) to disrupt bacterial cell walls and membranes, thereby inhibiting bacterial growth and reproduction. Additionally, nano-TiO2 coatings can interact with proteins and receptors on the osteoblast surface, activating intracellular signaling pathways, (e.g., FAK/MAPK pathway) that promote osteoblast adhesion, migration, and differentiation, thus accelerating the repair and regeneration of bone tissue (Figure 3)[52,53].
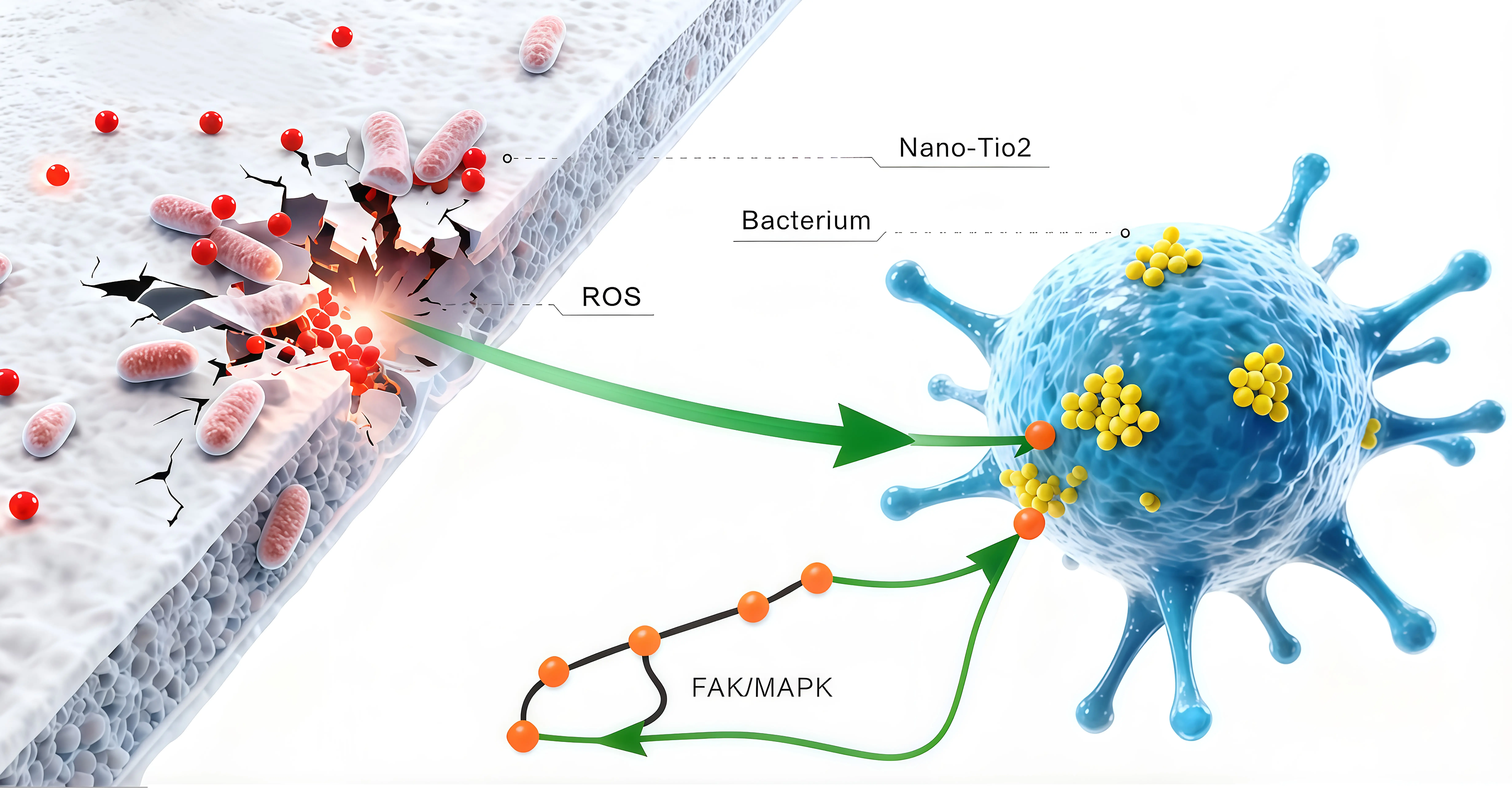
Figure 3. Schematic diagram of the mechanism by which nano-TiO2 exerts antibacterial effects by destroying bacterial structures through ROS and activates the FAK/MAPK pathway to promote osteogenesis. nano-TiO2: nano-titanium dioxide; ROS: reactive oxygen species; FAK: focal adhesion kinase; MAPK: mitogen-activated protein kinase.
5.3.3 Synergistic regulatory mechanisms of nanocomposites
During degradation, the acidic products generated by PLGA in PLGA/β-TCP/Mg nanocomposites neutralize the alkaline substances released by metallic magnesium, maintaining the stability of the local microenvironment and facilitating cellular growth and metabolism. Ca and phosphorus released from β-TCP promote osteoblast mineralization and bone tissue formation. The degradation products of PLGA and β-TCP regulate the composition and properties of the extracellular matrix, influence cellular behavior and function, and enhance bone tissue repair and regeneration[54,55].
Carbon nanotubes (CNTs) in CNT-reinforced nanocomposites possess unique nanostructures and mechanical properties, enabling them to interact with the cytoskeleton of osteoblasts to enhance cellular mechanical stability and signal transduction efficiency. The active sites on the CNT surface adsorb growth factors and extracellular matrix components, promoting osteoblast adhesion, proliferation, and differentiation. Additionally, CNTs regulate intracellular ROS levels, influencing cellular antioxidant capacity and signaling pathway activation to further modulate bone tissue repair and regeneration (Figure 4)[56,57].
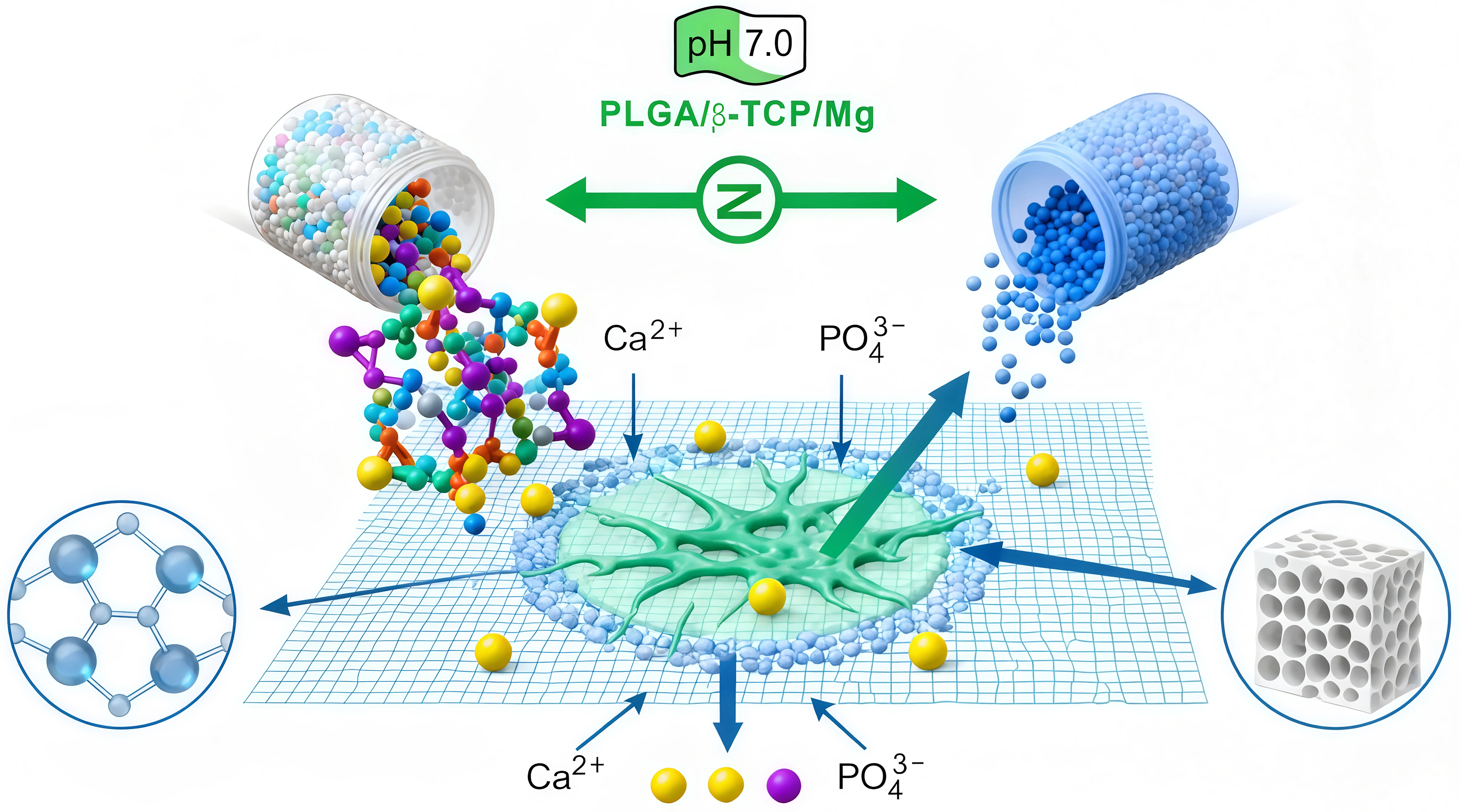
Figure 4. Schematic diagram of the mechanism by which carbon nanotube-reinforced magnesium alloy-based nanocomposites regulate osteoblast mechanical stability, signal transduction, and bone tissue repair and regeneration.
5.3.4 Cellular immunomodulatory mechanisms
Magnesium ions and nanomaterial components released during the degradation of integrated materials (magnesium alloys combined with nanotechnology) exert certain anti-inflammatory effects. Magnesium ions can inhibit the activation of inflammatory cells and the release of inflammatory factors, such as interleukin-1β and tumor necrosis factor-α. Additionally, the surface properties of nanomaterials can regulate the polarization state of macrophages, promoting their transition to an anti-inflammatory phenotype (M2 type) and thereby reducing the adverse effects of inflammatory responses on bone tissue repair[14,56].
By regulating the functions and behaviors of immune cells, integrated materials influence bone tissue repair and regeneration.
5.4 Innovation of integration of magnesium alloys and nanotechnology
5.4.1 Intelligent bone repair
Nanosensor technology has been integrated into degradable magnesium alloy implants to enable real-time monitoring of physiological parameters during bone repair, such as temperature, pressure, pH value, and magnesium ion concentration, with data transmitted via wireless technology. A feedback control system is established based on the monitored data to automatically adjust parameters (e.g., implant mechanical properties and drug release rate) according to bone repair progress, thereby achieving intelligent bone repair and improving therapeutic efficacy and rehabilitation speed. pH-responsive nanocoatings can release antibacterial ions in infected environments, while implants integrated with nanosensors enable wireless monitoring of degradation progress, providing real-time feedback for clinical applications[58,59].
5.4.2 Personalized custom implants
Nanotechnology is used to precisely regulate the microstructure, composition, and properties of magnesium alloys. When combined with computer-aided design and 3D printing technologies, personalized orthopedic implants can be customized based on the patient's individual anatomical structure and disease characteristics. Personalized implants can improve surgical success rates and patient rehabilitation quality, achieve better matching with the patient's bone tissue, provide stable support and good bioactivity, reduce postoperative complications, and enhance patient satisfaction and quality of life[60,61].
5.4.3 New strategies in tissue engineering
Nanotechnology is used to fabricate three-dimensional tissue engineering scaffolds with nanoscale bioactivity. When combined with the supportive properties and biocompatibility of magnesium alloys, this integration provides a new strategy for bone-tissue engineering. The 3D nano-tissue engineering scaffolds synergize with magnesium alloys to promote bone tissue regeneration through multiple mechanisms, such as the release of bioactive factors and induction of cell differentiation. Nanobiologically active signals in the scaffold activate the expression of osteogenesis-related genes and enhance osteoblast function, while the degradation products of magnesium alloys provide essential nutrients for bone regeneration[62,63].
6. Evolution and Summary of the Integrated Technology
6.1 Overview of development: From metallurgical optimization to nano-integration
The development of biodegradable magnesium alloys for bone repair has undergone a remarkable evolution, pivoting around the central challenge of controlling their degradation kinetics to match the bone healing process. The initial research phase primarily focused on metallurgical optimization, exploring the effects of different alloying elements (e.g., Ca, Zn, Y, Nd) on the microstructure, mechanical properties, and corrosion resistance of Mg alloys[11,12]. While this approach yielded improvements, the inherent rapid corrosion of Mg in the physiological environment remained a significant hurdle.
6.2 Limitations of alloy composition control
This limitation catalyzed a strategic shift towards surface modification technologies. Techniques such as MAO, fluoride conversion coating, and polymer coating were extensively investigated to create a physical barrier on the Mg alloy surface[38,51]. These
6.3 Transitional breakthroughs via surface modification
The most recent and transformative paradigm has been the deep integration with nanotechnology, which is the focus of this review. This paradigm moves beyond mere corrosion protection, aiming to create multifunctional and intelligent bone repair implants. As detailed in previous sections, nanotechnology enables precise control at the atomic and molecular level. This is manifested in three key dimensions: (1) Bulk material enhancement through nano-alloying and nanocomposites, which fundamentally improve mechanical strength and homogenize degradation; (2) Surface biofunctionalization through nanostructured coatings (e.g., nHA,
6.4 Nanotechnology-driven intelligent transformation
In summary, the integration of nanotechnology has transformed biodegradable magnesium alloys from a promising but challenging concept into a sophisticated platform technology. It has successfully established a dynamic balance between mechanical support, controlled degradation, and active bone regeneration. The key achievements of this integrated approach include the ability to
6.5 Safety considerations of nanomaterials
While nanotechnology significantly enhances the properties of magnesium alloys, the long-term retention of nanomaterials (CNTs, TiO2) in the body may pose nanotoxicity concerns, such as chronic inflammation, cytotoxicity, or risks of organ accumulation. Future research should systematically evaluate the metabolic pathways, degradation behavior, and long-term effects of nanoparticles on the immune system.
7. Future Perspectives
The integrated innovation of degradable magnesium alloys and nanotechnology in orthopedic repair holds enormous potential and broad prospects. Nanotechnology has significantly enhanced the performance of degradable magnesium alloys, enabling a qualitative leap in their bioactivity, antibacterial properties, osteogenic inductive capacity, and degradation controllability. Despite challenges such as complex material preparation processes and cost control, these issues are expected to be resolved through technological innovations and multidisciplinary collaborations. In the future, with the advancement of trends such as intelligent bone repair, personalized custom implants, and new tissue engineering strategies, this integrated innovation will lead orthopedic repair technology to new heights. It will drive transformations in the orthopedic medical device industry, clinical treatment concepts, and basic research fields, benefiting patients with orthopedic diseases and improving human health and quality of life.
8. Conclusion
This paper reviews the integration of biodegradable magnesium alloys with nanotechnology in bone repair, highlighting their significant advantages in mechanical properties, bioactivity, antimicrobial performance, and controllable degradation. Nanotechnology not only enhances the physicochemical properties of magnesium alloys but also endows them with
Authors contribution
Tan Y, Sun D, Liu R: Writing-original draft.
Zhang P, Yang Q, Alshorman J: Writing-review & editing.
Conflicts of interest
The authors declare no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (81960398).
Copyright
© The Author(s) 2026.
References
-
2. Hoveidaei AH, Sadat-Shojai M, Nabavizadeh SS, Niakan R, Shirinezhad A, MosalamiAghili S, et al. Clinical challenges in bone tissue engineering - A narrative review. Bone. 2025;192:117363.[DOI]
-
7. Niu Z, He T, Peng S. Recent advances of 2D nanomaterials integrated 3D-printed scaffolds for bone repair and regeneration. BME Horizon. 2025;3(1):202416.[DOI]
-
10. Andrussow I, Sun H, Kuchenbecker KJ, Martius G. Minsight: A fingertip-sized vision-based tactile sensor for robotic manipulation. Adv Intell Syst. 2023;5(8):2300042.[DOI]
-
11. Zhang T, Wang W, Liu J, Wang L, Tang Y, Wang K. A review on magnesium alloys for biomedical applications. Front Bioeng Biotechnol. 2022;10:953344.[DOI]
-
14. Wang Y, Qin Q, Liu X, Li Q, Zhang H, Yang D, et al. Biocompatibility, degradation behavior, and mechanical properties of magnesium alloy plates in vivo. Appl Bionics Biomech. 2023;2023:4436396.[DOI]
-
15. Wang GQ, Li GF, Liu HF. Research progress in application and surface modification of medical degradable magnesium alloys. Surf Technol. 2024;53(7):15-30. Chinese.[DOI]
-
16. Jiang HF, Chen MF, Liu DB. Research progress of biodegradable magnesium alloys for fracture fixation and bone repair materials. Tianjin Yi Yao. 2020;48(7):682-688. Chinese.[DOI]
-
21. Wang Y, Qin Q, Liu X, Li Q, Zhang H, Yang D, et al. Biocompatibility, degradation behavior, and mechanical properties of magnesium alloy plates in vivo. Appl Bionics Biomech. 2023;2023:4436396.[DOI]
-
23. Wang Y, Liang W, Liu X, Li Q, Xie Y, Jiang Y. Osteogenesis and degradation behavior of magnesium alloy plate in vivo. Eur J Inflamm. 2021;19:20587392211034078.[DOI]
-
24. Cao W, Zhu S, Wang W, Wang Y, Yang S, Sun H. Microarc oxidation-PBAT composite coating on EK30 biodegradable magnesium alloys to enhance corrosion resistance and cytocompatibility. Colloids Surf B Biointerfaces. 2025;253:114720.[DOI]
-
25. Zhao C, Wen M, Wang Q, Ouyang W, Xu D, Jia Z, et al. Tailoring the corrosion resistance and biological performance of Mg-Zn-Y-Nd bioimplants with Multiphasic, pore-sealed cerium-doped ceramic coatings via facile one-pot plasma electrolytic oxidation. J Mater Sci Technol. 2025;230:60-79.[DOI]
-
26. Muhammad S, Batool F, Irfan M, Ahmad M. Metal/metal oxides/silsesquioxanes as fillers for functional polymer composites. BME Horizon. 2023;1(2):76.[DOI]
-
27. Fernández-Lizárraga M, García-López J, Rodil SE, Ribas-Aparicio RM, Silva-Bermudez P. Evaluation of the biocompatibility and osteogenic properties of metal oxide coatings applied by magnetron sputtering as potential biofunctional surface modifications for orthopedic implants. Materials. 2022;15(15):5240.
-
28. Zheng J, Rahman N, Li L, Zhang J, Tan H, Xue Y, et al. Biofunctionalization of electrospun fiber membranes by LbL-collagen/chondroitin sulfate nanocoating followed by mineralization for bone regeneration. Mater Sci Eng C. 2021;128:112295.[DOI]
-
33. Sun T, Wang J, Huang H, Liu X, Zhang J, Zhang W, et al. Low-temperature deposition manufacturing technology: A novel 3D printing method for bone scaffolds. Front Bioeng Biotechnol. 2023;11:1222102.[DOI]
-
40. Subramani M, Huang SJ, Borodianskiy K. Effect of SiC nanoparticles on AZ31 magnesium alloy. Materials. 2022;15(3):1004.[DOI]
-
42. Sarian MN, Iqbal N, Sotoudehbagha P, Razavi M, Ahmed QU, Sukotjo C, et al. Potential bioactive coating system for high-performance absorbable magnesium bone implants. Bioact Mater. 2022;12:42-63.[DOI]
-
44. Qi D, Su J, Li S, Zhu H, Cheng L, Hua S, et al. 3D printed magnesium-doped β-TCP gyroid scaffold with osteogenesis, angiogenesis, immunomodulation properties and bone regeneration capability in vivo. Biomater Adv. 2022;136:212759.[DOI]
-
49. Liang L, Lin Z, Duan Z, Agbedor SO, Li N, Baker I, et al. Enhancing the immunomodulatory osteogenic properties of Ti-Mg alloy by Mg2+-containing nanostructures. Regen Biomater. 2024;11:rbae104.[DOI]
-
50. Wang J, Wang M, Chen F, Wei Y, Chen X, Zhou Y, et al. Nano-hydroxyapatite coating promotes porous calcium phosphate ceramic-induced osteogenesis via BMP/smad signaling pathway. Int J Nanomed. 2019;14:7987-8000.[DOI]
-
52. Krumdieck SP, Boichot R, Gorthy R, Land JG, Lay S, Gardecka AJ, et al. Nanostructured TiO2 anatase-rutile-carbon solid coating with visible light antimicrobial activity. Sci Rep. 2019;9:1883.[DOI]
-
53. Nosrati H, Heydari M. Titanium dioxide nanoparticles: A promising candidate for wound healing applications. Burns Trauma. 2025;13:tkae069.[DOI]
-
55. Hatt LP, Wirth S, Ristaniemi A, Ciric DJ, Thompson K, Eglin D, et al. Micro-porous PLGA/β-TCP/TPU scaffolds prepared by solvent-based 3D printing for bone tissue engineering purposes. Regen Biomater. 2023;10:rbad084.[DOI]
-
56. Chakraborty Banerjee P, Al-Saadi S, Choudhary L, Harandi SE, Singh R. Magnesium implants: Prospects and challenges. Materials. 2019;12(1):136.[DOI]
-
57. He LY, Zhang XM, Liu B, Tian Y, Ma WH. Effect of magnesium ion on human osteoblast activity. Braz J Med Biol Res. 2016;49(7):e5257.[DOI]
-
58. Li S, Man Z, Zuo K, Zhang L, Zhang T, Xiao G, et al. Advancement in smart bone implants: The latest multifunctional strategies and synergistic mechanisms for tissue repair and regeneration. Bioact Mater. 2025;51:333-382.[DOI]
-
59. Gao H, Shen H, Zhang X, Liu Y, Shang Y, Sun S, et al. Revolutionizing neural regeneration with smart responsive materials: Current insights and future prospects. Bioact Mater. 2025;52:393-421.[DOI]
-
60. Dong J, Li Y, Lin P, Leeflang MA, van Asperen S, Yu K, et al. Solvent-cast 3D printing of magnesium scaffolds. Acta Biomater. 2020;114:497-514.[DOI]
-
62. Yazdanpanah Z, Johnston JD, Cooper DML, Chen X. 3D bioprinted scaffolds for bone tissue engineering: State-of-the-art and emerging technologies. Front Bioeng Biotechnol. 2022;10:824156.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite



