The solid electrolyte interface (SEI) in lithium batteries, defined as a nanoscale passivation layer formed by the electrolyte’s decomposition at anode-electrolyte interfaces, is crucial for battery performance[1-4]. The SEI composition can evolve during a resting stage[5] or a long cycling process[6], which is governed by different electrolyte compositions and has a great impact on battery performance. Mengdi et al. have reviewed this issue recently[7]. However, its characterization has always been challenging[8-10]. Transmission electron microscopy (TEM) and atomic force microscopy are common methods to revolve the thickness and hardness of SEI layers. Meanwhile, X-ray photoelectron spectroscopy (XPS) is widely used to identify the chemical species with a nanoscale resolution, especially providing the ability to identify Li element[11-13].
However, because of the intrinsic fragility of SEI layers, both its morphology and composition tend to be changed at room temperature (RT). This can be induced by solvent evaporation or electrochemical reactions[13,14]. This problem can be largely avoided with the help of cryogenic techniques, such as cryo-TEM[15-17]. However, as amorphous phases do not have a clear lattice fringe, the SEI components and valence states can hardly be determined under TEM, and just lie within the capabilities of XPS. Over the past thirty years, the application of XPS tests has been dramatically increased in SEI detection (Figure 1A), with in-situ/operando methods occasionally introduced. Also, the influence of ionic sputtering and X-ray beams has been considered with the gas cluster ion beam developed. However, coupling XPS with the cryogenic technique is much less reported (there are only dozens of publications (Figure 1B), even after taking all research areas into consideration), falling behind cryo-TEM. So, an accurate detection of SEI components and the influence of temperature on them are far from clear.
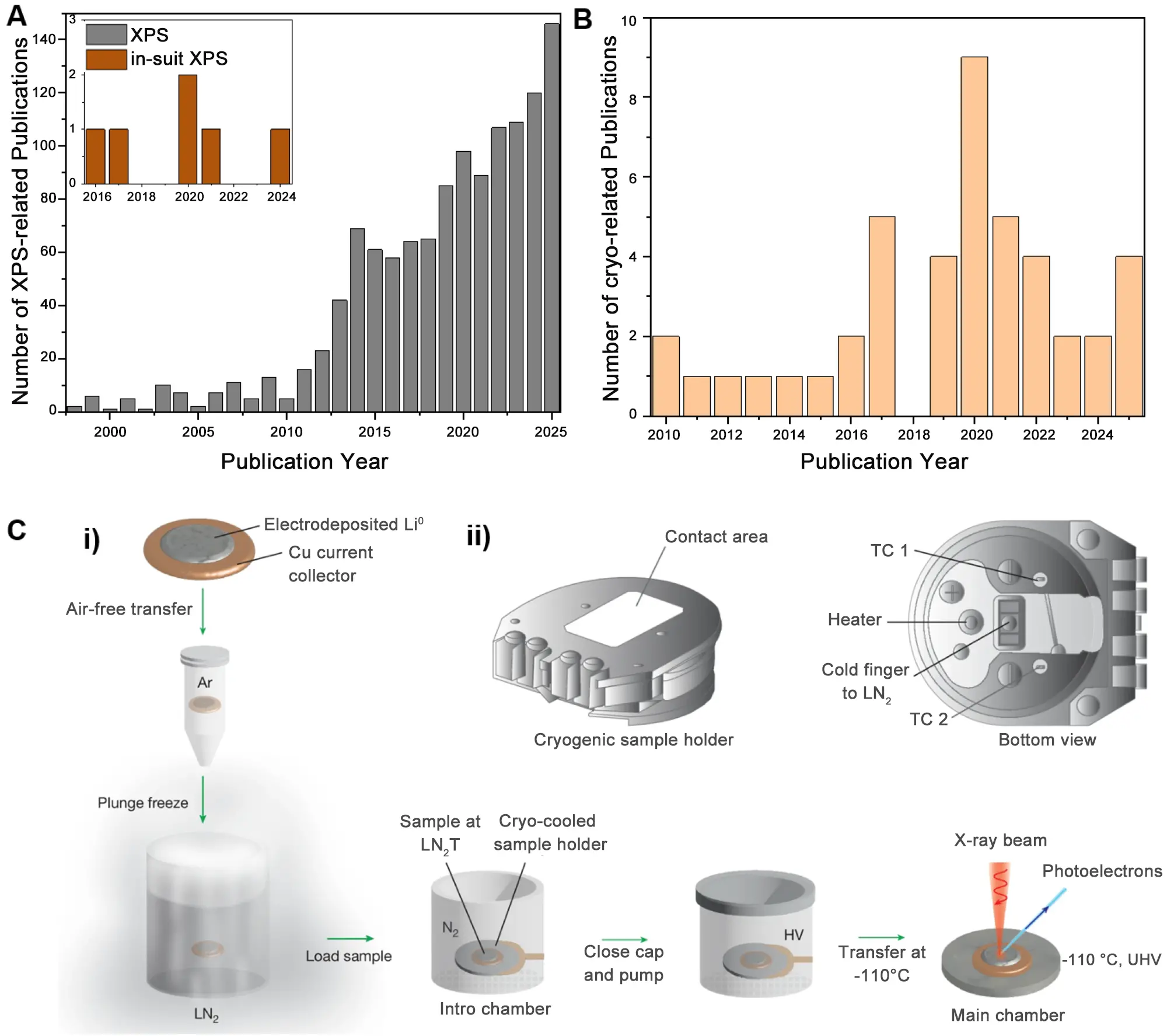
Figure 1. Development and application tendency of XPS techniques by statistics on the related publications. (A) Publications using the keywords “XPS” or “in situ XPS” together with “SEI”; (B) Publications using the keywords “cryo-XPS”; (C) Cryo-XPS setup: (i) sample preparation process for Cryo-XPS and (ii) cryogenic sample holder. Republished with permission from[18]. XPS: X-ray photoelectron spectroscopy; SEI: solid electrolyte interface.
To address the above issues, Shuchi et al.[18], inspired by cryo-electron microscopy (cryo-EM), developed a cryo-XPS method, publishing a report in Nature. The initial chemical species in SEI were carefully preserved by performing plunge freezing of the samples in liquid N2 (Figure 1C), and then XPS tests were conducted at -110 ℃. The low temperature effectively inhibits chemical reactions and volatile species, maintaining the real chemical state of the pristine SEI. The SEI evolution under the reaction effect, ultrahigh vacuum effect and beam effect were also systematically investigated. Moreover, a more precise relationship between SEI composition and battery performance could be built accordingly.
First of all, the influence of temperature was investigated by observing the evolution of SEI components in both cryo-XPS test and their RT-XPS (cryo-heated to RT) counterparts. Under cryo-XPS conditions, all F 1s, O 1s and N 1s spectra changed slightly over time. In contrast, once the temperature was raised to RT, significant changes happened and continued within 60 min. This phenomenon indicates that the composition of SEI can be stabilized at cryogenic temperature, but can evolve into a different one in RT. So, that the data obtained from cryo-XPS is more accurate than that from common RT-XPS. Later, the necessity of immediate plunge freezing and the reversibility of SEI evolution at RT were studied by re-cooling the SEI to cryo-temperature. The F 1s spectra reflect that the evolution of SEI chemical component is irreversible; therefore, it is necessary to perform an immediate plunge freezing.
Then, the reaction effect was investigated by comparing three XPS testing temperatures, including cryo-temperature, regular RT, and in-situ heating of the sample from cryo-temperature to RT. A localized high-concentration electrolyte was selected as a classical system. F 1s and O 1s signals were considered coming from LiF and Li2O, which are beneficial components for cycling performance. The peak intensity of LiF under both in-situ heating to RT and regular RT is significantly higher than that at cryo-temperature. It shows that the content of LiF is overestimated when utilizing an RT-XPS method. As the LiF amount is usually correlated with coulombic efficiency, RT-XPS would mislead their relationship. The increase of LiF content at RT mainly comes from a series of spontaneous decomposition reactions of certain SEI components and chemical corrosion of Li anode. In contrast, the peak of Li2O was clearly observed in cryo-XPS but almost undetectable in RT-XPS. This is mainly because the SEI reaction at RT caused Li2O to transform into other substances. As a consequence, the LiF and Li2O measured by RT-XPS deviated from the real contents in SEI, and therefore do not reflect the relationship between the SEI content and coulombic efficiency.
The ultrahigh vacuum (UHV) condition of the XPS chamber (≤ 10-6 Pa) can make SEI species volatilize and decompose, so that the SEI thickness will be reduced at RT. This UHV effect was observed by the potential Li0 peak at different XPS test temperatures. Compared to the Li 1s spectrum in cryo-XPS, there was a clear Li0 peak in RT-XPS. This is mainly due to the departure of volatile substances from the SEI, which makes the thickness of the SEI thinner at RT. Estimated from the photoelectron escape depth of the Li0 metal, the thickness of the SEI was ≤ 4.2 nm ± 20% at RT, while it was > 4.2 nm ± 20% at cryo-temperature. Additionally, the Li 1s spectrum also shows the number of chemical species inside SEI layers under different conditions, as most components in SEI form bonds with Li. So, a lower full-width half maximum (FWHM) in the spectrum corresponds to a more uniform composition in SEI, and vice versa. At cryo-temperature, the FWHM of the Li 1s spectrum of the SEI is higher than that at RT. It indicates the initial SEI is heterogeneous and has more components. In contrast, the SEI at RT is mainly composed of non-volatile substances and stable reaction products. This phenomenon was also reflected in the atomic concentration change with sputtering, when comparing a cryo-XPS test with the cryo-heated to RT-XPS test.
Considering that both electron and X-rays can cause beam damage to the samples, the authors also verified this beam effect in cryo-XPS. In cryo-TEM[19], the extent of beam damage was evaluated by comparing images before and after a long acquisition process. Similarly, five consecutive spectra at cryo-temperature and RT were collected in sequence. The intensity of F 1s peaks, which are attributed to LiF, only showed a slight increase at both cryo-temperature and RT. This proves that the beam damage effect has a minor impact on the SEI components. Furthermore, this LiF peak was compared between cryo-XPS and the cryo-heated to RT-XPS spectra from the same sample, where a more significant increase was observed once the temperature was raised than that observed among the five consecutive tests at RT. By this means, the beam effect can be decoupled from the reaction and UHV effects. Here, it shows that the evolution of SEI components at RT is mainly caused by the reaction and UHV effects, whereas the beam effect has only a slight influence. The scenario in TEM usually has a much more obvious beam damage. The authors attributed this difference to a low-energy X-ray beam (about 1.5 keV) in XPS and a high-energy electron beam (80-300 keV) in TEM. Moreover, Roberto et al.[20] suppressed this beam damage by reducing the X-ray dose in lab-based XPS, or by using a snapshot acquisition mode in synchrotron-based XPS.
Finally, the correlation between SEI composition and coulombic efficiency was provided, based on the unevolved SEI species from cryo-XPS. Different electrolyte chemistries were considered, which corresponded to the salt-, additive- and solvent-derived SEI. The (F + P)/C ratio was used for LiPF6 salt-based carbonate electrolytes, and the (F + S + N)/C ratio for LiFSI salt-based ether electrolytes. A Spearman coefficient (ρ) was used to measure the correlation between the ranked SEI composition and coulombic efficiency. It was found that the correlation was moderate (ρ = 0.6) when using the RT-XPS test, but highly positive (ρ = 0.9) when using the cryo-XPS test. Also, the correlation between F/C and CE was positive in cryo-XPS, confirming the beneficial impact of fluorinated species. However, it shows a moderate negative value in RT-XPS, which does not provide a rational result under this test condition. Moreover, the ratios and error ranges calculated from cryo-XPS tests are larger. This comes from the heterogeneity of the SEI components under cryo-XPS after effective preservation.
Overall, the work by Shuchi et al.[18] provides a cryo-XPS technique with an immediate plunge freezing method, which overcomes the limitations of RT-XPS in the study of SEI components. Both the problems of irreversible chemical composition changes and component volatilization under UHV conditions are avoided by this means. With the preserved SEI species obtained, the SEI content and columbic efficiency were found to have a significant positive correlation. This cryo-XPS test offers a new approach to get a more precise chemical composition of SEI layers and reliable correlations with battery performance, which may challenge most existing results. Also, this research will benefit more studies on sensitive SEI layers, as well as other issues in batteries (like interfacial phases etc.).
More broadly, the XPS technique faces bottleneck due to damages caused by not only the temperature, but also ionic sputtering and X-ray beams. Also, sample preparation is a critical issue when applying XPS to lithium batteries. It is still challenging to track the chemical state during a battery cycling process. Therefore, more integrated improvements are needed in the future, facilitating the study of broad areas (such as biology and pharmacy).
Authors contribution
Kong X: Data analysis, investigation, validation, writing-original draft.
Hao S: Conceptualization, project administration, supervision, validation, writing-review & editing.
All authors have given approval to the final version of the manuscript.
Conflicts of interest
All authors have no conflicts of interest to declare.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Funding
Financial support from the National Natural Science Foundation of China (22409099, 22579092) and the Fundamental Research Fund for the Central University, Nankai University (023/63253173) are gratefully acknowledged.
Copyright
© The Author(s) 2026.
References
-
1. Yu X, Manthiram A. Electrode–electrolyte interfaces in lithium-based batteries. Energy Environ Sci. 2018;11(3):527-543.[DOI]
-
2. Tan J, Matz J, Dong P, Shen J, Ye M. A growing appreciation for the role of LiF in the solid electrolyte interphase. Adv Energy Mater. 2021;11(16):2100046.[DOI]
-
3. Yao YX, Wan J, Liang NY, Yan C, Wen R, Zhang Q. Nucleation and growth mode of solid electrolyte interphase in Li-ion batteries. J Am Chem Soc. 2023;145(14):8001-8006.[DOI]
-
4. Chu L, Shi Y, Li Z, Sun C, Yan H, Ma J, et al. Solid electrolyte interphase on anodes in rechargeable lithium batteries. Nano Res. 2023;16(9):11589-11603.[DOI]
-
5. Yoon H, Kim D, Choi C, Oh JM, Shin J, Park H, et al. Unveiling the hidden interface: Pre-SEI governs Li morphology in anode-free Li metal batteries. Adv Mater. 2026;e21411.[DOI]
-
6. Fang Q, Xu S, Sha X, Liu D, Zhang X, Li W, et al. Interfacial degradation of silicon anodes in pouch cells. Energy Environ Sci. 2024;17(17):6368-6376.[DOI]
-
7. Hao M, Weng S, Zhong C, Li Y, Wang X. Structure and evolution of solid electrolyte interphase (SEI) at the electrode-electrolyte interface. Mater Today Energy. 2025;53:101998.[DOI]
-
8. Chae OB, Lucht BL. Interfacial issues and modification of solid electrolyte interphase for Li metal anode in liquid and solid electrolytes. Adv Energy Mater. 2023;13(14):2203791.[DOI]
-
9. Shen Z, Huang J, Xie Y, Wei D, Chen J, Shi Z. Solid electrolyte interphase on lithium metal anodes. ChemSusChem. 2024;17(11):e202301777.[DOI]
-
10. Huang S, Cheong LZ, Wang D, Shen C. Thermal stability of solid electrolyte interphase of lithium-ion batteries. Appl Surf Sci. 2018;454:61-67.[DOI]
-
11. Ushakova EE, Frolov A, Reveguk AA, Usachov DY, Itkis DM, Yashina LV. Solid electrolyte interface formation between lithium and PEO-based electrolyte. Appl Surf Sci. 2022;589:153014.[DOI]
-
12. Nojabaee M, Küster K, Starke U, Popovic J, Maier J. Solid electrolyte interphase evolution on lithium metal in contact with glyme-based electrolytes. Small. 2020;16(23):2000756.[DOI]
-
13. Oyakhire ST, Gong H, Cui Y, Bao Z, Bent SF. An X-ray photoelectron spectroscopy primer for solid electrolyte interphase characterization in lithium metal anodes. ACS Energy Lett. 2022;7(8):2540-2546.[DOI]
-
14. Quinn J, Wu B, Xu Y, Engelhard MH, Xiao J, Wang C. Tracking the oxidation of silicon anodes using cryo-EELS upon battery cycling. ACS Nano. 2022;16(12):21063-21070.[DOI]
-
15. Zhang Z, Cui Y, Vila R, Li Y, Zhang W, Zhou W, et al. Cryogenic electron microscopy for energy materials. Acc Chem Res. 2021;54(18):3505-3517.[DOI]
-
16. Zachman MJ, Tu Z, Choudhury S, Archer LA, Kourkoutis LF. Cryo-STEM mapping of solid–liquid interfaces and dendrites in lithium-metal batteries. Nature. 2018;560(7718):345-349.[DOI]
-
17. Wang X, Zhang M, Alvarado J, Wang S, Sina M, Lu B, et al. New insights on the structure of electrochemically deposited lithium metal and its solid electrolyte interphases via cryogenic TEM. Nano Lett. 2017;17(12):7606-7612.[DOI]
-
18. Shuchi SB, D’Acunto G, Sayavong P, Oyakhire ST, Sanroman Gutierrez KM, Risner-Jamtgaard J, et al. Cryogenic X-ray photoelectron spectroscopy for battery interfaces. Nature. 2025;646(8086):850-855.[DOI]
-
19. He Y, Jiang L, Chen T, Xu Y, Jia H, Yi R, et al. Progressive growth of the solid–electrolyte interphase towards the Si anode interior causes capacity fading. Nat Nanotechnol. 2021;16(10):1113-1120.[DOI]
-
20. Fantin R, Van Roekeghem A, Rueff JP, Benayad A. Surface analysis insight note: Accounting for X-ray beam damage effects in positive electrode-electrolyte interphase investigations. Surf Interface Anal. 2024;56(6):353-358.[DOI]
Copyright
© The Author(s) 2026. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Publisher’s Note
Share And Cite



