Anatomically porous-media heat transfer modeling for multi-organ supercooling perfusion cryopreservation
-
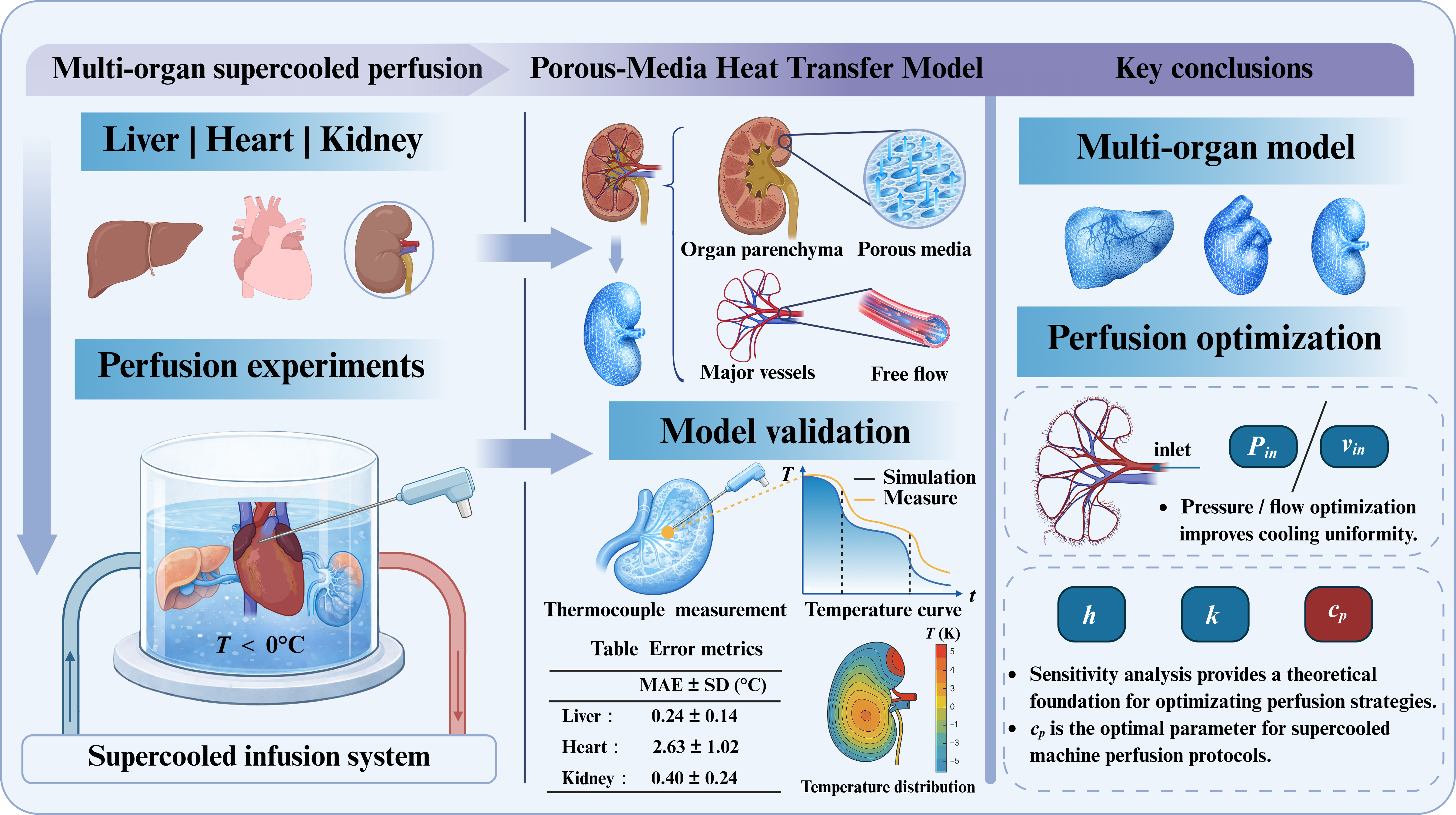
Supercooling perfusion extends organ-preservation time by maintaining grafts ice-free below 0 °C, but thermal non-uniformity and limited intra-organ temperature observability hinder protocol design, especially at large-organ scales. We developed ...
MoreSupercooling perfusion extends organ-preservation time by maintaining grafts ice-free below 0 °C, but thermal non-uniformity and limited intra-organ temperature observability hinder protocol design, especially at large-organ scales. We developed an anatomically based thermo-fluidic modeling framework for supercooled perfusion of the liver, heart, and kidney in a recirculating multi-organ configuration and validated the model experimentally. Three-dimensional organ geometries from the BodyParts3D repository were combined with a porous-media tissue representation and realistic perfusion boundary conditions to resolve transient intra-parenchymal temperature fields. A self-developed variable-frequency supercooled machine perfusion (MP) platform was used to measure temperatures in porcine livers, hearts, and kidneys using multiple thermocouples placed at anatomically corresponding locations. Simulated temperature trajectories agreed with measurements across organs, with mean absolute errors of 0.24 °C for the liver, 2.63 °C for the heart, and 0.4 °C for the kidney, and reproduced initial cooling followed by progressive approach to the perfusate temperature and stabilization. Spatial temperature maps captured organ-specific gradients consistent with convective heat extraction by perfusate delivery and conductive transport within tissue. Using the validated model, we performed parametric sweeps of the inlet perfusion parameter, perfusate thermophysical properties, and external convective heat-transfer coefficient to quantify their effects on cooling rate and temperature uniformity. Based on quantitative metrics, these parameters were found to influence cooling rate and intra-organ temperature uniformity to different degrees, while the magnitude of improvement differed among organs due to size and vascular characteristics. This study provides a validated, under the tested conditions, tool to predict intra-organ temperature evolution and a guide for thermodynamically optimizing supercooled MP protocols in multi-organ preservation.
Less -
Zaize Liu, ... Wei Rao
-
DOI: https://doi.org/10.70401/tx.2026.0018 - April 20, 2026
-
This article belongs to the Special Issue Recent Advances in Bio-thermophysics: Innovations and Development at the Intersection of Biology and Thermal Science