SIRT3 at the crossroads of ferroptosis: Multidimensional regulation of the mitochondrial deacetylase Sirtuin 3 (SIRT3) on ferroptosis
-
Ferroptosis, a regulated cell death modality, driven by iron-dependent lipid peroxidation, is intrinsically coupled with mitochondrial metabolic turbulence and redox dysregulation. While the mitochondrial sirtuin Sirtuin 3 (SIRT3) is canonically viewed ...
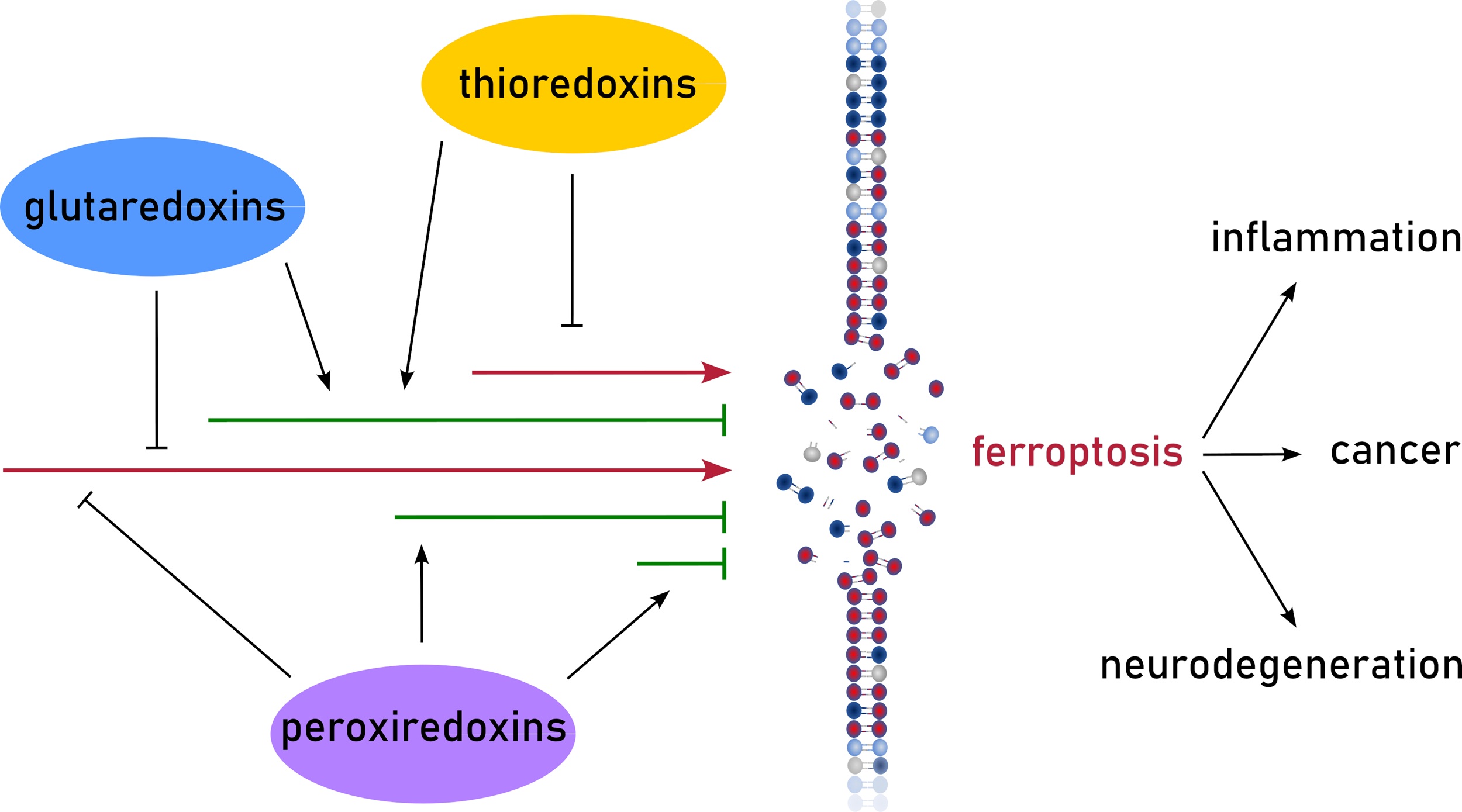
MoreFerroptosis, a regulated cell death modality, driven by iron-dependent lipid peroxidation, is intrinsically coupled with mitochondrial metabolic turbulence and redox dysregulation. While the mitochondrial sirtuin Sirtuin 3 (SIRT3) is canonically viewed as a master regulator of energy homeostasis, its defensive repertoire against ferroptosis extends far beyond the simplistic activation of antioxidant enzymes. In this review, we synthesize emerging evidence to construct an integrated “metabolic-structural” defense model orchestrated by SIRT3. We first delineate how SIRT3 functions as a metabolic rheostat, rewiring tricarboxylic acid (TCA) cycle flux via the deacetylation of isocitrate dehydrogenase 2 (IDH2) to sustain the nicotinamide adenine dinucleotide phosphate (NADPH)/glutathione (GSH) reservoir. Breaking away from the classical enzymatic paradigm, we highlight a novel non-enzymatic substrate regulatory mechanism where SIRT3 stabilizes the glutamate transporter SLC25A22 through specific deacetylation-ubiquitination crosstalk, thereby limiting ferroptotic susceptibility. Furthermore, we expand the SIRT3 signaling landscape by proposing a “SIRT3-nuclear factor erythroid 2-related factor 2 (Nrf2) deacetylation axis” that bridges mitochondrial stress signals to nuclear transcriptional defense, and by detailing its control over iron entry via the iron regulatory protein 1 (IRP1)-transferrin receptor 1 (TfR1) pathway. At the organelle level, we examine how SIRT3 remodels mitochondrial dynamics, upregulating optic atrophy-associated protein 1 (OPA1) while suppressing dynamin-related protein 1 (DRP1), to construct a “fusion network barrier” that dilutes ROS toxicity. We also posit a critical hypothesis: SIRT3 safeguards the integrity of mitochondria-associated endoplasmic reticulum membranes, preventing structural decoupling and calcium overload that triggers ferroptotic sensitization. Finally, we reconcile the context-dependent duality of SIRT3 in cancer and degenerative diseases, outlining future therapeutic strategies that leverage these multidimensional vulnerabilities.
Less -
Yixuan Chen, ... Rong Cai
-
DOI: https://doi.org/10.70401/fos.2026.0026 - April 20, 2026