Table of Contents
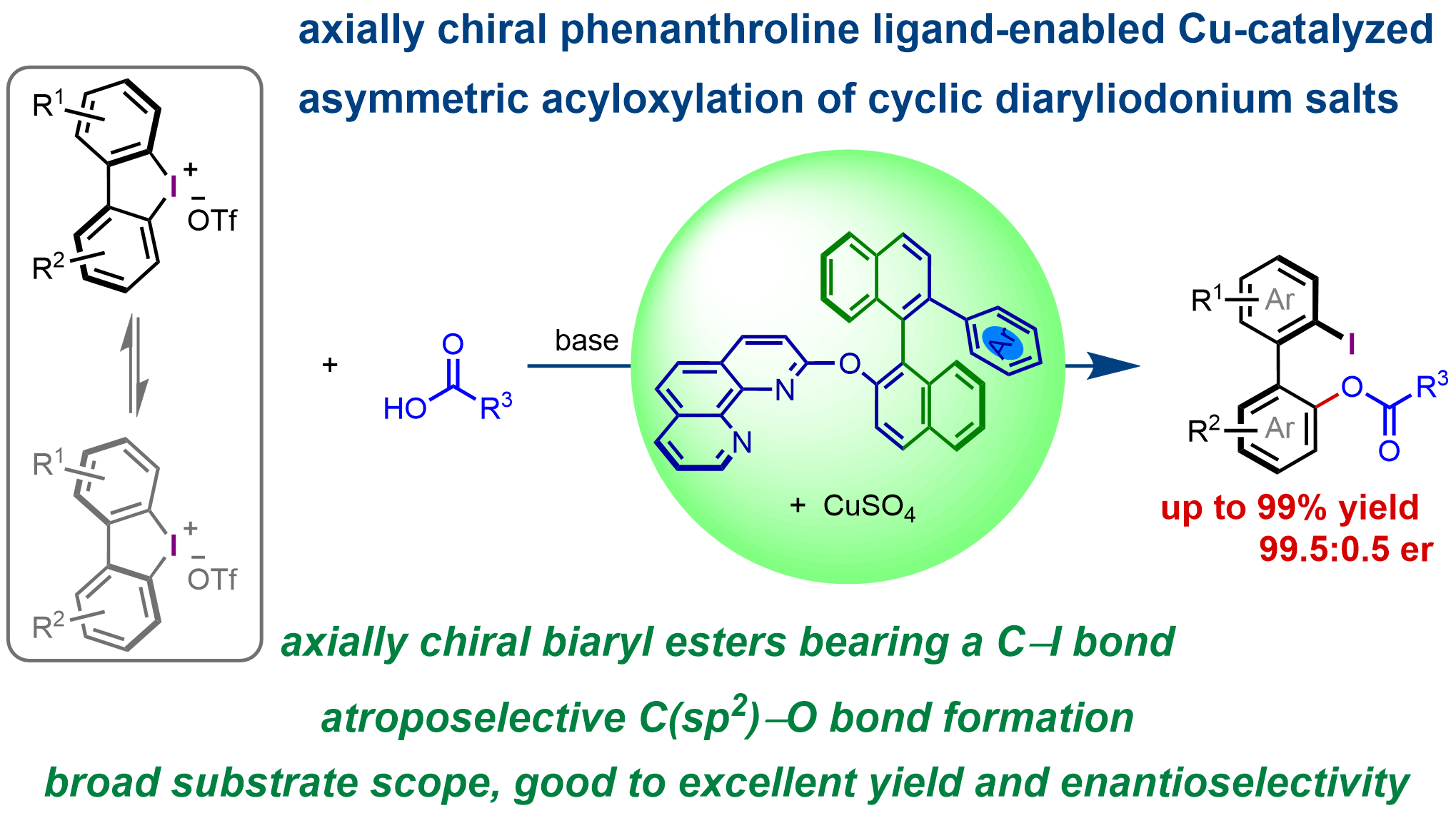
Axially chiral phenanthroline ligand-enabled Cu-catalyzed asymmetric acyloxylation of cyclic biaryliodonium salts
Chiral ligands play a crucial role in enantioselective transition-metal catalysis, not only by coordinating to metal centers and modulating their electronic properties, but also by providing the essential chiral environment for efficient enantiodiscrimination. ...
More.Chiral ligands play a crucial role in enantioselective transition-metal catalysis, not only by coordinating to metal centers and modulating their electronic properties, but also by providing the essential chiral environment for efficient enantiodiscrimination. For a given reaction, high enantioselectivities can often be achieved with different chiral ligands, offering valuable enantioinduction models, even when these ligands possess distinct structures and chiral environments. Herein, we report that a class of conformationally flexible C1-symmetric axially chiral phenanthroline ligands enables the formation of chiral copper catalysts capable of promoting the highly enantioselective ring-opening acyloxylation of cyclic biaryliodonium salts. Using this approach, a range of valuable axially chiral biaryl compounds bearing an ester group and a carbon–iodine bond were synthesized in good to excellent yield (up to 99%), with good to excellent enantioselectivity (up to 99.5:0.5 er).
Less.Shouyi Cen, ... Zhipeng Zhang
DOI:https://doi.org/10.70401/cc.2026.0023 - April 17, 2026
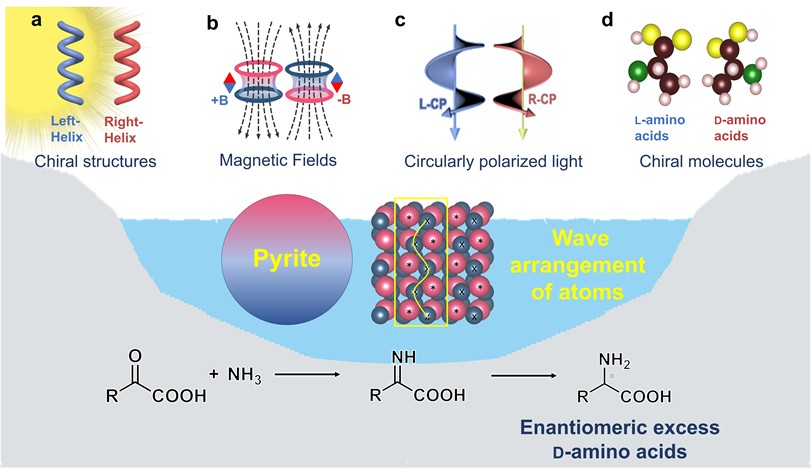
Pyrite interference in prebiotic chiral selection of amino acids
L-amino acids are ubiquitous in biology, yet the origin of their homochirality is an enduring mystery. Prebiotic chiral selection process of amino acids could significantly influence subsequent molecular evolution and the emergence of life. However, the ...
More.L-amino acids are ubiquitous in biology, yet the origin of their homochirality is an enduring mystery. Prebiotic chiral selection process of amino acids could significantly influence subsequent molecular evolution and the emergence of life. However, the distribution of chirality of amino acids remains convoluted, which poses obstacles to the in-depth study of prebiotic chiral selection of amino acids. Here, we propose that minerals in the prebiotic environment might have hindered the absolute selection of L-amino acids to induce a random distribution of L- and D-amino acids during the early stages of life’s evolution. Pyrite, abundant in the prebiotic world, predominantly selected D-amino acids in the photocatalytic reductive amination of α-keto acids, resisting the enantioselectivity of chiral driving forces like chiral structures, magnetic fields, circularly polarized light, and chiral molecules. This resistance is due to the wavy atomic arrangement of pyrite surfaces, which causes larger reaction energy differences between enantiomers. Our findings provide evidence to assess the validity of prebiotic chiral selection scenarios for biomolecules, an important step in understanding the origin of life.
Less.Ruiqi Li, ... Yuxi Fang
DOI:https://doi.org/10.70401/cc.2026.0021 - April 10, 2026
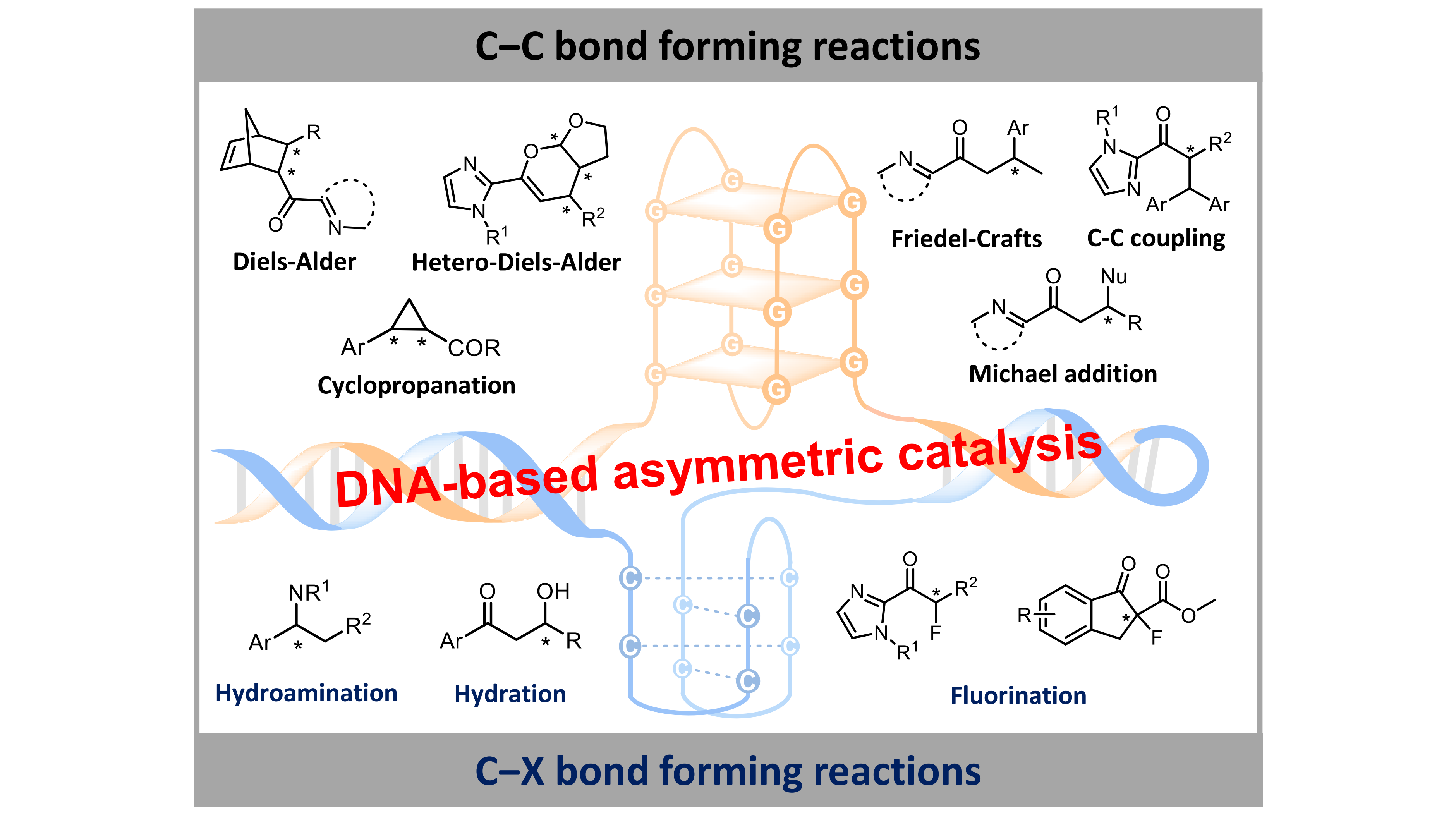
Programmable DNA as stereocontrol ligand for asymmetric catalysis in aqueous media
Owing to its intrinsic chirality, programmability and structural diversity, DNA functions as an effective chiral ligand for asymmetric catalysis in water. DNA hybrid catalysts are constructed by anchoring metal species into DNA scaffolds through covalent ...
More.Owing to its intrinsic chirality, programmability and structural diversity, DNA functions as an effective chiral ligand for asymmetric catalysis in water. DNA hybrid catalysts are constructed by anchoring metal species into DNA scaffolds through covalent or noncovalent strategies. These DNA-based catalytic systems exhibit potent reactivity and high enantioselectivity in various C–C, C–N, C–O, and C–F bond formation reactions in aqueous media. Importantly, their catalytic activity and product stereochemistry can be precisely directed by modulating DNA sequences, tertiary structures, metal species, and host-guest interaction. This review highlights recent advances in DNA-mediated asymmetric catalysis and discusses future prospects toward tailored stereocontrol under aqueous conditions.
Less.Shanmei Xu, ... Changhao Wang
DOI:https://doi.org/10.70401/cc.2026.0022 - April 10, 2026
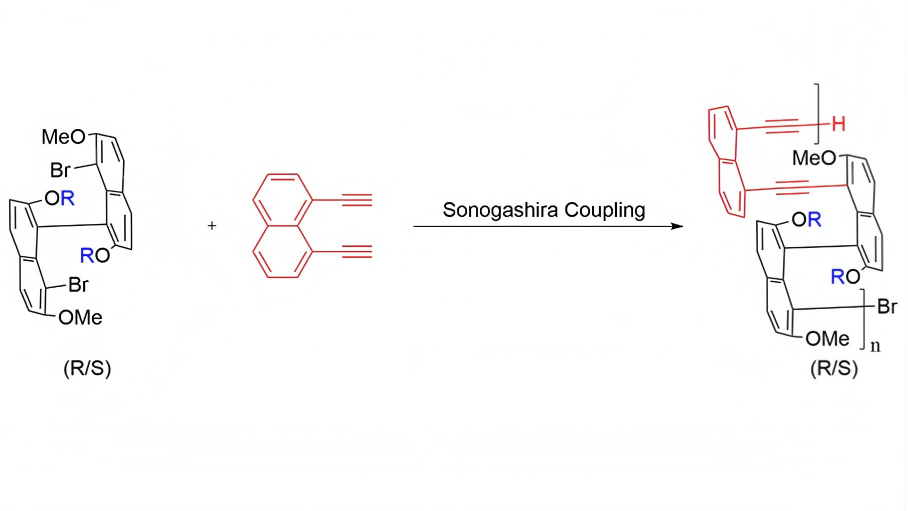
Enantiopure chiral multilayer polymers – design, asymmetric synthesis and property study
Axially chiral binaphthyl scaffolds provide a robust platform for encoding permanent stereochemistry into conjugated polymer backbones while promoting nonplanar organization and higher-order aggregation. Building on our group’s earlier development ...
More.Axially chiral binaphthyl scaffolds provide a robust platform for encoding permanent stereochemistry into conjugated polymer backbones while promoting nonplanar organization and higher-order aggregation. Building on our group’s earlier development of multilayer 3D folding polymers and related achiral/chiral multilayer polymer series, we designed and synthesized the first enantiopure chiral multilayer polymers from enantiopure binaphthyl-derived monomers via CuI-assisted, Pd-catalyzed Sonogashira polycondensation. To probe structure–property relationships, the monomer library was designed to contrast flexible alkoxy-substituted systems with a conformationally constrained tethered analogue. The resulting polymers were obtained in moderate yields, with number-average molecular weights on the order of 103-104 g mol-1 and relatively narrow dispersities. UV–visible spectra showed the expected aromatic π-π* absorptions. Photoluminescence measurements in tetrahydrofuran (THF)/H2O mixtures displayed a nonmonotonic solvent-composition dependence, with emission enhancement at intermediate water fractions followed by quenching in more water-rich mixtures. Dynamic light scattering indicated the presence of light-scattering dispersed aggregates in CHCl3 under the tested conditions, while scanning electron microscopy revealed hierarchical agglomerates in the dried state, in which submicrometer particulates coalesced into micrometer-scale clusters. Together, these results show that axial stereochemistry and aggregation/packing behavior cooperatively influence radiative and nonradiative decay pathways in chiral multilayer backbones, providing useful design guidance for aggregation-responsive emissive polymer materials.
Less.Hao Liu, ... Guigen Li
DOI:https://doi.org/10.70401/cc.2026.0020 - April 08, 2026
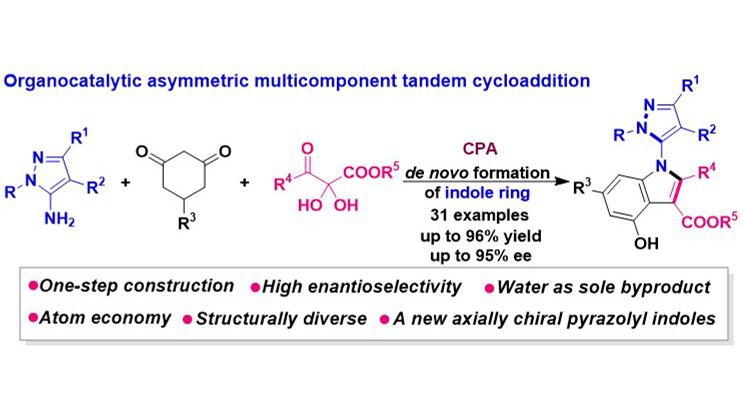
Catalytic atroposelective synthesis of C–N axially chiral 5-pyrazolyl indoles via multicomponent tandem cycloaddition
An organocatalytic asymmetric multicomponent tandem cycloaddition has been established for the construction of C–N axially chiral 5-pyrazolyl indole frameworks, which represent a novel class of five-five-membered indole scaffolds. Utilizing 5-aminopyrazoles, ...
More.An organocatalytic asymmetric multicomponent tandem cycloaddition has been established for the construction of C–N axially chiral 5-pyrazolyl indole frameworks, which represent a novel class of five-five-membered indole scaffolds. Utilizing 5-aminopyrazoles, cyclohexanediones, and 2,3-diketoester precursors, this methodology constructs the indole ring de novo in a single step, demonstrating high atom- and step-economy with water as the exclusive byproduct. The protocol enabled the efficient and high-enantioselective synthesis of structurally diverse C–N axially chiral 5-pyrazolyl indoles. This achievement not only introduces a new member to the family of axially chiral pyrazolyl indoles but also provides an efficient and novel strategy for their construction.
Less.Yu-Yu Chen, ... Feng Shi
DOI:https://doi.org/10.70401/cc.2026.0018 - April 02, 2026